Abstract
Cystic fibrosis is an autosomal recessive, monogenetic disorder caused by mutations in the cystic fibrosis transmembrane conductance regulator (CFTR) gene. The gene defect was first described 25 years ago and much progress has been made since then in our understanding of how CFTR mutations cause disease and how this can be addressed therapeutically. CFTR is a transmembrane protein that transports ions across the surface of epithelial cells. CFTR dysfunction affects many organs; however, lung disease is responsible for the vast majority of morbidity and mortality in patients with cystic fibrosis. Prenatal diagnostics, newborn screening and new treatment algorithms are changing the incidence and the prevalence of the disease. Until recently, the standard of care in cystic fibrosis treatment focused on preventing and treating complications of the disease; now, novel treatment strategies directly targeting the ion channel abnormality are becoming available and it will be important to evaluate how these treatments affect disease progression and the quality of life of patients. In this Primer, we summarize the current knowledge, and provide an outlook on how cystic fibrosis clinical care and research will be affected by new knowledge and therapeutic options in the near future. For an illustrated summary of this Primer, visit: http://go.nature.com/4VrefN
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 1 digital issues and online access to articles
$99.00 per year
only $99.00 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Weiler, C. A. & Drumm, M. L. Genetic influences on cystic fibrosis lung disease severity. Front. Pharmacol. 4, 40 (2013).
Jarvi, K. et al. Cystic fibrosis transmembrane conductance regulator and obstructive azoospermia. Lancet 345, 1578 (1995).
Gonska, T. et al. Role of cystic fibrosis transmembrane conductance regulator in patients with chronic sinopulmonary disease. Chest 142, 996–1004 (2012).
Wilschanski, M. et al. Mutations in the cystic fibrosis transmembrane regulator gene and in vivo transepithelial potentials. Am. J. Respir. Crit. Care Med. 174, 787–794 (2006).
Riordan, J. R. et al. Identification of the cystic fibrosis gene: cloning and characterization of complementary DNA. Science 245, 1066–1073 (1989).
Rommens, J. M. et al. Identification of the cystic fibrosis gene: chromosome walking and jumping. Science 245, 1059–1065 (1989).
Ratjen, F. & Grasemann, H. New therapies in cystic fibrosis. Curr. Pharm. Des. 18, 614–627 (2012).
O'Sullivan, B. P. & Freedman, S. D. Cystic fibrosis. Lancet 373, 1891–1904 (2009).
Walters, S., M. A . in Hodson and Geddes' Cystic Fibrosis 3rd edn (eds Hodson, M. E., Geddes D. M., Bush, A. ) 21–45 (Edward Arnold Ltd, 2007).
Farrell, P. M. The prevalence of cystic fibrosis in the European Union. J. Cyst. Fibros. 7, 450–453 (2008).
Festini, F. et al. Incidence of cystic fibrosis in the Albanian population. Pediatr. Pulmonol. 43, 1124–1129 (2008).
Festini, F., Taccetti, G., Cioni, M. L., Repetto, T. & De Martino, M. High incidence of cystic fibrosis in children born in Italy to Albanian immigrants. Thorax 58, 93 (2003).
Dupuis, A., Hamilton, D., Cole, D. E. & Corey, M. Cystic fibrosis birth rates in Canada: a decreasing trend since the onset of genetic testing. J. Pediatr. 147, 312–315 (2005).
Kulich, M., Rosenfeld, M., Goss, C. H. & Wilmott, R. Improved survival among young patients with cystic fibrosis. J. Pediatr. 142, 631–636 (2003).
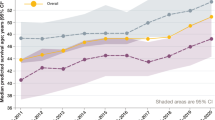
MacKenzie, T. et al. Longevity of patients with cystic fibrosis in 2000 to 2010 and beyond: survival analysis of the cystic fibrosis foundation patient registry. Ann. Intern. Med. 161, 233–241 (2014).
Cystic Fibrosis Foundation Cystic Fibrosis Foundation Patient Registry 2012 annual data report. (Cystic Fibrosis Foundation, 2013).
Dodge, J. A., Lewis, P. A., Stanton, M. & Wilsher, J. Cystic fibrosis mortality and survival in the UK: 1947–2003. Eur. Respir. J. 29, 522–526 (2007). References 15 and 17 show the improvements in survival in the US and the UK cystic fibrosis populations, respectively, over the past decade.
Cystic Fibrosis Canada. Canadian cystic fibrosis patient data registry report 2010. (Cystic Fibrosis Canada, 2010).
Kerem, E. et al. Factors associated with FEV1 decline in cystic fibrosis: analysis of the ECFS patient registry. Eur. Respir. J. 43, 125–133 (2014).
Rodman, D. M. et al. Late diagnosis defines a unique population of long-term survivors of cystic fibrosis. Am. J. Respir. Crit. Care Med. 171, 621–626 (2005).
Nick, J. A. et al. Effects of gender and age at diagnosis on disease progression in long-term survivors of cystic fibrosis. Am. J. Respir. Crit. Care Med. 182, 614–626 (2010).
Simmonds, N. J. et al. Cystic fibrosis and survival to 40 years: a study of cystic fibrosis transmembrane conductance regulator function. Eur. Respir. J. 37, 1076–1082 (2011).
George, P. M. et al. Improved survival at low lung function in cystic fibrosis: cohort study from 1990 to 2007. BMJ 342, d1008 (2011).
Accurso, F. J. et al. Effect of VX-770 in persons with cystic fibrosis and the G551D-CFTR mutation. N. Engl. J. Med. 363, 1991–2003 (2010).
Ramsey, B. W. et al. A CFTR potentiator in patients with cystic fibrosis and the G551D mutation. N. Engl. J. Med. 365, 1663–1672 (2011). This article shows the efficacy of CFTR modulation by the potentiator ivacaftor in patients with the G551D mutation.
Kerem, B. et al. Identification of the cystic fibrosis gene: genetic analysis. Science 245, 1073–1080 (1989).
Higgins, C. F. ABC transporters: from microorganisms to man. Annu. Rev. Cell Biol. 8, 67–113 (1992).
Klein, I., Sarkadi, B. & Varadi, A. An inventory of the human ABC proteins. Biochim. Biophys. Acta 1461, 237–262 (1999).
Collins, F. S. Cystic fibrosis: molecular biology and therapeutic implications. Science 256, 774–779 (1992).
Serohijos, A. W. et al. Phenylalanine-508 mediates a cytoplasmic-membrane domain contact in the CFTR 3D structure crucial to assembly and channel function. Proc. Natl Acad. Sci. USA 105, 3256–3261 (2008).
Drumm, M. L. et al. Genetic modifiers of lung disease in cystic fibrosis. N. Engl. J. Med. 353, 1443–1453 (2005).
Chillon, M. et al. Mutations in the cystic fibrosis gene in patients with congenital absence of the vas deferens. N. Engl. J. Med. 332, 1475–1480 (1995).
Sharer, N. et al. Mutations of the cystic fibrosis gene in patients with chronic pancreatitis. N. Engl. J. Med. 339, 645–652 (1998).
Cohn, J. A. et al. Relation between mutations of the cystic fibrosis gene and idiopathic pancreatitis. N. Engl. J. Med. 339, 653–658 (1998).
Sosnay, P. R. et al. Defining the disease liability of variants in the cystic fibrosis transmembrane conductance regulator gene. Nat. Genet. 45, 1160–1167 (2013).
Welsh, M. J. & Smith, A. E. Molecular mechanisms of CFTR chloride channel dysfunction in cystic fibrosis. Cell 73, 1251–1254 (1993).
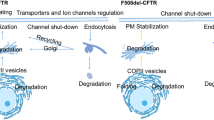
Okiyoneda, T. et al. Peripheral protein quality control removes unfolded CFTR from the plasma membrane. Science 329, 805–810 (2010).
Mendoza, J. L. et al. Requirements for efficient correction of DeltaF508 CFTR revealed by analyses of evolved sequences. Cell 148, 164–174 (2012).
Rabeh, W. M. et al. Correction of both NBD1 energetics and domain interface is required to restore ΔF508 CFTR folding and function. Cell 148, 150–163 (2012). References 38 and 39 delineate the importance of two distinct molecular defects conferred by the F508del. CFTR mutation, including their effect of correcting CFTR misfolding to restore CFTR expression. These papers show that to fully restore mutated CFTR to the cell surface, both NBD1 stability and interdomain assembly must be addressed.
Green, D. M. et al. Heritability of respiratory infection with Pseudomonas aeruginosa in cystic fibrosis. J. Pediatr. 161, 290–295 e1 (2012).
Garred, P. et al. Association of mannose-binding lectin gene heterogeneity with severity of lung disease and survival in cystic fibrosis. J. Clin. Invest. 104, 431–437 (1999).
Sun, L. et al. Multiple apical plasma membrane constituents are associated with susceptibility to meconium ileus in individuals with cystic fibrosis. Nat. Genet. 44, 562–569 (2012).
Blackman, S. M. et al. Genetic modifiers of cystic fibrosis-related diabetes. Diabetes 62, 3627–3635 (2013).
Dorfman, R. et al. Modifier gene study of meconium ileus in cystic fibrosis: statistical considerations and gene mapping results. Hum. Genet. 126, 763–778 (2009).
Romi, H. et al. Meconium ileus caused by mutations in GUCY2C, encoding the CFTR-activating guanylate cyclase 2C. Am. J. Hum. Genet. 90, 893–899 (2012).
Li, W. et al. Unraveling the complex genetic model for cystic fibrosis: pleiotropic effects of modifier genes on early cystic fibrosis-related morbidities. Hum. Genet. 133, 151–161 (2014).
Wright, F. A. et al. Genome-wide association and linkage identify modifier loci of lung disease severity in cystic fibrosis at 11p13 and 20q13.2. Nat. Genet. 43, 539–546 (2011).
Collaco, J. M., Blackman, S. M., McGready, J., Naughton, K. M. & Cutting, G. R. Quantification of the relative contribution of environmental and genetic factors to variation in cystic fibrosis lung function. J. Pediatr. 157, 802–807 e1–3 (2010).
Vanscoy, L. L. et al. Heritability of lung disease severity in cystic fibrosis. Am. J. Respir. Crit. Care Med. 175, 1036–1043 (2007).
Kolbe, E. W. et al. CLCA4 variants determine the manifestation of the cystic fibrosis basic defect in the intestine. Eur. J. Hum. Genet. 21, 691–694 (2013).
Bremer, L. A. et al. Interaction between a novel TGFB1 haplotype and CFTR genotype is associated with improved lung function in cystic fibrosis. Hum. Mol. Genet. 17, 2228–2237 (2008).
Yarden, J. et al. Association of tumour necrosis factor alpha variants with the CF pulmonary phenotype. Thorax 60, 320–325 (2005).
Goss, C. H., Newsom, S. A., Schildcrout, J. S., Sheppard, L. & Kaufman, J. D. Effect of ambient air pollution on pulmonary exacerbations and lung function in cystic fibrosis. Am. J. Respir. Crit. Care Med. 169, 816–821 (2004).
Collaco, J. M. et al. Interactions between secondhand smoke and genes that affect cystic fibrosis lung disease. JAMA 299, 417–424 (2008).
Martin, B. et al. Comparison of the US and Australian cystic fibrosis registries: the impact of newborn screening. Pediatrics 129, e348–e355 (2012).
Boyle, M. P., Sabadosa, K. A., Quinton, H. B., Marshall, B. C. & Schechter, M. S. Key findings of the US Cystic Fibrosis Foundation's clinical practice benchmarking project. BMJ Qual. Saf. 23 (Suppl. 1), i15–i22 (2014). This article reports the key outcomes of the quality improvement programme of cystic fibrosis care in the United States and shows the assessment of patterns of practice in the top-performing (20%) treatment centres, and provides insight into the features of practice that could lead to optimization of patient outcomes.
Ooi, C. Y. & Durie, P. R. Cystic fibrosis transmembrane conductance regulator (CFTR) gene mutations in pancreatitis. J. Cyst. Fibros. 11, 355–362 (2012).
Ratjen, F. & Doring, G. Cystic fibrosis. Lancet 361, 681–689 (2003).
Rowe, S. M., Miller, S. & Sorscher, E. J. Cystic fibrosis. N. Engl. J. Med. 352, 1992–2001 (2005).
Boucher, R. C. Airway surface dehydration in cystic fibrosis: pathogenesis and therapy. Annu. Rev. Med. 58, 157–170 (2007).
Adebamiro, A., Cheng, Y., Rao, U. S., Danahay, H. & Bridges, R. J. A segment of γ-ENaC mediates elastase activation of Na+ transport. J. Gen. Physiol. 130, 611–629 (2007).
Caldwell, R. A., Boucher, R. C. & Stutts, M. J. Neutrophil elastase activates near-silent epithelial Na+ channels and increases airway epithelial Na+ transport. Am. J. Physiol. Lung Cell. Mol. Physiol. 288, L813–819 (2005).
Passero, C. J. et al. TMPRSS4-dependent activation of the epithelial sodium channel requires cleavage of the γ-subunit distal to the furin cleavage site. Am. J. Physiol. Renal Physiol. 302, F1–8 (2012).
Matsui, H. et al. Evidence for periciliary liquid layer depletion, not abnormal ion composition, in the pathogenesis of cystic fibrosis airways disease. Cell 95, 1005–1015 (1998).
Button, B. et al. A periciliary brush promotes the lung health by separating the mucus layer from airway epithelia. Science 337, 937–941 (2012).
Chen, J. H. et al. Loss of anion transport without increased sodium absorption characterizes newborn porcine cystic fibrosis airway epithelia. Cell 143, 911–923 (2010).
Tuggle, K. L. et al. Characterization of defects in ion transport and tissue development in cystic fibrosis transmembrane conductance regulator (CFTR)-knockout rats. PLoS ONE 9, e91253 (2014).
Itani, O. A. et al. Human cystic fibrosis airway epithelia have reduced Cl− conductance but not increased Na+ conductance. Proc. Natl Acad. Sci. USA 108, 10260–10265 (2011).
Birket, S. E. et al. A functional anatomic defect of the cystic fibrosis airway. Am. J. Respir. Crit. Care Med. 190, 421–432 (2014).
Hoegger, M. J. et al. Cystic fibrosis. Impaired mucus detachment disrupts mucociliary transport in a piglet model of cystic fibrosis. Science 345, 818–822 (2014).
Corcoran, T. E. et al. Absorptive clearance of DTPA as an aerosol-based biomarker in the cystic fibrosis airway. Eur. Respir. J. 35, 781–786 (2010).
Schutte, A. et al. Microbial-induced meprin beta cleavage in MUC2 mucin and a functional CFTR channel are required to release anchored small intestinal mucus. Proc. Natl Acad. Sci. USA 111, 12396–12401 (2014).
Henderson, A. G. et al. Cystic fibrosis airway secretions exhibit mucin hyperconcentration and increased osmotic pressure. J. Clin. Invest. 124, 3047–3060 (2014).
Quinton, P. M. Cystic fibrosis: impaired bicarbonate secretion and mucoviscidosis. Lancet 372, 415–417 (2008).
Garcia, M. A., Yang, N. & Quinton, P. M. Normal mouse intestinal mucus release requires cystic fibrosis transmembrane regulator-dependent bicarbonate secretion. J. Clin. Invest. 119, 2613–2622 (2009).
Gustafsson, J. K. et al. Bicarbonate and functional CFTR channel are required for proper mucin secretion and link cystic fibrosis with its mucus phenotype. J. Exp. Med. 209, 1263–1272 (2012). References 70, 71 and 76 demonstrate that a defect in mucociliary clearance emanates from abnormalities of mucus in cystic fibrosis, including its adhesive properties, shown in mouse models in which the intestines are bicarbonate-dependent, porcine airway gland ducts submerged in saline and the human airway surface, even in the setting of adequate periciliary layer depth.
Keiser, N. W. et al. Defective innate immunity and hyper-inflammation in newborn CFTR-knockout ferret lungs. Am. J. Respir. Cell. Mol. Biol. 15 Oct 2014 [epub ahead of print].
Rogers, C. S. et al. Disruption of the CFTR gene produces a model of cystic fibrosis in newborn pigs. Science 321, 1837–1841 (2008).
Kunzelmann, K. & Mehta, A. CFTR: a hub for kinases and crosstalk of cAMP and Ca2+. FEBS J. 280, 4417–4429 (2013).
Garnett, J. P. et al. Novel role for pendrin in orchestrating bicarbonate secretion in cystic fibrosis transmembrane conductance regulator (CFTR)-expressing airway serous cells. J. Biol. Chem. 286, 41069–41082 (2011).
Pezzulo, A. A. et al. Reduced airway surface pH impairs bacterial killing in the porcine cystic fibrosis lung. Nature 487, 109–113 (2012).
Pohl, K. et al. A neutrophil intrinsic impairment affecting Rab27a and degranulation in cystic fibrosis is corrected by CFTR potentiator therapy. Blood 124, 999–1009 (2014).
Bruscia, E. M. et al. Macrophages directly contribute to the exaggerated inflammatory response in cystic fibrosis transmembrane conductance regulator−/− mice. Am. J. Respir. Cell. Mol. Biol. 40, 295–304 (2009).
Wang, X. et al. Hsp90 cochaperone Aha1 downregulation rescues misfolding of CFTR in cystic fibrosis. Cell 127, 803–815 (2006).
Bedrossian, C., Greenberg, S., Singer, D., Hansen, J. & Rosenberg, H. The lung in cystic fibrosis: a quantitative study including prevalence of pathologic findings among different age groups. Hum. Pathol. 7, 195–204 (1976).
Livraghi-Butrico, A. et al. Mucus clearance, MyD88-dependent and MyD88-independent immunity modulate lung susceptibility to spontaneous bacterial infection and inflammation. Mucosal Immunol. 5, 397–408 (2012).
Hybiske, K. et al. Effects of cystic fibrosis transmembrane conductance regulator and Delta F508CFTR on inflammatory response, ER stress, and Ca2+ of airway epithelia. Am. J. Physiol. Lung Cell. Mol. Physiol. 293, L1250–L1260 (2007).
Mott, L. S. et al. Progression of early structural lung disease in young children with cystic fibrosis assessed using CT. Thorax 67, 509–516 (2011).
Sly, P. D. et al. Risk factors for bronchiectasis in children with cystic fibrosis. N. Engl. J. Med. 368, 1963–1970 (2013). This article highlights that neutrophil elastase activity in bronchoalveolar lavage fluid in early life of children with cystic fibrosis undergoing bronchoscopy is associated with early bronchiectasis. This finding shows the importance of early detection of lung infection and inflammation, and the importance of its treatment.
Gehrig, S. et al. Lack of neutrophil elastase reduces inflammation, mucus hypersecretion, and emphysema, but not mucus obstruction, in mice with cystic fibrosis-like lung disease. Am. J. Respir. Crit. Care Med. 189, 1082–1092 (2014).
Vandivier, R. W. et al. Elastase-mediated phosphatidylserine receptor cleavage impairs apoptotic cell clearance in cystic fibrosis and bronchiectasis. J. Clin. Invest. 109, 661–670 (2002).
Berger, M., Sorensen, R. U., Tosi, M. F., Dearborn, D. G. & Doring, G. Complement receptor expression on neutrophils at an inflammatory site, the Pseudomonas-infected lung in cystic fibrosis. J. Clin. Invest. 84, 1302–1313 (1989).
Day, B. J., van Heeckeren, A. M., Min, E. & Velsor, L. W. Role for cystic fibrosis transmembrane conductance regulator protein in a glutathione response to bronchopulmonary pseudomonas infection. Infect. Immun. 72, 2045–2051 (2004).
Roum, J. H., Buhl, R., McElvaney, N. G., Borok, Z. & Crystal, R. G. Systemic deficiency of glutathione in cystic fibrosis. J. Appl. Physiol. 75, 2419–2424 (1993).
Knowles, M. R. & Boucher, R. C. Mucus clearance as a primary innate defense mechanism for mammalian airways. J. Clin. Invest. 109, 571–577 (2002).
Emerson, J., Rosenfeld, M., McNamara, S., Ramsey, B. & Gibson, R. L. Pseudomonas aeruginosa and other predictors of mortality and morbidity in young children with cystic fibrosis. Pediatr. Pulmonol. 34, 91–100 (2002).
Nixon, G. M. et al. Clinical outcome after early Pseudomonas aeruginosa infection in cystic fibrosis. J. Pediatr. 138, 699–704 (2001).
Gangell, C. et al. Inflammatory responses to individual microorganisms in the lungs of children with cystic fibrosis. Clin. Infect. Dis. 53, 425–432 (2011).
van Ewijk, B. E. et al. Prevalence and impact of respiratory viral infections in young children with cystic fibrosis: prospective cohort study. Pediatrics 122, 1171–1176 (2008).
Wat, D. et al. The role of respiratory viruses in cystic fibrosis. J. Cyst. Fibros. 7, 320–328 (2008).
Wark, P. A. et al. Viral infections trigger exacerbations of cystic fibrosis in adults and children. Eur. Respir. J. 40, 510–512 (2012).
Amin, R., Dupuis, A., Aaron, S. D. & Ratjen, F. The effect of chronic infection with Aspergillus fumigatus on lung function and hospitalization in patients with cystic fibrosis. Chest 137, 171–176 (2010).
Dasenbrook, E. C. et al. Association between respiratory tract methicillin-resistant Staphylococcus aureus and survival in cystic fibrosis. JAMA 303, 2386–2392 (2010).
Govan, J. R., Brown, A. R. & Jones, A. M. Evolving epidemiology of Pseudomonas aeruginosa and the Burkholderia cepacia complex in cystic fibrosis lung infection. Future Microbiol. 2, 153–164 (2007).
Ledson, M. J., Gallagher, M. J., Jackson, M., Hart, C. A. & Walshaw, M. J. Outcome of Burkholderia cepacia colonisation in an adult cystic fibrosis centre. Thorax 57, 142–145 (2002).
Aaron, S. D. et al. Infection with transmissible strains of Pseudomonas aeruginosa and clinical outcomes in adults with cystic fibrosis. JAMA 304, 2145–2153 (2010).
Armstrong, D. S. et al. Detection of a widespread clone of Pseudomonas aeruginosa in a pediatric cystic fibrosis clinic. Am. J. Respir. Crit. Care Med. 166, 983–987 (2002).
McCallum, S. J. et al. Superinfection with a transmissible strain of Pseudomonas aeruginosa in adults with cystic fibrosis chronically colonised by P. aeruginosa. Lancet 358, 558–560 (2001).
Cramer, N. et al. Molecular epidemiology of chronic Pseudomonas aeruginosa airway infections in cystic fibrosis. PLoS ONE 7, e50731 (2012).
Kidd, T. J. et al. Pseudomonas aeruginosa exhibits frequent recombination, but only a limited association between genotype and ecological setting. PLoS ONE 7, e44199 (2012).
Wiehlmann, L. et al. Effective prevention of Pseudomonas aeruginosa cross-infection at a cystic fibrosis centre — results of a 10-year prospective study. Int. J. Med. Microbiol. 302, 69–77 (2012).
Griffiths, A. L. et al. Effects of segregation on an epidemic Pseudomonas aeruginosa strain in a cystic fibrosis clinic. Am. J. Respir. Crit. Care Med. 171, 1020–1025 (2005).
Adjemian, J., Olivier, K. N. & Prevots, D. R. Nontuberculous mycobacteria among patients with cystic fibrosis in the United States: screening practices and environmental risk. Am. J. Respir. Crit. Care Med. 190, 581–586 (2014).
Qvist, T. et al. Epidemiology of nontuberculous mycobacteria among patients with cystic fibrosis in Scandinavia. J. Cyst. Fibros. 14, 46–52 (2014).
Bryant, J. M. et al. Whole-genome sequencing to identify transmission of Mycobacterium abscessus between patients with cystic fibrosis: a retrospective cohort study. Lancet 381, 1551–1560 (2013).
Rogers, G. B. et al. Bacterial diversity in cases of lung infection in cystic fibrosis patients: 16S ribosomal DNA (rDNA) length heterogeneity PCR and 16S rDNA terminal restriction fragment length polymorphism profiling. J. Clin. Microbiol. 41, 3548–3558 (2003).
Tunney, M. M. et al. Detection of anaerobic bacteria in high numbers in sputum from patients with cystic fibrosis. Am. J. Respir. Crit. Care Med. 177, 995–1001 (2008).
LiPuma, J. J. Expanding our understanding of respiratory microbiota in cystic fibrosis. Ann. Am. Thorac. Soc. 11, 1084–1085 (2014).
Worlitzsch, D. et al. Effects of reduced mucus oxygen concentration in airway Pseudomonas infections of cystic fibrosis patients. J. Clin. Invest. 109, 317–325 (2002).
Bragonzi, A. et al. Pseudomonas aeruginosa microevolution during cystic fibrosis lung infection establishes clones with adapted virulence. Am. J. Respir. Crit. Care Med. 180, 138–145 (2009).
Marvig, R. L., Sommer, L. M., Molin, S. & Johansen, H. K. Convergent evolution and adaptation of Pseudomonas aeruginosa within patients with cystic fibrosis. Nat. Genet. 47, 57–64 (2015).
Young, R. L. et al. Neutrophil extracellular trap (NET)-mediated killing of Pseudomonas aeruginosa: evidence of acquired resistance within the CF airway, independent of CFTR. PLoS ONE 6, e23637 (2011).
Regamey, N. et al. Distinct patterns of inflammation in the airway lumen and bronchial mucosa of children with cystic fibrosis. Thorax 67, 164–170 (2012).
Tan, H. L. et al. The Th17 pathway in cystic fibrosis lung disease. Am. J. Respir. Crit. Care Med. 184, 252–258 (2011).
Hector, A. et al. Regulatory T cell impairment in cystic fibrosis patients with chronic Pseudomonas infection. Am. J. Respir. Crit. Care Med. 191, 914–923 (2015).
Ooi, C. Y. et al. Does extensive genotyping and nasal potential difference testing clarify the diagnosis of cystic fibrosis among patients with single-organ manifestations of cystic fibrosis? Thorax 69, 254–260 (2014). This manuscript looks at the use of ancillary testing in the diagnosis of cystic fibrosis. Despite increasing sophistication of ion transport measurements and genetic testing, the diagnosis frequently remains in doubt, underscoring the importance of clinical acumen in these indeterminate cases.
Ooi, C. Y. et al. Comparing the American and European diagnostic guidelines for cystic fibrosis: same disease, different language? Thorax 67, 618–624 (2012). The article discusses the nomenclature of and the approach to infants with an equivocal newborn screening test. This is a very difficult area of uncertainty for paediatricians and families, and this consensus statement, whether the nomenclature is thought to be correct or not, offers important guidance.
Stewart, B., Zabner, J., Shuber, A. P., Welsh, M. J. & McCray, P. B. Jr. Normal sweat chloride values do not exclude the diagnosis of cystic fibrosis. Am. J. Respir. Crit. Care Med. 151, 899–903 (1995).
Knowles, M., Gatzy, J. & Boucher, R. Increased bioelectric potential difference across respiratory epithelia in cystic fibrosis. N. Engl. J. Med. 305, 1489–1495 (1981).
Ooi, C. Y. et al. Does integration of various ion channel measurements improve diagnostic performance in cystic fibrosis? Ann. Am. Thorac Soc. 11, 562–570 (2014).
Vermeulen, F., Proesmans, M., Boon, M. & De Boeck, K. Improved repeatability of nasal potential difference with a larger surface catheter. J. Cyst. Fibros. [online], (2014).
Veeze, H. J., Sinaasappel, M., Bijman, J., Bouquet, J. & de Jonge, H. R. Ion transport abnormalities in rectal suction biopsies from children with cystic fibrosis. Gastroenterology 101, 398–403 (1991).
Cohen-Cymberknoh, M. et al. Evaluation of the intestinal current measurement method as a diagnostic test for cystic fibrosis. Pediatr. Pulmonol. 48, 229–235 (2013).
Dekkers, J. F. et al. A functional CFTR assay using primary cystic fibrosis intestinal organoids. Nat. Med. 19, 939–945 (2013).
Quinton, P. et al. β-adrenergic sweat secretion as a diagnostic test for cystic fibrosis. Am. J. Respir. Crit. Care Med. 186, 732–739 (2012).
Lebecque, P. et al. Mutations of the cystic fibrosis gene and intermediate sweat chloride levels in children. Am. J. Respir. Crit. Care Med. 165, 757–761 (2002).
Bush, A. & Wallis, C. Time to think again: cystic fibrosis is not an ‘all or none’ disease. Pediatr. Pulmonol. 30, 139–144 (2000).
Sims, E. J. et al. Economic implications of newborn screening for cystic fibrosis: a cost of illness retrospective cohort study. Lancet 369, 1187–1195 (2007).
Farrell, P. M. et al. Nutritional benefits of neonatal screening for cystic fibrosis. Wisconsin Cyst. Fibrosis Neonatal Screen. Study Group. N. Engl. J. Med. 337, 963–969 (1997). This article is a major report on the only randomized controlled trial of screening. Screening led to improved nutritional outcomes but no respiratory improvements. This finding probably reflects suboptimal care in one clinic, making the important point that if cystic fibrosis newborn screening is instituted, the best standards of care for the screen-positive babies must be available.
Wilcken, B. & Gaskin, K. More evidence to favour newborn screening for cystic fibrosis. Lancet 369, 1146–1147 (2007).
Balfour-Lynn, I. M. Newborn screening for cystic fibrosis: evidence for benefit. Arch. Dis. Child 93, 7–10 (2008).
Wagener, J. S., Zemanick, E. T. & Sontag, M. K. Newborn screening for cystic fibrosis. Curr. Opin. Pediatr. 24, 329–335 (2012).
Ranganathan, S. C. et al. Airway function in infants newly diagnosed with cystic fibrosis. Lancet 358, 1964–1965 (2001).
Ranganathan, S. C. et al. The evolution of airway function in early childhood following clinical diagnosis of cystic fibrosis. Am. J. Respir. Crit. Care Med. 169, 928–933 (2004).
Southern, K. W. et al. A survey of newborn screening for cystic fibrosis in Europe. J. Cyst. Fibros. 6, 57–65 (2007).
Vernooij- van Langen, A. M. et al. Novel strategies in newborn screening for cystic fibrosis: a prospective controlled study. Thorax 67, 289–295 (2012).
Southern, K. W. Determining the optimal newborn screening protocol for cystic fibrosis. Thorax 67, 281–282 (2012).
Roussey, M. et al. Neonatal screening of cystic fibrosis: diagnostic problems with CFTR mild mutations. J. Inherit Metab. Dis. 30, 613 (2007).
Scotet, V. et al. Immunoreactive trypsin/DNA newborn screening for cystic fibrosis: should the R117H variant be included in CFTR mutation panels? Pediatrics 118, e1523–e1529 (2006).
O'Sullivan, B. P., Zwerdling, R. G., Dorkin, H. L., Comeau, A. M. & Parad, R. Early pulmonary manifestation of cystic fibrosis in children with the ΔF508/R117H-7T genotype. Pediatrics 118, 1260–1265 (2006).
Peckham, D., Conway, S. P., Morton, A., Jones, A. & Webb, K. Delayed diagnosis of cystic fibrosis associated with R117H on a background of 7T polythymidine tract at intron 8. J. Cyst. Fibros. 5, 63–65 (2006).
de Nooijer, R. A. et al. Assessment of CFTR function in homozygous R117H-7T subjects. J. Cyst. Fibros. 10, 326–332 (2011).
Cystic Fibrosis, F. et al. Cystic Fibrosis Foundation practice guidelines for the management of infants with cystic fibrosis transmembrane conductance regulator-related metabolic syndrome during the first two years of life and beyond. J. Pediatr. 155, S106–S116 (2009).
Mayell, S. J. et al. A European consensus for the evaluation and management of infants with an equivocal diagnosis following newborn screening for cystic fibrosis. J. Cyst. Fibros. 8, 71–78 (2009).
Ren, C. L., Desai, H., Platt, M. & Dixon, M. Clinical outcomes in infants with cystic fibrosis transmembrane conductance regulator (CFTR) related metabolic syndrome. Pediatr. Pulmonol. 46, 1079–1084 (2011).
Wielputz, M. O. et al. Magnetic resonance imaging detects changes in structure and perfusion, and response to therapy in early cystic fibrosis lung disease. Am. J. Respir. Crit. Care Med. 189, 956–965 (2014).
Aurora, P. et al. Multiple breath inert gas washout as a measure of ventilation distribution in children with cystic fibrosis. Thorax 59, 1068–1073 (2004).
Aurora, P. et al. Multiple-breath washout as a marker of lung disease in preschool children with cystic fibrosis. Am. J. Respir. Crit. Care Med. 171, 249–256 (2005).
Kraemer, R., Blum, A., Schibler, A., Ammann, R. A. & Gallati, S. Ventilation inhomogeneities in relation to standard lung function in patients with cystic fibrosis. Am. J. Respir. Crit. Care Med. 171, 371–378 (2005).
Lum, S. et al. Early detection of cystic fibrosis lung disease: multiple-breath washout versus raised volume tests. Thorax 62, 341–347 (2007).
Hoo, A. F. et al. Lung function is abnormal in 3-month-old infants with cystic fibrosis diagnosed by newborn screening. Thorax 67, 874–881 (2012).
Sly, P. D. et al. Lung disease at diagnosis in infants with cystic fibrosis detected by newborn screening. Am. J. Respir. Crit. Care Med. 180, 146–152 (2009).
Pillarisetti, N. et al. Infection, inflammation, and lung function decline in infants with cystic fibrosis. Am. J. Respir. Crit. Care Med. 184, 75–81 (2011).
Nguyen, T. T. et al. Evolution of lung function during the first year of life in newborn screened cystic fibrosis infants. Thorax 69, 910–917 (2013).
Brennan, L. C. et al. S7 Evolution of lung function during the first two years of life in infants with cystic fibrosis diagnosed by newborn screening. Thorax 68, A6–A7 (2013).
Amin, R. et al. The effect of dornase alfa on ventilation inhomogeneity in patients with cystic fibrosis. Eur. Respir. J. 37, 806–812 (2011).
Amin, R. et al. Hypertonic saline improves the LCI in paediatric CF patients with normal lung function. Thorax 65, 379–383 (2010).
Vermeulen, F., Proesmans, M., Boon, M., Havermans, T. & De Boeck, K. Lung clearance index predicts pulmonary exacerbations in young patients with cystic fibrosis. Thorax 69, 39–45 (2014).
Maisonneuve, P., Marshall, B. C., Knapp, E. A. & Lowenfels, A. B. Cancer risk in cystic fibrosis: a 20-year nationwide study from the United States. J. Natl Cancer Inst. 105, 122–129 (2013).
Thia, L. P. et al. Is chest CT useful in newborn screened infants with cystic fibrosis at 1 year of age? Thorax 69, 320–327 (2014).
Gustafsson, P. M., De Jong, P. A., Tiddens, H. A. & Lindblad, A. Multiple-breath inert gas washout and spirometry versus structural lung disease in cystic fibrosis. Thorax 63, 129–134 (2008).
Owens, C. M. et al. Lung Clearance Index and HRCT are complementary markers of lung abnormalities in young children with CF. Thorax 66, 481–488 (2011).
Stafler, P., Davies, J. C., Balfour-Lynn, I. M., Rosenthal, M. & Bush, A. Bronchoscopy in cystic fibrosis infants diagnosed by newborn screening. Pediatr. Pulmonol. 46, 696–700 (2011).
Wainwright, C. E. et al. Effect of bronchoalveolar lavage-directed therapy on Pseudomonas aeruginosa infection and structural lung injury in children with cystic fibrosis: a randomized trial. JAMA 306, 163–171 (2011).
King, V. V. Upper respiratory disease, sinusitis, and polyposis. Clin. Rev. Allergy 9, 143–157 (1991).
Henriksson, G. et al. Nasal polyps in cystic fibrosis: clinical endoscopic study with nasal lavage fluid analysis. Chest 121, 40–47 (2002).
Hansen, S. K. et al. Evolution and diversification of Pseudomonas aeruginosa in the paranasal sinuses of cystic fibrosis children have implications for chronic lung infection. ISME J. 6, 31–45 (2012).
Rubinstein, S., Moss, R. & Lewiston, N. Constipation and meconium ileus equivalent in patients with cystic fibrosis. Pediatrics 78, 473–479 (1986).
Blondeau, K. et al. Gastro-oesophageal reflux and aspiration of gastric contents in adult patients with cystic fibrosis. Gut 57, 1049–1055 (2008).
Ledder, O., Haller, W., Couper, R. T., Lewindon, P. & Oliver, M. Cystic fibrosis: an update for clinicians. Part 2: hepatobiliary and pancreatic manifestations. J. Gastroenterol. Hepatol. 29, 1954–1962 (2014).
Borowitz, D. et al. Gastrointestinal outcomes and confounders in cystic fibrosis. J. Pediatr. Gastroenterol. Nutr. 41, 273–285 (2005).
Lewindon, P. J., Robb, T. A., Moore, D. J., Davidson, G. P. & Martin, A. J. Bowel dysfunction in cystic fibrosis: importance of breath testing. J. Paediatr. Child Health 34, 79–82 (1998).
Konstan, M. W. et al. Growth and nutritional indexes in early life predict pulmonary function in cystic fibrosis. J. Pediatr. 142, 624–630 (2003).
Sharma, R. et al. Wasting as an independent predictor of mortality in patients with cystic fibrosis. Thorax 56, 746–750 (2001).
Corbett, K. et al. Cystic fibrosis-associated liver disease: a population-based study. J. Pediatr. 145, 327–332 (2004).
Rowland, M. et al. Outcome in cystic fibrosis liver disease. Am. J. Gastroenterol. 106, 104–109 (2011).
Moran, A. et al. Clinical care guidelines for cystic fibrosis-related diabetes: a position statement of the American Diabetes Association and a clinical practice guideline of the Cystic Fibrosis Foundation, endorsed by the Pediatric Endocrine Society. Diabetes Care 33, 2697–2708 (2010).
Lewis, C. et al. Diabetes-related mortality in adults with cystic fibrosis. Role of genotype and sex. Am. J. Respir. Crit. Care Med. 191, 194–200 (2015).
Neinstein, L. S., Stewart, D., Wang, C. I. & Johnson, I. Menstrual dysfunction in cystic fibrosis. J. Adolesc. Health Care 4, 153–157 (1983).
Stern, M., Wiedemann, B., Wenzlaff, P. & German Cystic Fibrosis Quality Assessment, G. From registry to quality management: the German Cystic Fibrosis Quality Assessment project 1995–2006. Eur. Respir. J. 31, 29–35 (2008).
Marshall, B. C. & Nelson, E. C. Accelerating implementation of biomedical research advances: critical elements of a successful 10 year Cystic Fibrosis Foundation healthcare delivery improvement initiative. BMJ Qual. Saf. 23 (Suppl. 1), i95–i103 (2014).
McCormick, J. et al. Comparative demographics of the European cystic fibrosis population: a cross-sectional database analysis. Lancet 375, 1007–1013 (2010).
Colombo, C. & Littlewood, J. The implementation of standards of care in Europe: state of the art. J. Cyst. Fibros. 10 (Suppl. 2), S7–S15 (2011).
Stern, M., Niemann, N., Wiedemann, B., Wenzlaff, P. & German, C. G. Benchmarking improves quality in cystic fibrosis care: a pilot project involving 12 centres. Int. J. Qual. Health Care 23, 349–356 (2011).
Schechter, M. S. Benchmarking to improve the quality of cystic fibrosis care. Curr. Opin. Pulm. Med. 18, 596–601 (2012).
Conway, S. et al. European Cystic Fibrosis Society standards of care: framework for the Cystic Fibrosis Centre. J. Cyst. Fibros. 13, S3–S22 (2014).
European Cystic Fibrosis Society. ECFS Patient Registry annual data report 2010. (European Cystic Fibrosis Society, 2010).
Cystic Fibrosis Trust. UK CF Registry annual report 2011. (Cystic Fibrosis Trust, 2013).
Goss, C. H. et al. Children and young adults with CF in the USA have better lung function compared with the UK. Thorax 70, 229–236 (2014).
Cystic Fibrosis Australia. 15th Annual Report from the Cystic Fibrosis Data Registry. (Cystic Fibrosis Australia, 2013).
Stern, M. et al. European Cystic Fibrosis Society standards of care: quality management in cystic fibrosis. J. Cyst. Fibros. 13, S43–S59 (2014).
Quittner, A. L. et al. Pulmonary medication adherence and health-care use in cystic fibrosis. Chest 146, 142–151 (2014).
Abbott, J., Dodd, M. & Webb, A. K. Health perceptions and treatment adherence in adults with cystic fibrosis. Thorax 51, 1233–1238 (1996).
Conway, S. P., Pond, M. N., Hamnett, T. & Watson, A. Compliance with treatment in adult patients with cystic fibrosis. Thorax 51, 29–33 (1996).
Plant, B. J., Goss, C. H., Plant, W. D. & Bell, S. C. Management of comorbidities in older patients with cystic fibrosis. Lancet Respir. Med. 1, 164–174 (2013).
Grasemann, H. & Ratjen, F. Early lung disease in cystic fibrosis. Lancet Respir. Med. 1, 148–157 (2013).
Flume, P. A. et al. Cystic fibrosis pulmonary guidelines: treatment of pulmonary exacerbations. Am. J. Respir. Crit. Care Med. 180, 802–808 (2009).
Smyth, A. R. et al. European Cystic Fibrosis Society Standards of Care: Best Practice guidelines. J. Cyst. Fibros. 13, S23–S42 (2014).
Okumura, M. J. et al. Improving transition from paediatric to adult cystic fibrosis care: programme implementation and evaluation. BMJ Qual. Saf. 23 (Suppl. 1), i64–i72 (2014).
Lester, M. K. & Flume, P. A. Airway-clearance therapy guidelines and implementation. Respir. Care 54, 733–753 (2009).
McIlwaine, M. P. et al. Long-term multicentre randomised controlled study of high frequency chest wall oscillation versus positive expiratory pressure mask in cystic fibrosis. Thorax 68, 746–751 (2013).
Schneiderman, J. E. et al. Longitudinal relationship between physical activity and lung health in patients with cystic fibrosis. Eur. Respir. J. 43, 817–823 (2014).
Cox, N. S., Alison, J. A. & Holland, A. E. Interventions for promoting physical activity in people with cystic fibrosis. Cochrane Database Syst. Rev. 12, CD009448 (2013).
Fuchs, H. J. et al. Effect of aerosolized recombinant human DNase on exacerbations of respiratory symptoms and on pulmonary function in patients with cystic fibrosis. N. Engl. J. Med. 331, 637–642 (1994).
Jones, A. P. & Wallis, C. E. Recombinant human deoxyribonuclease for cystic fibrosis. Cochrane Database Syst. Rev. 3, CD001127 (2003).
Shah, P. I. et al. Recombinant human DNase I in cystic fibrosis patients with severe pulmonary disease: a short-term, double-blind study followed by six months open-label treatment. Eur. Respir. J. 8, 954–958 (1995).
Quan, J. M. et al. A two-year randomized, placebo-controlled trial of dornase alfa in young patients with cystic fibrosis with mild lung function abnormalities. J. Pediatr. 139, 813–820 (2001).
Flume, P. A. et al. Cystic fibrosis pulmonary guidelines: chronic medications for maintenance of lung health. Am. J. Respir. Crit. Care Med. 176, 957–969 (2007). References 197 and 218 describe current standards of care of implementing a cystic fibrosis centre, and for the management of patients with chronic maintenance therapies, respectively.
Elkins, M. R. et al. A controlled trial of long-term inhaled hypertonic saline in patients with cystic fibrosis. N. Engl. J. Med. 354, 229–240 (2006).
Rosenfeld, M. et al. Inhaled hypertonic saline in infants and children younger than 6 years with cystic fibrosis: the ISIS randomized controlled trial. JAMA 307, 2269–2277 (2012).
Subbarao, P. et al. Lung clearance index as an outcome measure for clinical trials in young children with cystic fibrosis. A pilot study using inhaled hypertonic saline. Am. J. Respir. Crit. Care Med. 188, 456–460 (2013).
Bilton, D. et al. Inhaled dry powder mannitol in cystic fibrosis: an efficacy and safety study. Eur. Respir. J. 38, 1071–1080 (2011).
Aitken, M. L. et al. Long-term inhaled dry powder mannitol in cystic fibrosis: an international randomized study. Am. J. Respir. Crit. Care Med. 185, 645–652 (2012).
Ramsey, B. W. et al. Efficacy of aerosolized tobramycin in patients with cystic fibrosis. N. Engl. J. Med. 328, 1740–1746 (1993).
Ramsey, B. W. et al. Intermittent administration of inhaled tobramycin in patients with cystic fibrosis. N. Engl. J. Med. 340, 23–30 (1999).
Konstan, M. W. et al. Safety, efficacy and convenience of tobramycin inhalation powder in cystic fibrosis patients: The EAGER trial. J. Cyst. Fibros. 10, 54–61 (2011).
McCoy, K. S. et al. Inhaled aztreonam lysine for chronic airway Pseudomonas aeruginosa in cystic fibrosis. Am. J. Respir. Crit. Care Med. 178, 921–928 (2008).
Retsch-Bogart, G. Z. et al. A phase 2 study of aztreonam lysine for inhalation to treat patients with cystic fibrosis and Pseudomonas aeruginosa infection. Pediatr. Pulmonol. 43, 47–58 (2008).
Doring, G., Flume, P., Heijerman, H., Elborn, J. S. & Consensus Study, G. Treatment of lung infection in patients with cystic fibrosis: current and future strategies. J. Cyst. Fibros. 11, 461–479 (2012).
Tullis, D. E. et al. Inhaled aztreonam for chronic Burkholderia infection in cystic fibrosis: a placebo-controlled trial. J. Cyst. Fibros. 13, 296–305 (2014).
Wolter, J. et al. Effect of long term treatment with azithromycin on disease parameters in cystic fibrosis: a randomised trial. Thorax 57, 212–216 (2002).
Saiman, L. et al. Azithromycin in patients with cystic fibrosis chronically infected with Pseudomonas aeruginosa: a randomized controlled trial. JAMA 290, 1749–1756 (2003).
Saiman, L. et al. Effect of azithromycin on pulmonary function in patients with cystic fibrosis uninfected with Pseudomonas aeruginosa: a randomized controlled trial. JAMA 303, 1707–1715 (2010).
Clement, A. et al. Long term effects of azithromycin in patients with cystic fibrosis: A double blind, placebo controlled trial. Thorax 61, 895–902 (2006).
Konstan, M. W., Byard, P. J., Hoppel, C. L. & Davis, P. B. Effect of high-dose ibuprofen in patients with cystic fibrosis. N. Engl. J. Med. 332, 848–854 (1995).
Lands, L. C. & Stanojevic, S. Oral non-steroidal anti-inflammatory drug therapy for lung disease in cystic fibrosis. Cochrane Database Syst, Rev. 6, CD001505 (2013).
Auerbach, H. S., Williams, M., Kirkpatrick, J. A. & Colten, H. R. Alternate-day prednisone reduces morbidity and improves pulmonary function in cystic fibrosis. Lancet 2, 686–688 (1985).
Eigen, H., Rosenstein, B. J., FitzSimmons, S. & Schidlow, D. V. A multicenter study of alternate-day prednisone therapy in patients with cystic fibrosis. J. Pediatr. 126, 515–523 (1995).
Balfour-Lynn, I. M. & Welch, K. Inhaled corticosteroids for cystic fibrosis. Cochrane Database Syst. Rev. 11, CD001915 (2009).
Balfour-Lynn, I. M. et al. Multicenter randomized controlled trial of withdrawal of inhaled corticosteroids in cystic fibrosis. Am. J. Respir. Crit. Care Med. 173, 1356–1362 (2006).
Konstan, M. W. et al. A randomized double blind, placebo controlled phase 2 trial of BIIL 284 BS (an LTB4 receptor antagonist) for the treatment of lung disease in children and adults with cystic fibrosis. J. Cyst. Fibros. 13, 148–155 (2014).
Hirche, T. O. et al. Practical guidelines: lung transplantation in patients with cystic fibrosis. Pulm. Med. 2014, 621342 (2014).
Madden, B. P. et al. Noninvasive ventilation in cystic fibrosis patients with acute or chronic respiratory failure. Eur. Respir. J. 19, 310–313 (2002).
Gill, D. R. & Hyde, S. C. Delivery of genes into the CF airway. Thorax 69, 962–964 (2014).
Crystal, R. G. et al. Administration of an adenovirus containing the human CFTR cDNA to the respiratory tract of individuals with cystic fibrosis. Nat. Genet. 8, 42–51 (1994).
Harvey, B. G., Hackett, N. R., Ely, S. & Crystal, R. G. Host responses and persistence of vector genome following intrabronchial administration of an E1−E3−, adenovirus gene transfer vector to normal individuals. Mol. Ther. 3, 206–215 (2001).
Knowles, M. R. et al. A controlled study of adenoviral-vector-mediated gene transfer in the nasal epithelium of patients with cystic fibrosis. N. Engl. J. Med. 333, 823–831 (1995).
Moss, R. B. et al. Repeated adeno-associated virus serotype 2 aerosol-mediated cystic fibrosis transmembrane regulator gene transfer to the lungs of patients with cystic fibrosis: a multicenter, double-blind, placebo-controlled trial. Chest 125, 509–521 (2004).
Moss, R. B. et al. Repeated aerosolized AAV-CFTR for treatment of cystic fibrosis: a randomized placebo-controlled phase 2B trial. Hum. Gene Ther. 18, 726–732 (2007).
Griesenbach, U. et al. Assessment of F/HN-pseudotyped lentivirus as a clinically relevant vector for lung gene therapy. Am. J. Respir. Crit. Care Med. 186, 846–856 (2012).
Alton, E. W. et al. A randomised, double-blind, placebo-controlled phase IIB clinical trial of repeated application of gene therapy in patients with cystic fibrosis. Thorax 68, 1075–1077 (2013).
Bangel-Ruland, N. et al. Cystic fibrosis transmembrane conductance regulator-mRNA delivery: a novel alternative for cystic fibrosis gene therapy. J. Gene Med. 15, 414–426 (2013).
Wilschanski, M. et al. Gentamicin-induced correction of CFTR function in patients with cystic fibrosis and CFTR stop mutations. N. Engl. J. Med. 349, 1433–1441 (2003).
Linde, L. et al. Nonsense-mediated mRNA decay affects nonsense transcript levels and governs response of cystic fibrosis patients to gentamicin. J. Clin. Invest. 117, 683–692 (2007).
Kerem, E. et al. Ataluren for the treatment of nonsense-mutation cystic fibrosis: a randomised, double-blind, placebo-controlled phase 3 trial. Lancet Respir. Med. 2, 539–547 (2014).
Study of ataluren (PTC124®) in cystic fibrosis [online], (2014).
Van Goor, F. et al. Rescue of CF airway epithelial cell function in vitro by a CFTR potentiator, VX-770. Proc. Natl Acad. Sci. USA 106, 18825–18830 (2009).
Rowe, S. M. et al. Clinical mechanism of the cystic fibrosis transmembrane conductance regulator potentiator ivacaftor in G551D-mediated cystic fibrosis. Am. J. Respir. Crit. Care Med. 190, 175–184 (2014).
De Boeck, K. et al. Efficacy and safety of ivacaftor in patients with cystic fibrosis and a non-G551D gating mutation. J. Cyst. Fibros. 13, 674–680 (2014).
Sloane, P. A. et al. A pharmacologic approach to acquired cystic fibrosis transmembrane conductance regulator dysfunction in smoking related lung disease. PLoS ONE 7, e39809 (2012).
Flume, P. A. et al. Ivacaftor in subjects with cystic fibrosis who are homozygous for the F508del-CFTR mutation. Chest 142, 718–724 (2012).
Boyle, M. P. et al. A CFTR corrector (lumacaftor) and a CFTR potentiator (ivacaftor) for treatment of patients with cystic fibrosis who have a phe508del CFTR mutation: a phase 2 randomised controlled trial 2, 527–538 (2014).
Van Goor, F. et al. Correction of the F508del-CFTR protein processing defect in vitro by the investigational drug VX-809. Proc. Natl Acad. Sci. USA 108, 18843–18848 (2011).
Veit, G. et al. Some gating potentiators, including VX-770, diminish ΔF508–CFTR functional expression. Sci. Transl. Med. 6, 246ra97 (2014).
Cholon, D. M. et al. Potentiator ivacaftor abrogates pharmacological correction of ΔF508 CFTR in cystic fibrosis. Sci. Transl. Med. 6, 246ra96 (2014).
Okiyoneda, T. et al. Mechanism-based corrector combination restores DeltaF508-CFTR folding and function. Nat. Chem. Biol. 9, 444–454 (2013).
Accurso, F. J. et al. Denufosol tetrasodium in patients with cystic fibrosis and normal to mildly impaired lung function. Am. J. Respir. Crit. Care Med. 183, 627–634 (2011).
Ratjen, F. et al. Long term effects of denufosol tetrasodium in patients with cystic fibrosis. J. Cyst. Fibros. 11, 539–549 (2012).
Burrows, E. F., Southern, K. W. & Noone, P. G. Sodium channel blockers for cystic fibrosis. Cochrane Database Syst, Rev. 4, CD005087 (2014).
Quittner, A. L., Alpern, A. N. & Kimberg, C. I. in Kendig and Chernick's Disorders of the Respiratory Tract in Children. 8th edn (eds Wilmott, R. W., Boat, T. F., Bush, A., Chernick, V., Deterding, R. R. & Ratjen, F. ) 251–260 (Elsevier (Saunders), 2012).
Goss, C. H., Edwards, T. C., Ramsey, B. W., Aitken, M. L. & Patrick, D. L. Patient-reported respiratory symptoms in cystic fibrosis. J. Cyst. Fibros. 8, 245–252 (2009).
Schmidt, A. M. et al. Exercise and quality of life in patients with cystic fibrosis: A 12-week intervention study. Physiother Theory Pract. 27, 548–556 (2011).
Schechter, M. S. et al. Long-term effects of pregnancy and motherhood on disease outcomes of women with cystic fibrosis. Ann. Am. Thorac Soc. 10, 213–219 (2013).
Sawicki, G. S. et al. Longitudinal assessment of health-related quality of life in an observational cohort of patients with cystic fibrosis. Pediatr. Pulmonol. 46, 36–44 (2011).
Snyder, C. F. et al. Implementing patient-reported outcomes assessment in clinical practice: a review of the options and considerations. Qual. Life Res. 21, 1305–1314 (2012).
Sawicki, G. S., Sellers, D. E. & Robinson, W. M. High treatment burden in adults with cystic fibrosis: challenges to disease self-management. J. Cyst. Fibros. 8, 91–96 (2009).
Quittner, A. L. Measurement of quality of life in cystic fibrosis. Curr. Opin. Pulm. Med. 4, 326–331 (1998).
Goss, C. H. & Quittner, A. L. Patient-reported outcomes in cystic fibrosis. Proc. Am. Thorac. Soc. 4, 378–386 (2007).
Quittner, A. L. et al. Erratum to: Psychometric evaluation of the Cystic Fibrosis Questionnaire-Revised in a national, US sample. Qual. Life Res. 21, 1279–1290 (2012).
Donaldson, S. H. et al. Mucus clearance and lung function in cystic fibrosis with hypertonic saline. N. Engl. J. Med. 354, 241–250 (2006).
Sawicki, G. S. et al. Treatment complexity in cystic fibrosis: trends over time and associations with site-specific outcomes. J. Cyst. Fibros. 12, 461–467 (2013). References 277 and 281 show the treatment burden and the complexity in patients with cystic fibrosis, respectively.
Quittner, A. L. et al. Impact of socioeconomic status, race, and ethnicity on quality of life in patients with cystic fibrosis in the United States. Chest 137, 642–650 (2010).
Quittner, A. L. et al. Prevalence of depression and anxiety in patients with cystic fibrosis and parent caregivers: results of The International Depression Epidemiological Study across nine countries. Thorax 69, 1090–1097 (2014).
Smith, B. A., Modi, A. C., Quittner, A. L. & Wood, B. L. Depressive symptoms in children with cystic fibrosis and parents and its effects on adherence to airway clearance. Pediatr. Pulmonol. 45, 756–763 (2010).
Riekert, K. A., Bartlett, S. J., Boyle, M. P., Krishnan, J. A. & Rand, C. S. The association between depression, lung function, and health-related quality of life among adults with cystic fibrosis. Chest 132, 231–237 (2007).
Goldbeck, L. et al. Prevalence of symptoms of anxiety and depression in German patients with cystic fibrosis. Chest 138, 929–936 (2010).
Snell, C., Fernandes, S., Bujoreanu, I. S. & Garcia, G. Depression, illness severity, and healthcare utilization in cystic fibrosis. Pediatr. Pulmonol. 49, 1177–1181 (2014).
Wong, A. P. et al. Directed differentiation of human pluripotent stem cells into mature airway epithelia expressing functional CFTR protein. Nat. Biotech. 30, 876–882 (2012). References 135 and 288 describe model systems to assess response to CFTR modulators in vitro by either using intestinal organoids or skin-derived pluripotent stem cells transformed into airway epithelial cells, respectively.
Author information
Authors and Affiliations
Contributions
Introduction (F.R.); Epidemiology (C.H.G.), Mechanisms/pathophysiology (S.M.R. and S.C.B.); Diagnosis, screening and prevention (A.B., F.R. and S.M.R.); Management (S.C.B. and F.R.); Quality of life (A.L.Q.); Outlook (F.R. and A.B.); overview of the Primer (F.R.).
Corresponding author
Ethics declarations
Competing interests
S.M.R. has received grants and/or non-financial support from: Cystic Fibrosis Foundation Therapeutics, the US National Institutes of Health (NIH), Vertex Pharmaceuticals, PTC Therapeutics, Novartis, Forest Research Institute, Bayer Healthcare and Galapagos. C.H.G. has received grant funding and/or honoraria from: the NIH (grants P30 DK089507, R01HL103965, R01AI101307 and UM1HL119073), Food and Drug Administration (grant R01FD003704), the Cystic Fibrosis Foundation, Vertex Pharmaceuticals, Transave Inc., L. Hoffmann-La Roche Ltd, Johns Hopkins University, the European Cystic Fibrosis Society, Medscape and Gilead Sciences. He has also participated in Advisory Boards for KaloBios Pharmaceuticals and Transave Inc. A.L.Q. has received grants and/or consulting income from: NIH (grant R01 DC04797), European Union Health Commission (BESTCILIA), National Health and Medical Research Council of Australia, Cystic Fibrosis Foundation Clinical Research Grant, Novartis, Abbott Pharmaceuticals, Vertex Pharmaceuticals and Gilead Sciences. F.R. has received grants and/or consulting fees from: the Canadian Institutes of Health Research, National Heart, Lung, and Blood Institute, the Cystic Fibrosis Foundation, Genentech, Vertex Pharmaceuticals, Novartis, Gilead Sciences, Boehringer Ingelheim and Roche. S.C.B. has received grants, personal fees, speaker's fees and/or non-financial support from the National Health and Medical Research Council of Australia, the Cystic Fibrosis Foundation, the Office of Health and Medical Research, Queensland Health, the Queensland Children's Foundation, Vertex Pharmaceuticals, Novartis and Gilead. He has served on advisory boards for Vertex Pharmaceuticals, Novartis and Rempex and as a site principal investigator in several clinical trials sponsored by Vertex Pharmaceuticals. A.B. is supported by the UK National Institute of Health Research Respiratory Disease Biomedical Research Unit at the Royal Brompton and Harefield National Health Service Foundation Trust and Imperial College London.
Rights and permissions
About this article
Cite this article
Ratjen, F., Bell, S., Rowe, S. et al. Cystic fibrosis. Nat Rev Dis Primers 1, 15010 (2015). https://doi.org/10.1038/nrdp.2015.10
Published:
DOI: https://doi.org/10.1038/nrdp.2015.10
This article is cited by
-
Solvent effects in anion recognition
Nature Reviews Chemistry (2024)
-
Dual-color live imaging unveils stepwise organization of multiple basal body arrays by cytoskeletons
EMBO Reports (2024)
-
Longitudinal microbial and molecular dynamics in the cystic fibrosis lung after Elexacaftor–Tezacaftor–Ivacaftor therapy
Respiratory Research (2023)
-
Lung development and regeneration: newly defined cell types and progenitor status
Cell Regeneration (2023)
-
Fractional Exhalation Nitric Oxide (FeNO) changes in cystic fibrosis patients induced by compound honey syrup: a pretest–posttest clinical trial
BMC Pulmonary Medicine (2023)