Abstract
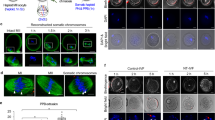
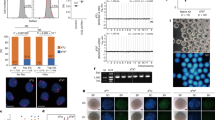
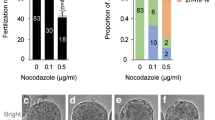
In this article, we describe detailed protocols for the isolation and transfer of spindle–chromosomal complexes between mature, metaphase II-arrested oocytes. In brief, the spindle–chromosomal complex is visualized using a polarized microscope and extracted into a membrane-enclosed karyoplast. Chromosomes are then reintroduced into an enucleated recipient egg (cytoplast), derived from another female, by karyoplast–cytoplast membrane fusion. Newly reconstructed oocytes consist of nuclear genetic material from one female and cytoplasmic components, including mitochondria and mitochondrial DNA (mtDNA), from another female. This approach yields developmentally competent oocytes suitable for fertilization and producing embryonic stem cells or healthy offspring. The protocol was initially developed for monkey oocytes but can also be used in other species, including mouse and human oocytes. Potential clinical applications include mitochondrial gene replacement therapy to prevent transmission of mtDNA mutations and treatment of infertility caused by cytoplasmic defects in oocytes. Chromosome transfer between the cohorts of oocytes isolated from two females can be completed within 2 h.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Cui, L.B., Huang, X.Y. & Sun, F.Z. Transfer of germinal vesicle to ooplasm of young mice could not rescue ageing-associated chromosome misalignment in meiosis of oocytes from aged mice. Hum. Reprod. 20, 1624–1631 (2005).
Liu, H., Wang, C.W., Grifo, J.A., Krey, L.C. & Zhang, J. Reconstruction of mouse oocytes by germinal vesicle transfer: maturity of host oocyte cytoplasm determines meiosis. Hum. Reprod. 14, 2357–2361 (1999).
Roberts, R.M. Prevention of human mitochondrial (mtDNA) disease by nucleus transplantation into an enucleated donor oocyte. Am. J. Med. Genet. 87, 265–266 (1999).
Paulson, R.J. et al. Pregnancy in the sixth decade of life: obstetric outcomes in women of advanced reproductive age. JAMA 288, 2320–2323 (2002).
Takeuchi, T., Ergun, B., Huang, T.H., Rosenwaks, Z. & Palermo, G.D. A reliable technique of nuclear transplantation for immature mammalian oocytes. Hum. Reprod. 14, 1312–1317 (1999).
Sato, A. et al. Gene therapy for progeny of mito-mice carrying pathogenic mtDNA by nuclear transplantation. Proc. Natl Acad. Sci. USA 102, 16765–16770 (2005).
Meirelles, F.V. & Smith, L.C. Mitochondrial genotype segregation in a mouse heteroplasmic lineage produced by embryonic karyoplast transplantation. Genetics 145, 445–451 (1997).
Meirelles, F.V. & Smith, L.C. Mitochondrial genotype segregation during preimplantation development in mouse heteroplasmic embryos. Genetics 148, 877–883 (1998).
Wilding, M. et al. Mitochondrial aggregation patterns and activity in human oocytes and preimplantation embryos. Hum. Reprod. 16, 909–917 (2001).
Tachibana, M. et al. Mitochondrial gene replacement in primate offspring and embryonic stem cells. Nature 461, 367–372 (2009).
Cohen, J. et al. Ooplasmic transfer in mature human oocytes. Mol. Hum. Reprod. 4, 269–280 (1998).
Tanaka, A. et al. Metaphase II karyoplast transfer from human in-vitro matured oocytes to enucleated mature oocytes. Reprod. Biomed. Online 19, 514–520 (2009).
Mitalipov, S.M. & Wolf, D.P. Nuclear transfer in nonhuman primates. Methods Mol. Biol. 348, 151–168 (2006).
Acknowledgements
This work was supported by start-up funds from the Oregon National Primate Research Center, the Oregon Stem Cell Center and grants from the National Institutes of Health HD057121, HD059946, HD063276, HD047721, HD047675, RR0000163 and U54 HD18185.
Author information
Authors and Affiliations
Contributions
S.M., M.S. and M.T. developed and wrote this protocol.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Video ~Chromosome transfer in mature oocytes~
Micromanipulation procedures for chromosome transfer involve: 1) MII oocyte positioning for chromosome-spindle detection, laser assisted zona drilling and chromosome-spindle aspiration into a karyoplast; 2) karyoplast exposure to HVJ-E; 3) and karyoplast transfer into a perivitelline space of a cytoplast. (MOV 6059 kb)
Rights and permissions
About this article
Cite this article
Tachibana, M., Sparman, M. & Mitalipov, S. Chromosome transfer in mature oocytes. Nat Protoc 5, 1138–1147 (2010). https://doi.org/10.1038/nprot.2010.75
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2010.75
This article is cited by
-
Genome transfer technique for bovine embryo production using the metaphase plate and polar body
Journal of Assisted Reproduction and Genetics (2023)
-
Mitochondrial Disease: Advances in Clinical Diagnosis, Management, Therapeutic Development, and Preventative Strategies
Current Genetic Medicine Reports (2018)
-
Polar bodies are efficient donors for reconstruction of human embryos for potential mitochondrial replacement therapy
Cell Research (2017)
-
Human oocytes reprogram adult somatic nuclei of a type 1 diabetic to diploid pluripotent stem cells
Nature (2014)
-
Towards germline gene therapy of inherited mitochondrial diseases
Nature (2013)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.