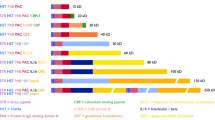
Abstract
Here, we present a cloning strategy for the production of recombinant proteins tagged with a polyhistidine sequence that can be cleaved by the exopeptidase, DAPase. The method can be used with most commonly available vectors and results in the expression of a His-tag protein that can be purified in its native form regardless of its natural sequence. This approach takes advantage of the TAGZyme system for the removal of amino-terminal affinity tags. Tag removal is accomplished either with DAPase (a recombinant dipeptidyl peptidase) alone or in combination with two accessory enzymes, Qcyclase and pGAPase. The system has been used for the production of intracellular proteins in Escherichia coli and can be applied to other expression hosts for the production of secreted proteins or proteins that require post-translational modification. The production of human interleukin 1β in E. coli is used as an example to illustrate this method. The complete protocol from initial PCR to the production of a detagged protein with its authentic N terminus can be performed within 5 days.
NOTE: In the version of this article initially published online, the text for Step 21 of the procedure was incorrect. It should read: “Prepare an overnight culture (1.8 l) and centrifuge in a Sorvall for 20 min at 10,000g, 4 °C.” This error has been corrected in all versions of the article.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
22 February 2007
altered some text in step 21
References
Arnau, J., Lauritzen, C., Petersen, G.E. & Pedersen, J. Current strategies for the use of affinity tags and tag removal for the purification of recombinant proteins. Protein Expr. Purif. 48, 1–13 (2006).
Pedersen, J., Lauritzen, C., Madsen, M.T. & Weis Dahl, S. Removal of N-terminal polyhistidine tags from recombinant proteins using engineered aminopeptidases. Protein Expr. Purif. 15, 389–400 (1999).
Cebe, R. & Geiser, M. Rapid and easy thermodynamic optimization of the 5′-end of mRNA dramatically increases the level of wild type protein expression in Escherichia coli. Protein Expr. Purif. 45, 374–380 (2006).
Svensson, J., Andersson, C., Reseland, J.E., Lyngstadaas, P. & Bulow, L. Histidine tag fusion increases expression levels of active recombinant amelogenin in Escherichia coli. Protein Expr. Purif. 48, 134–141 (2006).
Dahl, S.W. et al. Carica papaya glutamine cyclotransferase belongs to a novel plant enzyme subfamily: cloning and characterization of the recombinant enzyme. Protein Expr. Purif. 20, 27–36 (2000).
Looman, A.C. et al. Influence of the codon following AUG initiation codon on the expression of a modified lacZ gene in Escherichia coli. EMBO J. 6, 2489–2492 (1987).
Dalbøge, H., Bayne, S. & Pedersen, J. In vivo processing of N-terminal methionine in E.coli. FEBS Lett. 266, 1–3 (1990).
Schägger, H. Tricine SDS-PAGE. Nature Protocols 1, 16–22 (2006).
Cabrita, L.D., Dai, W. & Bottomley, S.P. A family of E. coli expression vectors for laboratory scale and high throughput soluble protein production. BMC Biotechnology 6, 12 (2006).
Acknowledgements
Current and past Unizyme staff are greatly acknowledged for their contribution to the development of the TAGZyme system.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Arnau, J., Lauritzen, C. & Pedersen, J. Cloning strategy, production and purification of proteins with exopeptidase-cleavable His-tags. Nat Protoc 1, 2326–2333 (2006). https://doi.org/10.1038/nprot.2006.388
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2006.388
This article is cited by
-
A novel protein purification scheme based on salt inducible self-assembling peptides
Microbial Cell Factories (2023)
-
Rapid online buffer exchange for screening of proteins, protein complexes and cell lysates by native mass spectrometry
Nature Protocols (2020)
-
Poly(amino acid)s/polypeptides as potential functional and structural materials
Polymer Journal (2015)
-
Exploiting the interactions between poly-histidine fusion tags and immobilized metal ions
Biotechnology Letters (2011)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.