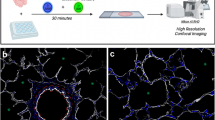
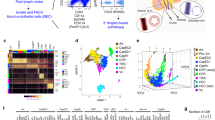
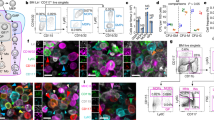
Abstract
The goal of many current studies of neovascularization is to define the phenotype of vascular cell populations of different origins and to determine how such cells promote assembly of vascular channel. Here, we describe a protocol to immunophenotype vascular cells by high-resolution imaging and by fluorescence-activated flow cytometry in an in vivo rodent model of pulmonary microvascular remodeling. Analysis of cells by this combined approach will characterize their phenotype, quantify their number and identify their role in the assembly of vascular channels.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Jones, R. Ultrastructural analysis of contractile cell development in lung microvessels in hyperoxic pulmonary hypertension. Fibroblasts and intermediate cells selectively reorganize nonmuscular segments. Am. J. Pathol. 141, 1491–1505 (1992).
Jones, R.C., Capen, D., Petersen, B., Jain, R.K. & Duda, D.G. A protocol for a lung neovascularization model in rodents. Nat. Protoc. 3, 378–387 (2008).
Jones, R.C. & Jacobson, M. Angiogenesis in the hypertensive lung: response to ambient oxygen tension. Cell Tissue Res. 300, 263–284 (2000).
Jones, R., Capen, D., Jacobson, M. & Munn, L. PDGF and microvessel wall remodeling in adult rat lung: imaging PDGF-AA and PDGF-Rα molecules in progenitor smooth muscle cells developing in experimental pulmonary hypertension. Cell Tissue Res. 326, 759–769 (2006).
Jones, R., Capen, D. & Jacobson, M. PDGF and microvessel wall remodeling in adult lung: imaging PDGF-Rβ and PDGF-BB molecules in progenitor smooth muscle cells developing in pulmonary hypertension. Ultrastruct. Pathol. 30, 267–281 (2006).
Duda, D.G. et al. Evidence for incorporation of bone marrow-derived endothelial cells into perfused blood vessels in tumors. Blood 107, 2774–2776 (2006).
Jones, R., Jacobson, M. & Steudel, W. Alpha-smooth-muscle actin and microvascular precursor smooth-muscle cells in pulmonary hypertension. Am. J. Respir. Cell Mol. Biol. 20, 582–594 (1999).
Jones, R., Steudel, W., White, S., Jacobson, M. & Low, R. Microvessel precursor smooth muscle cells express head-inserted smooth muscle myosin heavy chain (SM-B) isoform in hyperoxic pulmonary hypertension. Cell Tissue Res. 295, 453–465 (1999).
De Palma, M. et al. Tie2 identifies a hematopoietic lineage of proangiogenic monocytes required for tumor vessel formation and a mesenchymal population of pericyte progenitors. Cancer Cell 8, 211–226 (2005).
De Palma, M., Venneri, M.A., Roca, C. & Naldini, L. Targeting exogenous genes to tumor angiogenesis by transplantation of genetically modified hematopoietic stem cells. Nat. Med. 9, 789–795 (2003).
Duda, D.G. et al. Differential transplantability of tumor-associated stromal cells. Cancer Res. 64, 5920–5924 (2004).
Acknowledgements
Supported by NIH R01HL 070866 (to RJ), NIH R01CA 115767 (to RKJ) and by a Career Development Award to DGD (from the American Association for Cancer Research-Genentech BioOncology).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jones, R., Capen, D., Cohen, K. et al. A protocol for phenotypic detection and characterization of vascular cells of different origins in a lung neovascularization model in rodents. Nat Protoc 3, 388–397 (2008). https://doi.org/10.1038/nprot.2007.537
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2007.537
This article is cited by
-
A protocol for a lung neovascularization model in rodents
Nature Protocols (2008)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.