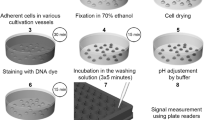
Abstract
One of the hallmarks of cell death is the cell surface–expression of phosphatidylserine. Expression of phosphatidylserine at the cell surface can be measured in vitro with the phosphatidylserine-binding protein annexin A5 conjugated to fluorochromes. This measurement can be made by flow cytometry or by confocal scanning-laser microscopy. The annexin A5 affinity assay comprises the incubation of cells stimulated to execute cell death with fluorescence-labeled annexin A5 and propidium iodide. Living cells are annexin A5–negative and propidium iodide negative, cells in the early phases of cell death are annexin A5 positive–and propidium iodide–negative, and secondary necrotic cells are annexin A5–positive and propidium iodide–positive. The entire procedure takes about 30 minutes for flow cytometry and 45 minutes for confocal scanning-laser microscopy. Various precautions and considerations are discussed further in the protocol described here.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Balasubramanian, K. & Schroit, A.J. Aminophospholipid asymmetry: a matter of life and death. Annu. Rev. Physiol. 65, 701–734 (2003).
Savill, J., Dransfield, I., Gregory, C. & Haslett, C. A blast from the past: clearance of apoptotic cells regulates immune responses. Nat. Rev. Immunol. 2, 965–975 (2002).
Vermes, I., Haanen, C., Steffens-Nakken, H. & Reutelingsperger, C. A novel assay for apoptosis. Flow cytometric detection of phosphatidylserine expression on early apoptotic cells using fluorescein labelled Annexin V. J. Immunol. Methods 184, 39–51 (1995).
van Engeland, M., Nieland, L.J., Ramaekers, F.C., Schutte, B. & Reutelingsperger, C.P. Annexin V-affinity assay: a review on an apoptosis detection system based on phosphatidylserine exposure. Cytometry 31, 1–9 (1998).
Kroemer, G. et al. Classification of cell death: recommendations of the Nomenclature Committee on Cell Death. Cell Death Differ. 12, 1463–1467 (2005).
Corsten, M.F., Hofstra, L., Narula, J. & Reutelingsperger, C.P. Counting heads in the war against cancer: defining the role of annexin A5 imaging in cancer treatment and surveillance. Cancer Res. 66, 1255–1260 (2006).
Martin, S.J. et al. Early redistribution of plasma membrane phosphatidylserine is a general feature of apoptosis regardless of the initiating stimulus: inhibition by overexpression of Bcl-2 and Abl. J. Exp. Med. 182, 1545–1556 (1995).
Martin, S.J., Finucane, D.M., Amarante-Mendes, G.P., O'Brien, G.A. & Green, D.R. Phosphatidylserine externalization during CD95-induced apoptosis of cells and cytoplasts requires ICE/CED-3 protease activity. J. Biol. Chem. 271, 28753–28756 (1996).
Foghsgaard, L. et al. Cathepsin B acts as a dominant execution protease in tumor cell apoptosis induced by tumor necrosis factor. J. Cell Biol. 153, 999–1010 (2001).
Callahan, M.K. et al. Phosphatidylserine expression and phagocytosis of apoptotic thymocytes during differentiation of monocytic cells. J. Leukoc. Biol. 74, 846–856 (2003).
Elliott, J.I. et al. Membrane phosphatidylserine distribution as a non-apoptotic signaling mechanism in lymphocytes. Nat. Cell Biol. 7, 808–816 (2005).
Dillon, S.R., Constantinescu, A. & Schlissel, M.S. Annexin V binds to positively selected B cells. J. Immunol. 166, 58–71 (2001).
Dias-Baruffi, M. et al. Dimeric galectin-1 induces surface exposure of phosphatidylserine and phagocytic recognition of leukocytes without inducing apoptosis. J. Biol. Chem. 278, 41282–41293 (2003).
van den Eijnde, S.M. et al. Transient expression of phosphatidylserine at cell-cell contact areas is required for myotube formation. J. Cell Sci. 114, 3631–3642 (2001).
Gerke, V., Creutz, C.E. & Moss, S.E. Annexins: linking Ca2+ signalling to membrane dynamics. Nat. Rev. Mol. Cell Biol. 6, 449–461 (2005).
van Engeland, M., Ramaekers, F.C., Schutte, B. & Reutelingsperger, C.P. A novel assay to measure loss of plasma membrane asymmetry during apoptosis of adherent cells in culture. Cytometry 24, 131–139 (1996).
Hughes, D. & Mehmet, H. Cell Proliferation and Apoptosis. 233–254, Ch. 9 (BIOS Scientific, Oxford, UK, 2003).
Pozarowski, P. et al. Interactions of fluorochrome-labeled caspase inhibitors with apoptotic cells: a caution in data interpretation. Cytometry A. 55, 50–60 (2003).
Gavrieli, Y., Sherman, Y. & Ben-Sasson, S.A. Identification of programmed cell death in situ via specific labeling of nuclear DNA fragmentation. J. Cell Biol. 119, 493–501 (1992).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
van Genderen, H., Kenis, H., Lux, P. et al. In vitro measurement of cell death with the annexin A5 affinity assay. Nat Protoc 1, 363–367 (2006). https://doi.org/10.1038/nprot.2006.55
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2006.55
This article is cited by
-
Low human and murine Mcl-1 expression leads to a pro-apoptotic plaque phenotype enriched in giant-cells
Scientific Reports (2019)
-
Monitoring the progression of cell death and the disassembly of dying cells by flow cytometry
Nature Protocols (2016)
-
Quantitative analysis of annexin V–membrane interaction by flow cytometry
European Biophysics Journal (2015)
-
Phosphatidylserine exposure during apoptosis reflects bidirectional trafficking between plasma membrane and cytoplasm
Cell Death & Differentiation (2013)
-
Fractional proliferation: a method to deconvolve cell population dynamics from single-cell data
Nature Methods (2012)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.