Abstract
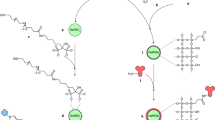
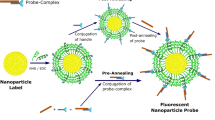
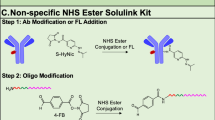
Molecular optical imaging has shown promise in visualizing molecular biomarkers with subcellular resolution both noninvasively and in real-time. Here, we use gold nanoparticles as optical probes to provide meaningful signal in the presence of targeted biomarkers. We present a novel conjugation technique to control the binding orientation of antibodies on the surface of gold nanoparticles to maximize antibody functionality. Briefly, a heterobifunctional linker, hydrazide-polyethylene glycol-dithiol, is used to directionally attach the Fc, or nonbinding region of the antibody, to the gold nanoparticle surface. The conjugation strategy allows for multiplexing various glycosylated antibodies on a single nanoparticle. We present a method to prepare multifunctional nanoparticles by incorporating targeting and delivery moieties on the same nanoparticle that addresses the challenge of imaging intracellular biomarkers. The time estimate for the entire protocol is ∼6 h.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Alivisatos, P. The use of nanocrystals in biological detection. Nat. Biotechnol. 22, 47–52 (2004).
Aaron, J. et al. Plasmon resonance coupling of metal nanoparticles for molecular imaging of carcinogenesis in vivo. J. Biomed. Opt. 12, 034007 (2007).
Kumar, S., Harrison, N., Richards-Kortum, R. & Sokolov, K. Plasmonic nanosensors for imaging intracellular biomarkers in live cells. Nano Lett. 7, 1338–1343 (2007).
Sokolov, K. et al. Real-time vital optical imaging of precancer using anti-epidermal growth factor receptor antibodies conjugated to gold nanoparticles. Cancer Res. 63, 1999–2004 (2003).
El-Sayed, I.H., Huang, X. & El-Sayed, M.A. Selective laser photo-thermal therapy of epithelial carcinoma using anti-EGFR antibody conjugated gold nanoparticles. Cancer Lett. 239, 129–135 (2006).
Huang, X., El-Sayed, I.H., Qian, W. & El-Sayed, M.A. Cancer cell imaging and photothermal therapy in the near-infrared region by using gold nanorods. J. Am. Chem. Soc. 128, 2115–2120 (2006).
Loo, C. et al. Nanoshell-enabled photonics-based imaging and therapy of cancer. Technol. Cancer Res. Treat. 3, 33–40 (2004).
Han, M.S., Lytton-Jean, A.K.R., Oh, B.-K., Heo, J. & Mirkin, C.A. Colorimetric screening of DNA-binding molecules with gold nanoparticle probes. Angew. Chem. Int. Ed. Engl. 45, 1807–1810 (2006).
Liu, G.L. et al. A nanoplasmonic molecular ruler for measuring nuclease activity and DNA footprinting. Nat. Nanotechnol. 1, 47–52 (2006).
Soennichsen, C., Reinhard, B.M., Liphardt, J. & Alivisatos, A.P. A molecular ruler based on plasmon coupling of single gold and silver nanoparticles. Nat. Biotechnol. 23, 741–745 (2005).
Kumar, S. & Richards-Kortum, R. Optical molecular imaging agents for cancer diagnostics and therapeutics. Nanomed. 1, 23–30 (2006).
El-Sayed, I.H., Huang, X. & El-Sayed, M.A. Surface plasmon resonance scattering and absorption of anti-EGFR antibody conjugated gold nanoparticles in cancer diagnostics: applications in oral cancer. Nano Lett. 5, 829–834 (2005).
Loo, C. et al. Gold nanoshell bioconjugates for molecular imaging in living cells. Opt. Lett. 30, 1012–1014 (2005).
Pathak, S., Davidson, M.C. & Silva, G.A. Characterization of the functional binding properties of antibody conjugated quantum dots. Nano Lett. 7, 1839–1845 (2007).
Aaron, J.S. et al. Increased optical contrast in imaging of epidermal growth factor receptor using magnetically actuated hybrid gold/iron oxide nanoparticles. Opt. Express 14, 12930–12943 (2006).
Spangler, B.D. & Tyler, B.J. Capture agents for a quartz crystal microbalance-continuous flow biosensor: functionalized self-assembled monolayers on gold. Anal. Chim. Acta 399, 51–62 (1999).
Won, J. et al. A magnetic nanoprobe technology for detecting molecular interactions in live cells. Science 309, 121–125 (2005).
Mallidi, S., Larson, T., Aaron, J., Sokolov, K. & Emelianov, S. Molecular specific optoacoustic imaging with plasmonic nanoparticles. Opt. Express 15, 6583–6588 (2007).
Larson, T.A., Bankson, J., Aaron, J. & Sokolov, K. Hybrid plasmonic magnetic nanoparticles as molecular specific agents for MRI/optical imaging and photothermal therapy of cancer cells. Nanotechnology 18, 325101 (2007).
Frens, G. Controlled nucleation for the regulation of the particle size in monodisperse gold suspensions. Nature 241, 20–22 (1973).
Cooper, J.A., Walker, S.B. & Pollard, T.D. Pyrene actin: documentation of the validity of a sensitive assay for actin polymerization. J. Muscle Res. Cell Motil. 4, 253–262 (1983).
Kouyama, T. & Mihashi, K. Fluorimetry Study of N-(1-pyrenyl)iodoacetamide-labelled F-actin. Local structural change of actin protomer both on polymerization and on binding of heavy meromyosin. Eur. J. Chem. 114, 33–38 (1981).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kumar, S., Aaron, J. & Sokolov, K. Directional conjugation of antibodies to nanoparticles for synthesis of multiplexed optical contrast agents with both delivery and targeting moieties. Nat Protoc 3, 314–320 (2008). https://doi.org/10.1038/nprot.2008.1
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2008.1
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.