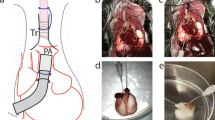
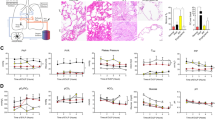
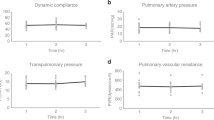
Abstract
Unlike transplantation of other solid organs, vascularized mouse lung transplantation has only recently been developed. In this protocol, we describe a detailed method for performing a vascularized and aerated mouse orthotopic lung transplant, which to date represents the most physiological mouse model of lung transplantation. The procedure is divided into two separate portions consisting of donor harvest followed by implantation using the cuff technique for bronchovascular anastomoses. After a training period spanning several months, the procedure can be successfully mastered and, in experienced hands, requires ∼90 min to perform. After an initial learning curve, perioperative survival is close to 100%. As the donor hematopoietic cells in the transplanted lung are replaced by those of the host over time, thereby creating a 'chimeric lung,' this model represents a novel research tool for the study of transplantation biology as well as other disease processes, such as malignancies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Trulock, E.P. et al. Registry of the International Society for Heart and Lung Transplantation: twenty-fourth official adult lung and heart-lung transplantation report-2007. J. Heart Lung Transplant. 26, 782–795 (2007).
Truong, W., Emamaullee, J.A., Merani, S., Anderson, C.C. & James Shapiro, A.M. Human islet function is not impaired by the sphingosine-1-phosphate receptor modulator FTY720. Am. J. Transplant. 7, 2031–2038 (2007).
Hertz, M.I., Jessurun, J., King, M.B., Savik, S.K. & Murray, J.J. Reproduction of the obliterative bronchiolitis lesion after heterotopic transplantation of mouse airways. Am. J. Pathol. 142, 1945–1951 (1993).
Belperio, J.A. et al. Role of CXCR2/CXCR2 ligands in vascular remodeling during bronchiolitis obliterans syndrome. J. Clin. Invest. 115, 1150–1162 (2005).
Belperio, J.A. et al. Critical role for the chemokine MCP-1/CCR2 in the pathogenesis of bronchiolitis obliterans syndrome. J. Clin. Invest. 108, 547–556 (2001).
Djamali, A. & Odorico, J.S. Fas-mediated cytotoxicity is not required for rejection of murine nonvascularized heterotopic cardiac allografts. Transplantation 66, 1793–1801 (1998).
Neuringer, I.P. et al. Epithelial kinetics in mouse heterotopic tracheal allografts. Am. J. Transplant. 2, 410–419 (2002).
Okazaki, M. et al. Maintenance of airway epithelium in acutely rejected orthotopic vascularized mouse lung transplants. Am. J. Respir. Cell Mol. Biol. 37, 625–630 (2007).
Khalifah, A.P. et al. Respiratory viral infections are a distinct risk for bronchiolitis obliterans syndrome and death. Am. J. Respir. Crit. Care Med. 170, 181–187 (2004).
D'Ovidio, F. et al. The effect of reflux and bile acid aspiration on the lung allograft and its surfactant and innate immunity molecules SP-A and SP-D. Am. J. Transplant. 6, 1930–1938 (2006).
Iacono, A.T. et al. A randomized trial of inhaled cyclosporine in lung-transplant recipients. N. Engl. J. Med. 354, 141–150 (2006).
Okazaki, M. et al. Sphingosine 1-phosphate inhibits ischemia reperfusion injury following experimental lung transplantation. Am. J. Transplant. 7, 751–758 (2007).
Mizuta, T., Kawaguchi, A., Nakahara, K. & Kawashima, Y. Simplified rat lung transplantation using a cuff technique. J. Thorac. Cardiovasc. Surg. 97, 578–581 (1989).
Okazaki, M. et al. A mouse model of orthotopic vascularized aerated lung transplantation. Am. J. Transplant. 7, 1672–1679 (2007).
Gelman, A.E. et al. CD4+ T lymphocytes are not necessary for the acute rejection of vascularized mouse lung transplants. J. Immunol. 180, 4754–4762 (2008).
Krieger, N.R., Yin, D.P. & Fathman, C.G. CD4+ but not CD8+ cells are essential for allorejection. J. Exp. Med. 184, 2013–2018 (1996).
Yan, S.F. et al. Egr-1, a master switch coordinating upregulation of divergent gene families underlying ischemic stress. Nat. Med. 6, 1355–1361 (2000).
Panoskaltsis-Mortari, A., Tram, K.V., Price, A.P., Wendt, C.H. & Blazar, B.R. A new murine model for bronchiolitis obliterans post-bone marrow transplant. Am. J. Respir. Crit. Care Med. 176, 713–723 (2007).
Blankenstein, T. The role of tumor stroma in the interaction between tumor and immune system. Curr. Opin. Immunol. 17, 180–186 (2005).
Folkman, J. Role of angiogenesis in tumor growth and metastasis. Semin. Oncol. 29, 15–18 (2002).
Laufer, T.M., DeKoning, J., Markowitz, J.S., Lo, D. & Glimcher, L.H. Unopposed positive selection and autoreactivity in mice expressing class II MHC only on thymic cortex. Nature 383, 81–85 (1996).
Liao, Y.-P. et al. Ionizing radiation affects human MART-1 melanoma antigen processing and presentation by dendritic cells. J. Immunol. 173, 2462–2469 (2004).
Ganss, R., Ryschich, E., Klar, E., Arnold, B. & Hammerling, G.J. Combination of T-cell therapy and trigger of inflammation induces remodeling of the vasculature and tumor eradication. Cancer Res. 62, 1462–1470 (2002).
Acknowledgements
A.S.K. is supported by (i) the American Association for Thoracic Surgery Norman Shumway Research Scholarship; (ii) American Cancer Society Internal Research Grant through the Alvin J Siteman Cancer Center; (iii) Melanoma Research Foundation; and (iv) a grant jointly sponsored by The National Cancer Institute and The Thoracic Surgery Foundation for Research and Education (1K08CA131097). D.K. is supported by a grant jointly sponsored by The National Heart, Lung and Blood Institute and The Thoracic Surgery Foundation for Research and Education (1K08HL083983-01). We thank Arlene Aligori for the medical illustrations.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Krupnick, A., Lin, X., Li, W. et al. Orthotopic mouse lung transplantation as experimental methodology to study transplant and tumor biology. Nat Protoc 4, 86–93 (2009). https://doi.org/10.1038/nprot.2008.218
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2008.218
This article is cited by
-
Metabolites released from apoptotic cells act as tissue messengers
Nature (2020)
-
Premedication with a cathepsin C inhibitor alleviates early primary graft dysfunction in mouse recipients after lung transplantation
Scientific Reports (2019)
-
Airway brushing as a new experimental methodology to detect airway gene expression signatures in mouse lung squamous cell carcinoma
Scientific Reports (2018)
-
Bioengineering of functional human induced pluripotent stem cell-derived intestinal grafts
Nature Communications (2017)
-
Lineage-negative progenitors mobilize to regenerate lung epithelium after major injury
Nature (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.