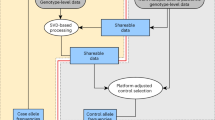
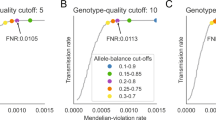
Abstract
This protocol details the steps for data quality assessment and control that are typically carried out during case-control association studies. The steps described involve the identification and removal of DNA samples and markers that introduce bias. These critical steps are paramount to the success of a case-control study and are necessary before statistically testing for association. We describe how to use PLINK, a tool for handling SNP data, to perform assessments of failure rate per individual and per SNP and to assess the degree of relatedness between individuals. We also detail other quality-control procedures, including the use of SMARTPCA software for the identification of ancestral outliers. These platforms were selected because they are user-friendly, widely used and computationally efficient. Steps needed to detect and establish a disease association using case-control data are not discussed here. Issues concerning study design and marker selection in case-control studies have been discussed in our earlier protocols. This protocol, which is routinely used in our labs, should take approximately 8 h to complete.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Zondervan, K.T. & Cardon, L.R. Designing candidate gene and genome-wide case–control association studies. Nat. Protoc. 2, 2492–2501 (2007).
The Wellcome Trust Case Control Consortium. Genome-wide association study of 14,000 cases of seven common diseases and 3,000 shared controls. Nature 447, 661–678 (2007).
Anderson, C.A. et al. Investigation of Crohn's disease risk loci in ulcerative colitis further defines their molecular relationship. Gastroenterology 136, 396–399 (2009).
Teo, Y.Y. et al. A genotype calling algorithm for the Illumina BeadArray platform. Bioinformatics 23, 2741–2746 (2007).
Clayton, D.G. et al. Population structure, differential bias and genomic control in a large-scale, case–control association study. Nat. Genet. 37, 1243–1246 (2005).
Marchini, J., Howie, B., Myers, S.R., McVean, G. & Donnelly, P. A new multipoint method for genome-wide association studies by imputation of genotypes. Nat. Genet. 39, 906–913 (2007).
Silverberg, M.S. et al. Ulcerative colitis-risk loci on chromosomes 1p36 and 12q15 found by genome-wide association study. Nat. Genet. 41, 216–220 (2009).
Pompanon, F., Bonin, A., Bellemain, E. & Taberlet, P. Genotyping errors: causes, consequences and solutions. Nat. Rev. Genet. 6, 847–859 (2005).
Price, A.L. et al. Long-range LD can confound genome scans in admixed populations. Am. J. Hum. Genet. 83, 132–135 (2008).
Purcell, S. et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am. J. Hum. Genet. 81, 559–575 (2007).
Cardon, L.R. & Palmer, L.J. Population stratification and spurious allelic association. Lancet 361, 598–604 (2003).
Campbell, C.D. et al. Demonstrating stratification in a European American population. Nat. Genet. 37, 868–872 (2005).
Risch, N. & Merikangas, K. The future of genetic studies of complex human diseases. Science 273, 1616–1617 (1996).
Patterson, N., Price, A.L. & Reich, D. Population structure and eigenanalysis. PLoS Genet. 2, e190 (2006).
Price, A.L. et al. Principal components analysis corrects for stratification in genome-wide association studies. Nat. Genet. 38, 904–909 (2006).
The International HapMap Consortium. A haplotype map of the human genome. Nature 437, 1299–1320 (2005).
The International HapMap Consortium. The International HapMap Project. Nature 426, 789–796 (2003).
Fisher, S.A. et al. Genetic determinants of ulcerative colitis include the ECM1 locus and five loci implicated in Crohn's disease. Nat. Genet. 40, 710–712 (2008).
Wittke-Thompson, J.K., Pluzhnikov, A. & Cox, N.J. Rational inferences about departures from Hardy–Weinberg equilibrium. Am. J. Hum. Genet. 76, 967–986 (2005).
Meyre, D. et al. Genome-wide association study for early-onset and morbid adult obesity identifies three new risk loci in European populations. Nat. Genet. 41, 157–159 (2009).
Moskvina, V., Craddock, N., Holmans, P., Owen, M.J. & O'Donovan, M.C. Effects of differential genotyping error rate on the type I error probability of case–control studies. Hum. Hered. 61, 55–64 (2006).
Plagnol, V., Cooper, J.D., Todd, J.A. & Clayton, D.G. A method to address differential bias in genotyping in large-scale association studies. PLoS Genet. 3, e74 (2007).
Morris, A.P. & Zeggini, E. An evaluation of statistical approaches to rare variant analysis in genetic association studies. Genet. Epidemiol. 34, 188–193 (2010).
Pettersson, F.H. et al. Marker selection for genetic case–control association studies. Nat. Protoc. 4, 743–752 (2009).
R Development Core Team. R: a language and environment for statistical computing. (2005).
Aulchenko, Y.S., Ripke, S., Isaacs, A. & van Duijn, C.M. GenABEL: an R library for genome-wide association analysis. Bioinformatics 23, 1294–1296 (2007).
Pettersson, F., Morris, A.P., Barnes, M.R. & Cardon, L.R. Goldsurfer2 (Gs2): a comprehensive tool for the analysis and visualization of genome wide association studies. BMC Bioinformatics 9, 138 (2008).
Pettersson, F., Jonsson, O. & Cardon, L.R. GOLDsurfer: three dimensional display of linkage disequilibrium. Bioinformatics 20, 3241–3243 (2004).
Acknowledgements
C.A.A. was funded by the Wellcome Trust (WT91745/Z/10/Z). A.P.M. was supported by a Wellcome Trust Senior Research Fellowship. K.T.Z. was supported by a Wellcome Trust Research Career Development Fellowship.
Author information
Authors and Affiliations
Contributions
C.A.A. wrote the first draft of the article. C.A.A. wrote scripts and performed analyses. C.A.A., F.H.P., G.M.C., A.P.M. and K.T.Z. revised the article. C.A.A., L.R.C., A.P.M. and K.T.Z. designed the protocol.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Data
Simulated dataset for use with the protocol, contains the following files: hapmap3r2_CEU.CHB.JPT.YRI.founders.no-at-cg-snps.bed hapmap3r2_CEU.CHB.JPT.YRI.founders.no-at-cg-snps.bim 8813 2010-03-09 11:12 hapmap3r2_CEU.CHB.JPT.YRI.founders.no-at-cg-snps.fam hapmap3r2_CEU.CHB.JPT.YRI.no-at-cg-snps.txt high-LD-regions.txt imiss-vs-het.Rscript pca-populations.txt plot-IBD.Rscript plot-pca-results.Rscript raw-GWA-data.map raw-GWA-data.ped (file size ~2.5GB uncompressed) raw-GWA-data.prune.in run-diffmiss-qc.pl run-IBD-QC.pl (ZIP 451047 kb)
Rights and permissions
About this article
Cite this article
Anderson, C., Pettersson, F., Clarke, G. et al. Data quality control in genetic case-control association studies. Nat Protoc 5, 1564–1573 (2010). https://doi.org/10.1038/nprot.2010.116
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2010.116
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.