Abstract
Non-invasive imaging of transgene expression requires the appropriate combination of a reporter gene and a reporter probe. [18F]FEAU positron emission tomography (PET) is used for the assessment of herpes simplex virus type-1 thymidine kinase gene expression. Hybrid AAV phage (termed AAVP) can be adapted to transduce mammalian cells by targeting to a specific receptor. We evaluated a targeted AAVP vector using [18F]FEAU PET. This protocol describes [18F]FEAU production and dosing, micro-PET imaging and image analysis. 2-Deoxy-2-trifluoromethanesulfonyl-1,3,5-tri-O-benzoyl-α-D-ribofuranose is radio-fluorinated, converted into its 1-bromo derivative and coupled with protected 5-ethyl uracil. The coupled product is hydrolyzed and purified using HPLC. Tumor-bearing animals targeted with either retroviral or AAVP vectors are anesthetized and injected with [18F]FEAU (0.1 mCi per mouse); this is followed 2 h after injection by imaging on a micro-PET. Production of [18F]FEAU requires approximately 3.5 h from the end of bombardment. PET imaging studies require 2–3 h (depending on the number of animals) after synthesis of [18F]FEAU.
Similar content being viewed by others
Introduction
A paradigm for non-invasive imaging of transgene expression involves the appropriate combination of a reporter gene and a reporter substrate or probe1,2,3. This imaging paradigm is essentially an in vivo radiotracer enzyme assay, where the reporter gene product (an enzyme) selectively converts a reporter probe into a metabolite that is trapped within the transduced cells. Model systems have been established and validated in tissue culture and experimental animals using herpes simplex virus type-1 thymidine kinase (HSV1-tk) gene as a reporter gene and radiolabeled pyrimidine nucleoside analogs and acycloguanosine analogs as reporter probes4,5,6,7,8,9,10. The use of this paradigm and the reporter gene technology have been extended. Reporter genes can be used (i) to image vector targeting and level of suicide gene (HSV1-tk) expression11; (ii) to image the regulation of endogenous genes and signal-transduction pathways12,13,14,15,16; and (iii) to monitor and quantitatively assess the expression of a second transgene that is cis-linked to the reporter gene by an internal ribosome entry site sequence14,15.
We developed the acycloguanosine derivatives [18F]FHPG and [18F]FHBG5,6,7,8 and a series of 18F-labeled pyrimidine nucleoside derivatives, including [18F]FMAU, [18F]FIAU, [18F]FFAU, [18F]FCAU, [18F]FBAU and [18F]FEAU (Fig. 1), for positron emission tomography (PET) imaging of HSV1-tk gene expression17,18,19,20,21,22. We have investigated these pyrimidine and guanosine analogs in human colon cancer cells (HT-29) to compare their efficacies6,7,8,18,20,21,22. The pyrimidine derivatives have been found to have two advantages over the acycloguanosine derivatives. First, pyrimidine nucleosides are much more (log-order) sensitive than the guanosine derivatives [18F]FHPG and [18F]FHBG4,20. Second, pyrimidine derivatives follow renal clearance20, and therefore the background activity is negligible, whereas [18F]FHBG is cleared through a hepatobiliary pathway, resulting in the retention of large amounts of radioactivity in the intestine and other organs13,16. Among the pyrimidine nucleosides, [18F]FFAU has the highest sensitivity and specificity for HSV1-tk, and FEAU has lower sensitivity and specificity than FFAU in HT-29 cells22. Other than FFAU and FEAU, pyrimidine analogs have some non-specific uptake22. These results suggest that FFAU and FEAU are the imaging agents most specific for HSV1-tk. However, when we compared these tracers in human breast cancer cells (MDA-MB-468), total uptake of [18F]FFAU in HSV1-tk-expressing cells was 150-fold less than that of FEAU23. This result suggests that [18F]FEAU may be the best choice of PET imaging agent for HSV1-tk gene expression in terms of sensitivity and specificity; however, further studies are necessary to investigate the efficacy of these nucleosides in a variety of cancer cells. Recently, others have demonstrated that FEAU has the highest uptake ratio between tk-positive transduced cells and wild-type cells when compared with FHBG, FIAU, FMAU, FHPG and FLT24. It has been reported that a combination of high accumulation and high selectivity for both HSV1-tk and HSV1-sr39tk makes FEAU a promising candidate for PET imaging of HSV-tk gene expression25. [18F]FEAU has also been used to monitor the response of tumors using PET after systemic Sindbis/tk (vector) treatment as a basis for determining the levels of tissue distribution of vectors in living animals26.
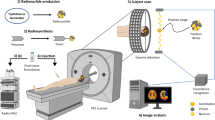
Bacteriophages (phages) are bacterial viruses that can be adapted to transduce mammalian cells by targeting to a specific receptor27,28,29,30,31,32. We have recently developed and evaluated a novel phage-based targeted vector, a genetic chimera of filamentous phage and recombinant AAV (termed AAVP)28. We have demonstrated that it is possible to assess targeted AAVP vectors non-invasively in experimental tumors. The expression of the reporter gene (HSV1-tk) can be successfully imaged using [18F]FEAU and PET in pre-clinical tumor models28. This protocol covers the following aspects of this approach. (i) Radiotracer ([18F]FEAU) production is a seven-step synthesis, shown schematically in Figure 2. (ii) This is followed by radiotracer dosing and administration. (iii) Animal preparation and tumor growth: use nine mice per group to generate statistically meaningful results. Animals are divided into two groups, with group 1 receiving targeted AAVP vector and group 2 receiving non-targeted AAVP vector administration. Negative control groups consist of mice with tumors produced from parental tumor cells; positive controls are mice with tumors produced from tumor cells stably transfected with HSV1-tk. (iv) After micro-PET calibration, the animals are imaged using [18F]FDG (18fluoro-2-deoxyglucose) PET to observe viable tumor cells on day 4 after the AAVP vector administration. Animals are then imaged with [18F]FEAU PET to observe HSV1-tk gene expression (from day 5 onward). (v) The protocol ends with image reconstruction, analysis and interpretation33.
See ref. 33 for methods for (i) the cloning and generation of the targeted AAVP vector; (ii) transduction of mammalian cells in culture by the targeted AAVP vector and specific inhibition using synthetic peptides; (iii) real-time transgene expression in mouse models after systemic administration33.
Materials
Reagents
-
Cells (see REAGENT SETUP)
-
Minimal essential medium (MEM; Irvine Scientific, Santa Ana, CA)
-
DMEM (Gibco, Gaithersburg, MD)
-
L-GIn
-
Penicillin/streptomycin
-
Animals (see REAGENT SETUP)
-
Oxygen-18 enriched water ([18O]H2O) (any suitable source as used by cyclotron operators)
-
K2CO3 (Aldrich, St. Louis, MO)
Caution
Irritant.
-
Kryptofix 2.2.2 (Aldrich)
Caution
Toxic, use in a fume hood.
-
Anhydrous acetonitrile (MeCN; Aldrich)
Caution
Flammable liquid, toxic.
-
Ethyl acetate (EtOAc; Aldrich)
Caution
Flammable liquid, irritant.
-
1,3,5-Tri-O-benzoyl-α-D-ribofuranose (Aldrich)
-
Anhydrous pyridine (Aldrich)
Caution
Irritant, use in a fume hood.
-
Trifluoromethane sulfonic anhydride (Aldrich)
Caution
Corrosive and moisture sensitive.
-
Hexane (Aldrich)
Caution
Flammable liquid, irritant.
-
Acetone (Aldrich)
Caution
Flammable liquid, irritant.
-
Dichloromethane (CH2Cl2 ; Aldrich)
Caution
Flammable liquid, cancer suspecting agent.
-
1,2-Dichloroethane (Aldrich)
Caution
Flammable liquid, cancer suspecting agent.
-
1,1,1,3,3,3-Hexamethyldisilazane (Aldrich)
Caution
Flammable liquid, corrosive.
-
Chlorotrimethylsilane (TMSCl; Aldrich)
Caution
Flammable liquid.
-
5-Ethyl uracil (Aldrich)
Caution
Irritant.
-
Hydrogen bromide in acetic acid (HBr/AcOH, 30% (wt/wt); Aldrich)
Caution
Highly toxic, use in fume hood.
-
Hydrochloric acid (HCl, 1 M; Aldrich)
Caution
Highly toxic and corrosive.
-
Sodium methoxide (NaOMe, 0.5 M; Aldrich)
Caution
Flammable and corrosive.
-
Methanol (MeOH; Aldrich)
Caution
Flammable and toxic.
-
Toluene (Aldrich)
Caution
Flammable and toxic.
-
Saline (Abbott Laboratories, Chicago, IL)
-
2-Deoxy-2-trifluoromethanesulfonyl-1,3,5-tri-O-benzoyl-α-D-ribofuranose 1; prepared in-house17,19,34,35 (see NMR data)
-
Compound 2 (see REAGENT SETUP)
-
Standard solution of FEAU (1 mg ml−1 in 10% MeCN/water (vol/vol)); prepared in-house19,35,36 (see REAGENT SETUP)
-
2,4-Bis-trimethylsilyl-5-ethyl uracil 4; prepared in-house19 (see PROCEDURE)
-
RGD-4C AAVP-HSV1 vector28
-
Non-targeted AAVP-HSV1-tk vector28
Equipment
-
Round-bottomed flasks (100 ml and other sizes)
-
Pear-shaped flask (15 ml)
-
V-vials (5 ml)
-
Rotary evaporator
-
Rubber septum
-
Aluminum seals
-
Crimper
-
Thin layer chromatography (TLC) plates
-
HPLC system (see EQUIPMENT SETUP)
-
TLC developing jar
-
High-vacuum pump (see EQUIPMENT SETUP)
-
Vacuum manifold
-
Flash chromatographic column
-
Silica gel
-
Reflux condenser
-
Glass adapter
-
Needles
-
Syringe (gas tight)
-
Regular glass and plastic syringe (Becton Dickinson)
-
Teflon tubing (flanged), connectors and end fittings
-
Inert gas (argon or nitrogen)
-
Gas flow meter
-
Balloons
-
Silica gel Sep-Pack cartridge (900 mg; Alltech Associates)
-
Anion exchange cartridge to trap the [18F]fluoride from [18O]H2O (ABX, Germany, cat. no. chromafix 30PS-HCO3); use a setup similar to that used in the production of [18F]FDG
-
Millipore filter (0.22 μm; Millipore)
-
Dose calibrator (Capintec) and a survey meter (any suitable make)
-
Micro-PET scanner (R-4; Concord Microsystems) (see EQUIPMENT SETUP)
-
Cyclotron or a source of [18F]fluoride (GE or any other suitable make)
-
Radiation badge including ring badge
-
Hot cell (to perform radiochemical synthesis) with manipulators
-
Semi-automated chemistry box (see EQUIPMENT SETUP)
-
One column C18 reverse-phase Econosil semi-preparative column (10 × 250 mm2) for purification (Alltech Associates)
-
One analytical column C18 reverse-phase Econosil (4 × 250 mm2) for QC analysis (Alltech Associates)
-
NMR spectrometer or a source for chemical analysis of the precursor compound
-
HPLC column (see EQUIPMENT SETUP)
Reagent setup
-
Compound 2 Preparation of compound 2 is performed in the cyclotron area using an FDG box (GE) or similar.
-
K2CO3/kryptofix solution Prepare a stock solution of K2CO3/kryptofix 2.2.2 by mixing a solution of kryptofix 2.2.2 (12 mg ml−1 in MeCN) and K2CO3 (2.75 mg ml−1 in water) in a ratio of 80:20 (vol/vol), and store this solution in a refrigerator as a shelf reagent for routine use (discard after 6 months).
-
Standard solution of cold FEAU Prepare a stock solution of cold FEAU, 1 mg ml−1 HPLC solvent (10% MeCN in water), and store this solution in a refrigerator. FEAU can be synthesized according to the previously reported method35,36 or following the synthetic scheme in Figure 2.
-
Cells Any cancer cells can be used7,8,18. For phage vector-targeted studies, we have used the following cell lines: Kaposi's sarcoma KS1767 (ATCC, Manassas, VA), EF43-FGF4 mouse mammary tumor cells, DU145 prostate carcinoma cells and UC3 bladder carcinoma cells28. The Kaposi's sarcoma cells were maintained in MEM; the other cell lines were cultured in DMEM. All media were supplemented with 10% FBS (Gibco), L-GIn and penicillin/streptomycin. All the tumors established by these cells in mice responded to systemic administration of targeted RGD-4C AAVP-HSV1-tk.
-
Animals To induce s.c. tumors, use athymic or nude mice. We used either female (6-week-old) mice for stably transduced tumor implantation with HT-29 cells or male (1-month-old) mice for AAVP vector HSV1-tk gene expression studies.
Critical
Approval of the protocol by the appropriate Institutional Animal Care and Use Committee is required.
Equipment setup
-
Semi-automated chemistry box (custom made, Fig. 3) used for preparation of compounds 3, 5 and 6 (Fig. 2). Check the gas flow from the argon or nitrogen tank through the box to ensure that all lines are clear and there is no leak in the system. Upload HBr/acetic acid, toluene (0.5 ml), compound 4, NaOMe/MeOH (0.4 ml) and HPLC solvent (10% MeCN/water, 1.2 ml, containing 0.1 ml 1 N HCl) in the reserve vials.
Figure 3: A schematic drawing of the chemistry box for coupling 18F-labeled sugar with the protected 5-ethyl uracil in [18F]FEAU synthesis. The first part (preparation of [18F]sugar, compound 2) is performed on a separate box, such as an FDG box. This box has solenoid valves (NR, 24 V DC, P/N HP225K032) operated using a remote-control switch box. Each solenoid valve opens a v-vial to transfer reagents from the v-vial to the reaction vial ([18F]sugar). The 18F-labeled sugar produced is then passed through a Sep-Pack (silica) cartridge. The cartridge is eluted with EtOAc and collected in the reaction vial. The solvent is evaporated using a pump under a flow of nitrogen or argon. HBr/acetic acid and CH2Cl–CH2Cl are added to the dry sugar. Subsequently, toluene, compound 4, NaOMe and HPLC solvents are added sequentially, as necessary, using the remote-control switch box.
-
HPLC The production of [18F]FEAU requires two HPLC systems, each with a UV detector (e.g., Waters or Agilent) and a radioactivity detector (Bioscans, Washington, DC). One system is for purification and the other is for quality control analysis. UV wavelength is 254 nm.
-
HPLC columns On the day of the experiment, equilibrate HPLC columns, both semi-preparative and analytical, with the appropriate HPLC solvent (10% MeCN/water) before starting the radiosynthesis of [18F]FEAU. Set up the UV lamp at 254 nm. Inject the cold standard solution of FEAU into the HPLC, elute (isocratic) with 10% MeCN solvent (4 ml min−1, semi-preparative; 1 ml min−1, analytical) and record the retention time of FEAU in both the semi-preparative and analytical HPLC systems.
-
High-vacuum pump Set up the high vacuum pump with the cooling trap filled with dry ice and isopropanol.
-
Micro-PET scanner Calibrate the micro-PET animal scanner (Concord Microsystems, R-4) according to the manufacturer's protocol.
Procedure
Radiotracer ([18F]FEAU) production: preparation of precursor, 2-deoxy-2-trifluoromethanesulfonyl-1,3,5-tri-O-benzoyl-α-D-ribofuranose 1
-
1
Dissolve 1,3,5-tri-O-benzoyl-α-D-ribofuranose (1.0 g) in dry pyridine (20 ml) in a round-bottomed flask (100 ml) containing a magnetic bar under argon and cool to 0 °C in an ice bath.
Caution
Perform experiments in a fume hood for chemical synthesis, and use gloves for handling toxic chemicals.
-
2
Add trifluoromethane sulfonic anhydride (1.3 equiv.), stir the solution for 15 min at 0 °C, warm to room temperature (∼22 °C) and continue stirring for another hour. Monitor the reaction by TLC as follows: transfer a small aliquot of the reaction mixture using a dry syringe to a small flask, pump off pyridine under vacuum and re-dissolve in CH2Cl2. Spot the solution using a capillary tube on the TLC plate and develop the plate in a developing jar using 15% EtOAc/hexane (vol/vol). Check the plate under a UV lamp. The Rf values are 0.4 for the starting material and 0.5 for the product.
-
3
When the reaction is complete (by TLC), remove pyridine using a vacuum pump, chromatograph the crude product on a silica gel column (20 × 3 cm, approximately 100 g) and elute the column with 15% EtOAc/hexane at a flow rate of 20 ml min−1.
-
4
Collect fractions (10 ml) until the compound is completely eluted (check fractions by spotting on a silica gel plate and visualizing under a UV lamp).
-
5
Combine the fractions that match with the product (Rf = 0.5) in a round-bottomed flask and evaporate solvent using a rotary evaporator.
-
6
Connect the flask to a high-vacuum pump and continue pumping for approximately 4 h (compound is a white solid).
-
7
Check the identity of the white solid product using 1H NMR and 19F NMR spectroscopy, and compare the results with those reported previously34,35 (see analytical data).
-
8
Divide the product into 5-mg aliquots in small vials and seal the vials under argon.
Pause point
Store the precursor at 4 °C in a desiccator for future radio-fluorination.
Radiotracer ([18F]FEAU) production: preparation of 2,4-bis-trimethylsilyl-5-ethyl uracil 4
-
9
Weigh 50 mg 5-ethyl uracil and transfer to a pear-shaped flask (15 ml) under argon.
-
10
Add MeCN (1.0 ml), 1,1,1,3,3,3-hexamethyldisilazane (0.5 ml) and TMSCl (0.5 ml) into the flask.
-
11
Connect a reflux condenser to the flask and reflux the reaction mixture at 80 °C for 1 h (until a clear solution is formed).
-
12
Cool the reaction mixture to approximately 40 °C, remove the condenser, connect the flask containing 4 with an adapter and put under a vacuum pump to remove excess reagents and solvent.
-
13
When all reagents are evaporated, disconnect the flask from the vacuum pump and flush with argon to prevent air entering into the flask.
-
14
Dissolve the residue (liquid) in 1,2-dichloroethane (4.0 ml) and transfer the solution to a sealed vial under argon.
Caution
Use a glove box under argon to avoid any exposure to moisture.
Pause point
Store the solution at 20 °C in a desiccator for future coupling experiments.
Radiotracer ([18F]FEAU) production: production of 18F isotope in a cyclotron
-
15
Bombard [18O]H2O (1 ml) with protons following the reaction 18O(p, n)18F for 15 min or longer (depending on the amount of activity required), and pass the 18F-enriched [18O]H2O from the target through an anion exchange cartridge.
Caution
The product is radioactive. Use radiation badge and ring badge.
-
16
Elute the radioactive material from the cartridge with a solution of K2CO3 and kryptofix 2.2.2 (1.2 ml) into a pre-crimped v-vial and transfer the v-vial to the radiochemistry lab in a hot cell (or to a FDG production box for fluorination).
Radiotracer ([18F]FEAU) production: preparation of [2-18F]fluoro-1,3,5-tri-O-benzoyl-α-D-arabinofuranose 2
-
17
Put the [18F]fluoride obtained in Step 16 into a heating block.
Caution
Radioactive material, use gloves; all experiments should be performed in a hot cell.
-
18
Remove water from the [18F]fluoride solution by azeotropic evaporation with dry MeCN (1–2 ml) at 110 °C under a stream of argon (dryness is observed by visualization).
-
19
Dissolve 2-deoxy-2-trifluoromethanesulfonyl-1,3,5-tri-O-benzoyl-α-D-ribofuranose 1 (5 mg, stored as a shelf compound) in dry MeCN (0.5 ml).
-
20
Transfer the precursor solution (Step 19) to the dry [18F]fluoride in the v-vial (Step 18) using a semi-automated box system or manual addition with manipulators.
-
21
Heat the reaction mixture at 80 °C for 15 min.
-
22
Cool the reaction mixture to 40 °C. Connect the v-vial with a silica gel Sep-Pack cartridge (pre-treated with hexane) using Teflon tubing (approximately 2 feet long, 1/16 inch i.d.).
-
23
Pass the reaction mixture through the Sep-Pack cartridge and discard the eluent (Fig. 3).
-
24
Connect the Sep-Pack to a second v-vial (Fig. 3). Elute the product with EtOAc (2 ml) into the pre-crimped v-vial (Fig. 3).
Radiotracer ([18F]FEAU) production: preparation of [2-18F]fluoro-1-bromo-3,5-di-O-benzoyl-α-D-arabinofuranose 3
-
25
Remove solvent from the product 2 ([18F]sugar) obtained in Step 24 by blowing a stream of nitrogen or argon at 80 °C under vacuum.
-
26
Add 1,2-dichloroethane (0.4 ml) and HBr in AcOH (30%, 0.1 ml) to the dry compound 2 (through the semi-automated box system or by manual addition using manipulators), and heat the mixture for 10 min at 80 °C in a heating block.
-
27
Evaporate the solvent to 50% by volume under a stream of argon. Add 0.5–0.7 ml toluene (through the semi-automated box) and continue evaporation to dryness (visual inspection).
Radiotracer ([18F]FEAU) production: coupling of the sugar with the base; preparation of protected [18F]FEAU 5
-
28
Add the solution of 2,4-bis-trimethylsilyl-5-ethyl uracil 4 as follows: withdraw the solution of 4 in a dry gas-tight syringe (0.4 ml) and add to the reaction vial ([18F]sugar) containing the 1-bromo-[2-18F]fluorosugar (through the semi-automated box system or by manual addition using manipulators). After adding the protected 5-ethyl uracil 4 into the v-vial from Step 27, heat the reaction mixture at 120 °C for 30 min in a heating block.
-
29
Cool the reaction mixture to 80 °C and evaporate the solvent by blowing argon or nitrogen under vacuum.
Radiotracer ([18F]FEAU) production: hydrolysis of the protecting groups; preparation of [18F]FEAU 6
-
30
Add NaOMe (0.1 ml, 0.5 M solution) and MeOH (0.4 ml) into the reaction vial from Step 29 (through the semi-automated box system or by manual addition using manipulators). Heat the vial containing the reaction mixture for 5 min at 80 °C, and then evaporate solvent under a flow of argon or nitrogen gas and vacuum.
-
31
Cool and add (through the semi-automated box system or by manual addition using manipulators) 1.2 ml HPLC solvent (10% MeCN/water) containing 0.1 ml HCl solution (1 M).
-
32
Inject the crude product onto the semi-preparative column (through the semi-automated box system or by manual addition using manipulators) pre-equilibrated with 10% MeCN/water and purify the product using HPLC. Elute the column with 10% MeCN/water at a flow rate of 4 ml min−1. Collect the appropriate fraction with similar retention time to the standard FEAU (follow radioactive peak) into a flask to obtain the desired product [18F]FEAU 6 (Fig. 4).
-
33
Evaporate solvent on a rotary evaporator, re-dissolve the product in saline (2–5 ml, depending on the desired activity concentration) and filter through a Millipore filter for dose preparation.
-
34
Measure radioactivity on a dose calibrator (Capintec), calculate activity concentration in each dose, analyze the product on an analytical HPLC for QC and then inject the product with standard cold compound to establish its identity and purity (Fig. 5).
Figure 5
Radiotracer dosing and administration
-
35
Prepare the radiotracer, either [18F]FDG or [18F]FEAU, at a concentration of 2 mCi ml−1 (74 MBq ml−1) in saline to inject the animals for PET imaging. For autoradiography, prepare a solution at a concentration of 5 mCi ml−1 (185 MBq ml−1).
Caution
Radioactive, should be performed in a hot cell or behind lead shielding.
Animal preparation and tumor growth
-
36
Anesthetize animals (nine mice per group) by gas inhalation (2% vol/vol isoflurane in oxygen) and inject (s.c.) tumor cells of interest into the deltoid muscle area of each mouse. This will produce tumor xenografts located away from urinary system (particularly bladder), thus avoiding artifacts caused by accumulation of high activity in urine. To generate xenografts, use a tumor cell dose adequate for each line.
-
37
When the tumor xenograft is approximately 0.5 cm in diameter, inject a single i.v. dose of RGD-4C AAVP-HSV-tk as described in an accompanying protocol28,33.
-
38
Take another group of tumor-bearing mice (n = 9) and inject them with non-targeted AAVP-HSV-tk. This group will serve as the experimental control.
Animal imaging on a micro-PET using radiotracer (day 4 after AAVP injection): [18F]FDG scan
-
39
Remove food from animal cages 5 h before injecting [18F]FDG for PET imaging.
-
40
Anesthetize the animals by gas inhalation (2% vol/vol isoflurane in oxygen) and inject [18F]FDG (100 μCi, 3.7 MBq, per animal) either via the caudal vein or through supra-orbital administration. Injections of [18F]FDG into the animals must be performed at an interval of 15 min to ensure a similar uptake period before imaging.
-
41
After [18F]FDG injection, monitor the animal until full recovery from anesthesia and put back into the cage. For PET imaging, anesthetize the animal once again 55 min after [18F]FDG injection so that it will be ready for imaging at 1 h after injection.
-
42
Place the animal on the scanner bed on a warming pad to maintain thermal regulation, and continue administration of the anesthetic gas so that the animal cannot move during scanning. If the animal moves during imaging, clean images cannot be obtained.
-
43
Image the animals at 1 h after injection (static 15-min acquisition).
-
44
After scanning, remove the animal from the scanner bed and image the next animal. Continue imaging all animals in the same way. Change the absorbing pad after each animal imaging session.
[18F]FEAU scan (day 5)
-
45
Scan the animals (static 15-min acquisition) using [18F]FEAU PET 2 h after injection as described above (Steps 40–44).
-
46
Image all animals again on day 7 and day 10 after the injection of the AAVP to determine when maximum gene expression occurs.
-
47
Treat the same group of animals (imaged with [18F]FDG and [18F]FEAU) with ganciclovir (80 mg kg−1d−1, i.p.) for 3 d and repeat PET scans with [18F]FEAU followed by [18F]FDG before killing.
Image reconstruction, analysis and interpretation
-
48
Reconstruct images using a standard reconstruction protocol such as the ordered subset expectation maximization algorithm (number of iterations = 4; number of subsets = 16) with 2D mode, without back filter.
-
49
Estimate regional tumor radioactivity concentrations (kBq cm−3) from the maximum pixels within regions of interest drawn around the tumor on transaxial slices of the reconstructed image sets.
-
50
Quantify radioactivity uptake ([18F]FDG or [18F]FEAU) in tumors and other organs as follows: draw the region of interest on images, calculate the volume and activity uptake (nBq mm−3) in the region of interest and then convert into percentage injected dose per gram (%ID g−1).
-
51
Calculate the mean level of activity uptake and s.d. Compare the control tissue (muscle in opposite deltoid area) and tumors. Calculate the tumor/muscle radiotracer uptake ratio between tumor and muscle by dividing the radioactivity concentration in the tumor by the radioactivity concentration in the muscle in the same animal. Calculate erythematic mean and s.d. of animal groups. In the case of non-Gaussian distribution and difference in inter-group variances, calculate medians and ranges instead.
-
52
Use two-tailed Student's t-test to compare unrelated (independent) groups with equal variances (F-test) and Gaussian distribution of parameters; otherwise, use non-parametric tests (e.g., Mann–Whitney U-test) to compare control and treated groups.
Troubleshooting
Troubleshooting advice can be found in Table 1.
Timing
The procedure for production of [18F]FEAU requires approximately 3.5 h, including bombardment for isotope production, synthesis and dose preparation. Steps 15–16: 45 min; Steps 17–18: 20 min; Steps 19–24: 30 min; Steps 25–27: 35 min; Steps 28–30: 40 min; Steps 31–34: 45 min. For tumor growth, injection of AAVP and imaging before and after therapy, see refs. 28,33.
Anticipated results
Preparation of 2-deoxy-2-trifluoromethanesulfonyl-1,3,5-tri-O-benzoyl-α-D-ribofuranose 1
The overall yield for compound 1 (Steps 1–6) should be more than 95%. In case of low yields, refer to Table 1. See NMR (analytical data); TLC: 15% acetone/hexane, Rf = 0.5.
Preparation of 2,4-bis-trimethylsilyl-5-ethyl uracil 4
The yield of compound 4 (Steps 9–14) is quantitative, more than 99%. In case of low yields, refer to Table 1.
Preparation of [2-18F]fluoro-1,3,5-tri-O-benzoyl-α-D-arabinofuranose 2
The radiochemical yield in these steps (Steps 17–24) should be in the range 35–55%. In case of low yields, refer to Table 1.
Yield in the coupling reaction (Steps 25–34) should be 70–80% (d.c.). In case of low yields, refer to Table 1.
Overall yield of [18F]FEAU (Steps 17–34) should be 15–20% from the end of bombardment.
In a typical experiment, 10 mCi (370 MBq) [18F]FEAU is expected from 100 mCi (3.7 GBq) [18F]fluoride.
PET images with [18F]FDG reflect tumor uptake for glucose metabolism, an indication of viable cancer cells.
PET images with [18F]FEAU reflect tumor uptake for HSV1-tk gene expression at the targeted tumor, an indication of HSV1-tk gene expression. The gene product HSV1-tk (enzyme) phosphorylated [18F]FEAU inside the transgene-expressing cells and 18F[FEAU] is trapped.
Maximum gene expression takes place on day 10 after the targeted AAVP vector injection.
Figure 6 is a molecular PET image of HSV1-tk targeted gene expression. The upper panel shows PET images of the control group of animals, where only uptake of FDG is observed. The lower panel shows PET images of the experimental group of animals injected with the targeting vector. PET with [18F]FDG reflects glucose metabolism, and PET with [18F]FEAU uptake reflects HSV1-tk gene expression. Following ganciclovir treatment, no [18F]FEAU uptake is observed. In contrast, some [18F]FDG uptake is observed, which suggests that some cancer cells remain viable following treatment, and only transduced cells are killed by the treatment.
Upper panel: non-targeted AAVP-HSV-tk tumor-bearing animals imaged with [18F]FDG and [18F]FEAU. Lower panel: RGD-4C AAVP-HSV-tk tumor-bearing animals imaged with [18F]FDG and [18F]FEAU. The two left-hand images are before treatment and the two right-hand images are after treatment. [18F]FDG uptake indicates proliferating cells and [18F]FEAU uptake indicates HSV1-tk gene expression. The arrows point to tumors.
Analytical data
Compound 1: 1H NMR (300 MHz, CDCl3) δ: 8.10-8.00 (m, 6H, aromatic), 7.76-7.65 (m, 3H, aromatic), 7.53-7.41 (m, 6H, aromatic), 6.88 (d, J = 4.2 Hz, 1H, H-1), 5.80 (m, 1H, H-2), 5.56 (m, 1H, H-3), 4.87 (q, J = 3 Hz, 1H, H-4), 4.70 (dd, 2H, H-5).
19F NMR (300 MHz, CDCl3), δ: −74.46 (s).
FEAU: 1H NMR (300 MHz, DMSO-d6), δ: 10.7 (bs, 1H, NH), 7.55 (s, 1H, H-6′), 6.1 (dd, J = 14 Hz, 4 Hz, 1H, H-1′), 5.89 (s, 1H, OH), 5.17 (s, 1H, OH), 5.05 (d, J = 53 Hz, 1H, H-2′), 4.23 (d, J = 20 Hz, 1H, H-3′), 3.75 (d, J = 3 Hz, 1H, H-4′), 3.67–3.56 (m, 2H, H-5′), 2.20 (q, J = 7.4 Hz, 2H, ethyl CH2), 1.00 (t, J = 7.4 Hz, 3H, ethyl CH3).
19F NMR (300 MHz, DMSO-d6), δ: −197.2 (m).
References
Tjuvajev, J.G. et al. Imaging the expression of transfected genes in vivo . Cancer Res. 55, 6126–6132 (1995).
Tjuvajev, J.G. et al. Noninvasive imaging of herpes virus thymidine kinase gene transfer and expression: a potential method for monitoring clinical gene therapy. Cancer Res. 56, 4087–4095 (1996).
Tjuvajev, J.G. et al. Imaging herpes virus thymidine kinase gene transfer and expression by positron emission tomography. Cancer Res. 58, 4333–4341 (1998).
Tjuvajev, J.G. et al. Comparison of radiolabeled nucleoside probes (FIAU, FHBG, and FHPG) for PET imaging of HSV1-tk gene expression. J. Nucl. Med. 43, 1072–1083 (2002).
Alauddin, M.M., Conti, P.S., Mazza, S.M., Hamzeh, F.H. & Lever, J.R. 9-[(3-[18F]fluoro-1-hydroxy-2-propoxy)methyl]guanine ([18F]FHPG): a potential imaging agent of viral infection and gene therapy using PET. Nucl. Med. Biol. 23, 787–792 (1996).
Alauddin, M.M. & Conti, P.S. Synthesis and preliminary evaluation of 9-(4-[18F]-fluoro-3-hydroxymethylbutyl)guanine ([18F]FHBG): a new potential imaging agent for viral infection and gene therapy using PET. Nucl. Med. Biol. 25, 175–180 (1998).
Alauddin, M.M., Shahinian, A., Kundu, R.K., Gordon, E.M. & Conti, P.S. Evaluation of 9-(3-[18F]-fluoro-1-hydroxy-2-propoxy)methyl]guanine ([18F]-FHPG) in vitro and in vivo as a probe for PET imaging of gene incorporation and expression in tumors. Nucl. Med. Biol. 26, 371–376 (1999).
Alauddin, M.M., Shahinian, A., Gordon, E.M., Bading, J.R. & Conti, P.S. Pre-clinical evaluation of the penciclovir analog 9-(4-[18F]-fluoro-3-hydroxymethylbutyl)guanine for in vivo measurement of suicide gene expression with PET. J. Nucl. Med. 42, 1682–1690 (2001).
Jacobs, A. et al. Positron emission tomography-based imaging of transgene expression mediated by replication-conditional, oncolytic herpes simplex virus type-1 mutant vectors in vivo . Cancer Res. 61, 2983–2995 (2001).
Haberkorn, U. et al. Monitoring gene therapy with herpes simplex virus thymidine kinase in hepatoma cells: uptake of specific substrates. J. Nucl. Med. 38, 287–294 (1997).
Tjuvajev, J.G. et al. Imaging adenoviral-mediated herpes virus thymidine kinase gene transfer and expression in vivo . Cancer Res. 59, 5186–5193 (1999).
Haberkorn, U. et al. Ganciclovir uptake in human mammary carcinoma cells expressing herpes simplex virus thymidine kinase. Nucl. Med. Biol. 25, 367–373 (1998).
Gambhir, S.S. et al. Imaging adenoviral-directed reporter gene expression in living animals with positron emission tomography. Proc. Natl. Acad. Sci. USA 96, 2333–2338 (1999).
Yu, Y. et al. Quantification of gene expression by imaging reporter gene expression in living animals. Nat. Med. 6, 933–937 (2000).
Tjuvajev, J.G. et al. A general approach to the non-invasive imaging of transgenes using cis-linked herpes virus thymidine kinase. Neoplasia 1, 315–320 (1999).
Iyer, M. et al. 8-[18F]-Fluoropenciclovir: an improved reporter probe for imaging HSV-tk reporter gene expression in vivo using PET. J. Nucl. Med. 42, 96–105 (2001).
Alauddin, M.M., Fissekis, J.D. & Conti, P.S. Synthesis of [18F]-labeled 2′-deoxy-2′-fluoro-5-methyl-1-β-d-arabinofuranosyluracil ([18F]-FMAU). J. Labelled Compd. Radiopharm. 45, 583–590 (2002).
Alauddin, M.M., Shahinian, A., Gordon, E.M. & Conti, P.S. Evaluation of 2′-deoxy-2′-fluoro-5-methyl-1-β-d-arabinofuranosyluracil as a potential gene imaging agent for HSV-tk expression in vivo . Mol. Imaging 1, 74–81 (2002).
Alauddin, M.M., Fissekis, J.D. & Conti, P.S. A general synthesis of 2′-deoxy-2′-[18F]fluoro-5-methyl-1-β-d-arabinofuranosyluracil and its 5-substituted nucleosides. J. Labelled Compd. Radiopharm. 4, 285–289 (2003).
Alauddin, M.M. et al. Synthesis and evaluation of 2′-deoxy-2′-[18F]- fluoro-5-fluoro-1-beta-d-arabinofuranosyluracil as a potential PET imaging agent for gene expression. J. Nucl. Med. 45, 2063–2069 (2004).
Alauddin, M.M. et al. Synthesis of 2′-deoxy-2′-[18F]fluoro-5-bromo-1-beta-d-arabinofuranosyluracil ([18F]FBAU) and 2′-deoxy-2′-[18F]fluoro-5-chloro-1-beta-d-arabinofuranosyluracil ([18F]FCAU), and their biological evaluation as markers for gene expression. Nucl. Med. Biol. 31, 399–405 (2004).
Alauddin, M.M. et al. In vivo evaluation of 2′-deoxy-2′-[18F]fluoro-5-iodo-1-beta-d-arabinofuranosyluracil ([18F]-FIAU) and 2′-deoxy-2′-[18F]fluoro-5-ethyl-1-beta-d-arabinofuranosyluracil ([18F]-FEAU) as markers for suicide gene expression. Eur. J. Nucl. Med. Mol. Imaging (Jan 6 2007, E. Pub).
Alauddin, M.M., Shahinian, A. & Conti, P.S. Evaluation of 2′-deoxy-2′-fluoro-5-substituted-1-beta-d-arabinofuranosyluracils as markers for suicide gene expression in breast cancer cells. J. Nucl. Med. 46, 35 (2005).
Buursma, A.R. et al. 18F-FEAU as a radiotracer for herpes simplex virus thymidine kinase gene expression: in vitro comparison with other PET tracers. Nucl. Med. Commun. 27, 25–30 (2006).
Kang, K.W., Min, J.J., Chen, X. & Gambhir, S.S. Comparison of [14C]FMAU, [3H]FEAU, [14C]FIAU and [3H]PCV for monitoring reporter gene expression with wild type and mutant herpes simplex virus type 1 thymidine kinase in cell culture. Mol. Imaging Biol. 7, 296–303 (2005).
Tseng, J.C. et al. Tumor specific in vivo transfection with HSV-1 thymidine kinase gene using a Sindbis viral vector as a basis for prodrug ganciclovir activation and PET. J. Nucl. Med. 47, 1136–1143 (2006).
Hajitou, A., Pasqualini, R. & Arap, W. Vascular targeting: recent advances and therapeutic perspectives. Trends Cardiovasc. Med. 16, 80–88 (2006).
Hajitou, A. et al. A hybrid vector for ligand-directed tumor targeting and molecular imaging. Cell 125, 385–398 (2006).
Ivanenkov, V.V., Felici, F. & Menon, A.G. Targeted delivery of multivalent phage display vectors into mammalian cells. Biochim. Biophys. Acta 1448, 463–472 (1999).
Larocca, D. et al. Gene transfer to mammalian cells using genetically targeted filamentous bacteriophage. FASEB J. 13, 727–734 (1999).
Poul, M.A. & Marks, J.D. Targeted gene delivery to mammalian cells by filamentous bacteriophage. J. Mol. Biol. 288, 203–211 (1999).
Sergeeva, A., Kolonin, M.G., Molldrem, J.J., Pasqualini, R. & Arap, W. Display technologies: application for the discovery of drug and gene delivery agents. Adv. Drug Deliv. Rev. 58, 1622–1654 (2006).
Hajitou, A. et al. Design and construction of targeted AAVP for mammalian cell transduction. Nat. Protoc. DOI: 10.1038/nprot.2007.51.
Alauddin, M.M. et al. Stereospecific fluorination of 1,3,5-tri-O-benzoyl-α-d-ribofuranise-2-sufonate esters: preparation of a versatile intermediate for synthesis of 2′-[18F]-fluoro-arabinonucleosides. J. Fluorine Chem. 106, 87–91 (2000).
Tann, C.H., Brodfuehrer, P.R., Brundidge, S.P., Sapino, C. Jr & Howell, H.G. Fluorocarbohydrtaes in synthesis. An efficient synthesis of 1-(2-deoxy-2-fluoro-β-d-arabimofuranosyluracil)-5-iodouracil (β-FIAU) and 1-(2-deoxy-2-fluoro-β-d-arabimofuranosyl)thymine (β-FMAU). J. Org. Chem. 50, 3644–3647 (1985).
Watanabe, K.A. et al. Nucleosides. 129. Synthesis of antiviral nucleosides: 5-alkenyl-1-(2-deoxy-2-fluoro-β-d-arabionofuranosyl)uracils. J. Med. Chem. 27, 91–94 (1984).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Soghomonyan, S., Hajitou, A., Rangel, R. et al. Molecular PET imaging of HSV1-tk reporter gene expression using [18F]FEAU. Nat Protoc 2, 416–423 (2007). https://doi.org/10.1038/nprot.2007.49
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2007.49
This article is cited by
-
Targeted AAVP-based therapy in a mouse model of human glioblastoma: a comparison of cytotoxic versus suicide gene delivery strategies
Cancer Gene Therapy (2020)
-
Imaging of Sleeping Beauty-Modified CD19-Specific T Cells Expressing HSV1-Thymidine Kinase by Positron Emission Tomography
Molecular Imaging and Biology (2016)
-
Targeting mammalian organelles with internalizing phage (iPhage) libraries
Nature Protocols (2013)
-
Synthesis of a probe for monitoring HSV1-tk reporter gene expression using chemical exchange saturation transfer MRI
Nature Protocols (2013)
-
PET imaging of T cells derived from umbilical cord blood
Leukemia (2009)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.