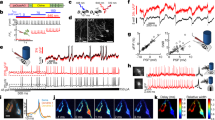
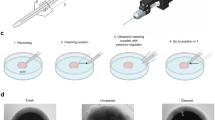
Abstract
Deciphering neuronal circuitry is central to understanding brain function and dysfunction, yet it remains a daunting task. To facilitate the dissection of neuronal circuits, a process requiring functional analysis of synaptic connections and morphological identification of interconnected neurons, we present here a method for stable simultaneous octuple patch-clamp recordings. This method allows physiological analysis of synaptic interconnections among 4–8 simultaneously recorded neurons and/or 10–30 sequentially recorded neurons, and it allows anatomical identification of >85% of recorded interneurons and >99% of recorded principal neurons. We describe how to apply the method to rodent tissue slices; however, it can be used on other model organisms. We also describe the latest refinements and optimizations of mechanics, electronics, optics and software programs that are central to the realization of a combined single- and two-photon microscopy–based, optogenetics- and imaging-assisted, stable, simultaneous quadruple–viguple patch-clamp recording system. Setting up the system, from the beginning of instrument assembly and software installation to full operation, can be completed in 3–4 d.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Scanziani, M. & Hausser, M. Electrophysiology in the age of light. Nature 461, 930–939 (2009).
Stuart, G.J. & Sakmann, B. Active propagation of somatic action potentials into neocortical pyramidal cell dendrites. Nature 367, 69–72 (1994).
Spruston, N., Schiller, Y., Stuart, G. & Sakmann, B. Activity-dependent action potential invasion and calcium influx into hippocampal CA1 dendrites. Science 268, 297–300 (1995).
Markram, H., Lubke, J., Frotscher, M., Roth, A. & Sakmann, B. Physiology and anatomy of synaptic connections between thick tufted pyramidal neurones in the developing rat neocortex. J. Physiol. (Lond) 500, 409–440 (1997).
Schiller, J., Schiller, Y., Stuart, G. & Sakmann, B. Calcium action potentials restricted to distal apical dendrites of rat neocortical pyramidal neurons. J. Physiol. (Lond) 505, 605–616 (1997).
Feldmeyer, D., Egger, V., Lubke, J. & Sakmann, B. Reliable synaptic connections between pairs of excitatory layer 4 neurones within a single 'barrel' of developing rat somatosensory cortex. J. Physiol. 521 Pt 1: 169–190 (1999).
Larkum, M.E., Zhu, J.J. & Sakmann, B. A new cellular mechanism for coupling inputs arriving at different cortical layers. Nature 398, 338–341 (1999).
Reyes, A. & Sakmann, B. Developmental switch in the short-term modification of unitary EPSPs evoked in layer 2/3 and layer 5 pyramidal neurons of rat neocortex. J. Neurosci. 19, 3827–3835 (1999).
Larkum, M.E. & Zhu, J.J. Signaling of layer 1 and whisker-evoked Ca2+ and Na+ action potentials in distal and terminal dendrites of rat neocortical pyramidal neurons in vitro and in vivo. J Neurosci 22, 6991–7005 (2002).
Waters, J., Larkum, M., Sakmann, B. & Helmchen, F. Supralinear Ca2+ influx into dendritic tufts of layer 2/3 neocortical pyramidal neurons in vitro and in vivo. J. Neurosci. 23, 8558–8567 (2003).
Luo, L., Callaway, E.M. & Svoboda, K. Genetic dissection of neural circuits. Neuron 57, 634–660 (2008).
Brown, S.P. & Hestrin, S. Cell-type identity: a key to unlocking the function of neocortical circuits. Curr. Opin. Neurobiol. 19, 415–421 (2009).
Wu, G.K., Tao, H.W. & Zhang, L.I. From elementary synaptic circuits to information processing in primary auditory cortex. Neurosci. Biobehav. Rev. 35, 2094–2104 (2011).
Le Be, J.V. & Markram, H. Spontaneous and evoked synaptic rewiring in the neonatal neocortex. Proc. Natl. Acad. Sci. USA 103, 13214–13219 (2006).
Lefort, S., Tomm, C., Floyd Sarria, J.C. & Petersen, C.C. The excitatory neuronal network of the C2 barrel column in mouse primary somatosensory cortex. Neuron 61, 301–316 (2009).
Perin, R., Berger, T.K. & Markram, H. A synaptic organizing principle for cortical neuronal groups. Proc. Natl. Acad. Sci. USA 108, 5419–5424 (2011).
Jiang, X., Wang, G., Lee, A.J., Stornetta, R.L. & Zhu, J.J. The organization of two new cortical interneuronal circuits. Nat. Neurosci. 16, 210–218 (2013).
Lee, A.J. et al. Canonical organization of layer 1 neuron-led cortical inhibitory and disinhibitory interneuronal circuits. Cereb. Cortex http://dx.doi.org/10.1093/cercor/bhu020 (2014).
Komai, S., Denk, W., Osten, P., Brecht, M. & Margrie, T.W. Two-photon targeted patching (TPTP) in vivo. Nat. Protoc. 1, 647–652 (2006).
Kitamura, K., Judkewitz, B., Kano, M., Denk, W. & Hausser, M. Targeted patch-clamp recordings and single-cell electroporation of unlabeled neurons in vivo. Nat. Methods 5, 61–67 (2008).
Suter, B.A. et al. Ephus: multipurpose data acquisition software for neuroscience experiments. Front. Neural Circuits 4, 100 (2010).
Pologruto, T.A., Sabatini, B.L. & Svoboda, K. ScanImage: flexible software for operating laser-scanning microscopes. Biomed. Eng. Online 2, 13 (2003).
Brown, S.P. & Hestrin, S. Intracortical circuits of pyramidal neurons reflect their long-range axonal targets. Nature 457, 1133–1136 (2009).
Yu, Y.C., Bultje, R.S., Wang, X. & Shi, S.H. Specific synapses develop preferentially among sister excitatory neurons in the neocortex. Nature 458, 501–504 (2009).
Buchanan, K.A. et al. Target-specific expression of presynaptic NMDA receptors in neocortical microcircuits. Neuron 75, 451–466 (2012).
Couey, J.J. et al. Recurrent inhibitory circuitry as a mechanism for grid formation. Nat. Neurosci. 16, 318–324 (2013).
Xue, M., Atallah, B.V. & Scanziani, M. Equalizing excitation-inhibition ratios across visual cortical neurons. Nature 511, 596–600 (2014).
Geiger, J.R. et al. Patch-clamp recording in brain slices with improved slicer technology. Pflugers Arch. 443, 491–501 (2002).
Zhu, J.J. Maturation of layer 5 neocortical pyramidal neurons: amplifying salient layer 1 and layer 4 inputs by Ca2+ action potentials in adult rat tuft dendrites. J. Physiol. (Lond) 526, 571–587 (2000).
Davie, J.T. et al. Dendritic patch-clamp recording. Nat. Protoc. 1, 1235–1247 (2006).
Zhu, J.J. Activity level-dependent synapse-specific AMPA receptor trafficking regulates transmission kinetics. J. Neurosci. 29, 6320–6335 (2009).
Petreanu, L., Huber, D., Sobczyk, A. & Svoboda, K. Channelrhodopsin-2-assisted circuit mapping of long-range callosal projections. Nat. Neurosci. 10, 663–668 (2007).
Zhao, S. et al. Cell type-specific channelrhodopsin-2 transgenic mice for optogenetic dissection of neural circuitry function. Nat. Methods 8, 745–752 (2011).
Constantinople, C.M. & Bruno, R.M. Deep cortical layers are activated directly by thalamus. Science 340, 1591–1594 (2013).
Zhu, Y. & Zhu, J.J. Rapid arrival and integration of ascending sensory information in layer 1 nonpyramidal neurons and tuft dendrites of layer 5 pyramidal neurons of the neocortex. J. Neurosci. 24, 1272–1279 (2004).
Murayama, M. & Larkum, M.E. In vivo dendritic calcium imaging with a fiberoptic periscope system. Nat. Protoc. 4, 1551–1559 (2009).
Tang, Q., Brecht, M. & Burgalossi, A. Juxtacellular recording and morphological identification of single neurons in freely moving rats. Nat. Protoc. 9, 2369–2381 (2014).
Zhu, Y., Stornetta, R.L. & Zhu, J.J. Chandelier cells control excessive cortical excitation: characteristics of whisker-evoked synaptic responses of layer 2/3 nonpyramidal and pyramidal neurons. J. Neurosci. 24, 5101–5108 (2004).
Wickersham, I.R. et al. Monosynaptic restriction of transsynaptic tracing from single, genetically targeted neurons. Neuron 53, 639–647 (2007).
Osakada, F. & Callaway, E.M. Design and generation of recombinant rabies virus vectors. Nat. Protoc. 8, 1583–1601 (2013).
Kim, J. et al. mGRASP enables mapping mammalian synaptic connectivity with light microscopy. Nat. Methods 9, 96–102 (2012).
Feng, L., Kwon, O., Lee, B., Oh, W.C. & Kim, J. Using mammalian GFP reconstitution across synaptic partners (mGRASP) to map synaptic connectivity in the mouse brain. Nat. Protoc. 9, 2425–2437 (2014).
Sun, Q.Q., Wang, X. & Yang, W. Laserspritzer: a simple method for optogenetic investigation with subcellular resolutions. PLoS ONE 9, e101600 (2014).
Hochbaum, D.R. et al. All-optical electrophysiology in mammalian neurons using engineered microbial rhodopsins. Nat. Methods 11, 825–833 (2014).
Kozloski, J., Hamzei-Sichani, F. & Yuste, R. Stereotyped position of local synaptic targets in neocortex. Science 293, 868–872 (2001).
Flytzanis, N.C. et al. Archaerhodopsin variants with enhanced voltage-sensitive fluorescence in mammalian and Caenorhabditis elegans neurons. Nat. Commun. 5, 4894 (2014).
St-Pierre, F. et al. High-fidelity optical reporting of neuronal electrical activity with an ultrafast fluorescent voltage sensor. Nat. Neurosci. 17, 884–889 (2014).
Edwards, F.A., Konnerth, A., Sakmann, B. & Takahashi, T. A thin slice preparation for patch clamp recordings from neurones of the mammalian central nervous system. Pflugers Arch. 414, 600–612 (1989).
Chen, X. et al. LOTOS-based two-photon calcium imaging of dendritic spines in vivo. Nat. Protoc. 7, 1818–1829 (2012).
Debanne, D. et al. Paired-recordings from synaptically coupled cortical and hippocampal neurons in acute and cultured brain slices. Nat. Protoc. 3, 1559–1568 (2008).
Marx, M., Gunter, R.H., Hucko, W., Radnikow, G. & Feldmeyer, D. Improved biocytin labeling and neuronal 3D reconstruction. Nat. Protoc. 7, 394–407 (2012).
Tomer, R., Ye, L., Hsueh, B. & Deisseroth, K. Advanced CLARITY for rapid and high-resolution imaging of intact tissues. Nat. Protoc. 9, 1682–1697 (2014).
Lim, C.S. et al. Pharmacological rescue of Ras signaling, GluA1-dependent synaptic plasticity, and learning deficits in a fragile X model. Genes Dev. 28, 273–289 (2014).
Zhu, J.J. & Connors, B.W. Intrinsic firing patterns and whisker-evoked synaptic responses of neurons in the rat barrel cortex. J. Neurophysiol. 81, 1171–1183 (1999).
Stern, E.A., Maravall, M. & Svoboda, K. Rapid development and plasticity of layer 2/3 maps in rat barrel cortex in vivo. Neuron 31, 305–315 (2001).
Lee, A.K., Epsztein, J. & Brecht, M. Head-anchored whole-cell recordings in freely moving rats. Nat. Protoc. 4, 385–392 (2009).
Acknowledgements
We thank R. Andrade, M. Beenhakker, T. Figl, V. Iyer, J. Kim, P. Neumann and K. Svoboda for technical advice and invaluable discussions, and we thank members of the Zhu laboratory for comments and technical assistance. Although we discuss here a few approaches for setting up a single- and two-photon optogenetics- and imaging-aided simultaneous multiple patch-clamp recordings system using mechanics, electronics, optics and software that we have some experience with, we anticipate alternative approaches to achieve the same goal with other instruments and software programs. We hope that this protocol will inspire more scientists to contribute their wisdom and experience to this subject. This work was supported in part by postdoctoral fellowships from the Epilepsy Foundation (G.W. and X.J.), National Institutes of Health (NIH) predoctoral training fellowships (D.R.W. and L.C.M.), a French governmental Bourse du mérite graduate fellowship and Aquimob traveling fellowship (T.L.), a Cultural and Educational Expert award from the State Administration of Foreign Experts Affairs of China (Y.S. and J.J.Z.), Chinese Ministry of Education Project 111 Program grant B13026 (Y.S. and J.J.Z.) and NIH grants R15NS081628 (Q.-Q.S. and W.Y.) and R01NS053570 (J.J.Z). This paper is the part of a dissertation in partial fulfillment of the requirements of the M.S. degree at the Bordeaux University, France (T.L.).
Author information
Authors and Affiliations
Contributions
G.W. and J.J.Z. developed the optogenetics- and imaging-assisted simultaneous multiple patch-clamp recording system with inputs from D.R.W., T.L., X.J. and Y.S. Q.-Q.S. developed the laserspritzer technique. G.W., D.R.W. and W.Y. performed the experiments with technical assistance from Y.W., L.C.M., Q.-Q.S. and J.J.Z. G.W., D.R.W. and J.J.Z. wrote the manuscript with contributions from all other authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Wang, G., Wyskiel, D., Yang, W. et al. An optogenetics- and imaging-assisted simultaneous multiple patch-clamp recording system for decoding complex neural circuits. Nat Protoc 10, 397–412 (2015). https://doi.org/10.1038/nprot.2015.019
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2015.019
This article is cited by
-
Immediate reuse of patch-clamp pipettes after ultrasonic cleaning
Scientific Reports (2024)
-
Multimodal Nature of the Single-cell Primate Brain Atlas: Morphology, Transcriptome, Electrophysiology, and Connectivity
Neuroscience Bulletin (2024)
-
Optogenetics in cardiology: methodology and future applications
International Journal of Arrhythmia (2022)
-
A biosensing system employing nanowell microelectrode arrays to record the intracellular potential of a single cardiomyocyte
Microsystems & Nanoengineering (2022)
-
In-Cell Nanoelectronics: Opening the Door to Intracellular Electrophysiology
Nano-Micro Letters (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.