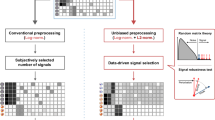
Abstract
In this protocol we describe a method to obtain telomere length parameters using Southern blots of terminal restriction fragments (TRFs). We use this approach primarily for epidemiological studies that examine leukocyte telomere length. However, the method can be adapted for telomere length measurements in other cells whose telomere lengths are within its detection boundaries. After extraction, DNA is inspected for integrity, digested, resolved by gel electrophoresis, transferred to a membrane, hybridized with labeled probes and exposed to X-ray film using chemiluminescence. Although precise and highly accurate, the method requires a considerable amount of DNA (3 μg per sample) and it measures both the canonical and noncanonical components of telomeres. The method also provides parameters of telomere length distribution in each DNA sample, which are useful in answering questions beyond those focusing on the mean length of telomeres in a given sample. A skilled technician can measure TRF length in ∼130 samples per week.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
Blasco, M.A. Telomeres and human disease: ageing, cancer and beyond. Nat. Rev. Genet. 6, 611–622 (2005).
Calado, R.T. & Young, N.S. Telomere diseases. N. Engl. J. Med. 361, 2353–2365 (2009).
Oeseburg, H., de Boer, R.A., van Gilst, W.H. & van der Harst, P. Telomere biology in healthy aging and disease. Pflugers Arch. 459, 259–268.
Zakian, V.A. The ends have arrived. Cell 139, 1038–1040 (2009).
Harley, C.B., Futcher, A.B. & Greider, C.W. Telomeres shorten during ageing of human fibroblasts. Nature 345, 458–460 (1990).
Canela, A., Vera, E., Klatt, P. & Blasco, M.A. High-throughput telomere length quantification by FISH and its application to human population studies. Proc. Natl Acad. Sci. USA 104, 5300–5305 (2007).
Lansdorp, P.M. et al. Heterogeneity in telomere length of human chromosomes. Hum. Mol. Genet. 5, 685–691 (1996).
Baerlocher, G.M., Vulto, I., de Jong, G. & Lansdorp, P.M. Flow cytometry and FISH to measure the average length of telomeres (flow FISH). Nat. Protoc. 1, 2365–2376 (2006).
Cawthon, R.M. Telomere measurement by quantitative PCR. Nucleic Acids Res. 30, e47 (2002).
Cawthon, R.M. Telomere length measurement by a novel monochrome multiplex quantitative PCR method. Nucleic Acids Res. 37, e21 (2009).
Baird, D.M., Rowson, J., Wynford-Thomas, D. & Kipling, D. Extensive allelic variation and ultrashort telomeres in senescent human cells. Nat. Genet. 33, 203–207 (2003).
Bendix, L., Horn, P.B., Jensen, U.B., Rubelj, I. & Kolvraa, S. The load of short telomeres, estimated by a new method, Universal STELA, correlates with number of senescent cells. Aging Cell 9, 383–397 (2010).
Hunt, S.C. et al. Leukocyte telomeres are longer in African Americans than in whites: the National Heart, Lung, and Blood Institute Family Heart Study and the Bogalusa Heart Study. Aging Cell 7, 451–458 (2008).
Kimura, M. et al. Offspring's leukocyte telomere length, paternal age, and telomere elongation in sperm. PLoS Genet. 4, e37 (2008).
Nawrot, T.S., Staessen, J.A., Gardner, J.P. & Aviv, A. Telomere length and possible link to X chromosome. Lancet 363, 507–510 (2004).
Valdes, A.M. et al. Obesity, cigarette smoking, and telomere length in women. Lancet 366, 662–664 (2005).
Vasan, R.S. et al. Association of leukocyte telomere length with circulating biomarkers of the renin–angiotensin–aldosterone system: the Framingham Heart Study. Circulation 117, 1138–1144 (2008).
Codd, V. et al. Common variants near TERC are associated with mean telomere length. Nat. Genet. 42, 197–199 (2010).
Levy, D. et al. Genome-wide association identifies OBFC1 as a locus involved in human leukocyte telomere biology. Proc. Natl Acad. Sci. USA 107, 9293–9298 (2010).
Alter, B.P. et al. Very short telomere length by flow fluorescence in situ hybridization identifies patients with dyskeratosis congenita. Blood 110, 1439–1447 (2007).
Rufer, N. et al. Telomere fluorescence measurements in granulocytes and T lymphocyte subsets point to a high turnover of hematopoietic stem cells and memory T cells in early childhood. J. Exp. Med. 190, 157–167 (1999).
Sambrook, J. & Russell, D.W. Isolation of high-molecular-weight DNA from mammalian cells using proteinase K and phenol. Cold Spring Harb. Protoc. doi:10.1101/pdb.prot4036 (2006).
de Lange, T. et al. Structure and variability of human chromosome ends. Mol. Cell. Biol. 10, 518–527 (1990).
Baird, D.M. et al. Telomere instability in the male germline. Hum. Mol. Genet. 15, 45–51 (2006).
Counter, C.M. et al. Telomere shortening associated with chromosome instability is arrested in immortal cells which express telomerase activity. EMBO J. 11, 1921–1929 (1992).
Levy, M.Z., Allsopp, R.C., Futcher, A.B., Greider, C.W. & Harley, C.B. Telomere end-replication problem and cell aging. J. Mol. Biol. 225, 951–960 (1992).
Allsopp, R.C. et al. Telomere length predicts replicative capacity of human fibroblasts. Proc. Natl Acad. Sci. USA 89, 10114–10118 (1992).
Vaziri, H. et al. Loss of telomeric DNA during aging of normal and trisomy 21 human lymphocytes. Am. J. Hum. Genet. 52, 661–667 (1993).
Cattan, V. et al. Chronic oxidative stress induces a tissue-specific reduction in telomere length in CAST/Ei mice. Free Radic. Biol. Med. 44, 1592–1598 (2008).
Herbert, B.S., Shay, J.W. & Wright, W.E. Analysis of telomeres and telomerase. Curr. Protoc. Cell. Biol. Chapter 18 Unit 18 6 (2003).
Acknowledgements
Telomere research at the Center for Human Development and Aging, University of Medicine and Dentistry of New Jersey, New Jersey Medical School was supported by the National Institute of Aging (NIH grants R01AG021593, R01AG20132, R01AG030678 and 5F30AG032858).
Author information
Authors and Affiliations
Contributions
M.K. oversaw the development and refinement of the TRF method, generated data shown in the figures and contributed to the writing of the article. R.S. assembled the elements of the paper, focusing on the presentation and writing of the experimental design and methodology. S.H. participated in the writing of the article, focusing on the statistical component of the introduction. J.S. oversaw the modeling and curve-fitting statistics for the TRF length distribution and contributed to the writing of the article. X.L. and X.C. routinely measured TRF length in our laboratory and provided R.S. with information to construct detailed protocols. C.H., who was a central figure in the development of the quantitative method for measuring the TRF distribution, contributed to the writing of the article. A.A. oversaw all elements of the project and participated in the writing of the article.
Corresponding author
Ethics declarations
Competing interests
C.B.H. holds stock in and consults for Geron Corporation, and is involved in the commercialization of telomere length assays.
Rights and permissions
About this article
Cite this article
Kimura, M., Stone, R., Hunt, S. et al. Measurement of telomere length by the Southern blot analysis of terminal restriction fragment lengths. Nat Protoc 5, 1596–1607 (2010). https://doi.org/10.1038/nprot.2010.124
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2010.124
This article is cited by
-
Fructose-1,6-bisphosphatase 1 dephosphorylates and inhibits TERT for tumor suppression
Nature Chemical Biology (2024)
-
ZNF524 directly interacts with telomeric DNA and supports telomere integrity
Nature Communications (2023)
-
Unraveling the causal genes and transcriptomic determinants of human telomere length
Nature Communications (2023)
-
Experimental and Computational Approaches to Measure Telomere Length: Recent Advances and Future Directions
Current Hematologic Malignancy Reports (2023)
-
The association of leukocyte telomere length with exceptional longevity among older women
GeroScience (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.