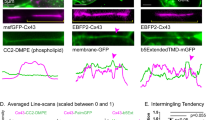
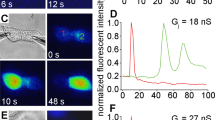
Abstract
Vertebrate gap junction channels are formed by a family of more than 20 connexin proteins. These gap junction proteins are expressed with overlapping cellular and tissue specificity, and coding region mutations can cause human hereditary diseases. Here we present a summary of what has been learned from voltage clamp studies performed on cell pairs either endogenously expressing gap junctions or in which connexins are exogenously expressed. General protocols presented here are currently used to transfect mammalian cells with connexins and to study the biophysical properties of the heterologously expressed connexin channels. Transient transfection is accomplished overnight with maximal expression occurring at about 36 h; stable transfectants normally can be generated within three or four weeks through colony selection. Electrophysiological protocols are presented for analysis of voltage dependence and single-channel conductance of gap junction channels as well as for studies of chemical gating of these channels.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
Sohl, G. & Willecke, K. Gap junctions and the connexin protein family. Cardiovasc. Res. 62, 228–232 (2004).
Kumar, N.M. & Gilula, N.B. The gap junction communication channel. Cell 84, 381–388 (1996).
Spray, D.C., Rozental, R. & Srinivas, M. Prospects for rational development of pharmacological gap junction channel blockers. Curr. Drug Targets 3, 455–464 (2002).
Spray, D.C. Illuminating gap junctions. Nat. Methods 2, 12–14 (2005).
Bennett, M.V. Physiology of electrotonic junctions. Ann. NY Acad. Sci. 137, 509–539 (1966).
Spray, D.C., Harris, A.L. & Bennett, M.V. Voltage dependence of junctional conductance in early amphibian embryos. Science 204, 432–434 (1979).
Spray, D.C., Harris, A.L. & Bennett, M.V. Equilibrium properties of a voltage-dependent junctional conductance. J. Gen. Physiol 77, 77–93 (1981).
Spray, D.C., Harris, A.L. & Bennett, M.V. Gap junctional conductance is a simple and sensitive function of intracellular pH. Science 211, 712–715 (1981).
Harris, A.L., Spray, D.C. & Bennett, M.V. Kinetic properties of a voltage-dependent junctional conductance. J. Gen. Physiol. 77, 95–117 (1981).
Spray, D.C., Ginzberg, R.D., Morales, E.A., Gatmaitan, Z. & Arias, I.M. Electrophysiological properties of gap junctions between dissociated pairs of rat hepatocytes. J. Cell Biol. 103, 135–144 (1986).
Rook, M.B., Jongsma, H.J. & van Ginneken, A.C. Properties of single gap junctional channels between isolated neonatal rat heart cells. Am. J. Physiol. 255, H770–H782 (1988).
Burt, J.M. & Spray, D.C. Single-channel events and gating behavior of the cardiac gap junction channel. Proc. Natl. Acad. Sci. USA 85, 3431–3434 (1988).
Veenstra, R.D. & DeHaan, R.L. Measurement of single channel currents from cardiac gap junctions. Science 233, 972–974 (1986).
Neyton, J. & Trautmann, A. Single-channel currents of an intercellular junction. Nature 317, 331–335 (1985).
Kumar, N.M. & Gilula, N.B. Cloning and characterization of human and rat liver cDNAs coding for a gap junction protein. J. Cell Biol. 103, 767–776 (1986).
Paul, D.L. Molecular cloning of cDNA for rat liver gap junction protein. J. Cell Biol. 103, 123–134 (1986).
Beyer, E.C., Paul, D.L. & Goodenough, D.A. Connexin43: a protein from rat heart homologous to a gap junction protein from liver. J. Cell Biol. 105, 2621–2629 (1987).
Dahl, G., Miller, T., Paul, D., Voellmy, R. & Werner, R. Expression of functional cell-cell channels from cloned rat liver gap junction complementary DNA. Science 236, 1290–1293 (1987).
Oh, S. et al. Changes in permeability caused by connexin 32 mutations underlie X-linked Charcot-Marie-Tooth disease. Neuron 19, 927–938 (1997).
Ressot, C., Gomes, D., Dautigny, A., Pham-Dinh, D. & Bruzzone, R. Connexin32 mutations associated with X-linked Charcot-Marie-Tooth disease show two distinct behaviors: loss of function and altered gating properties. J. Neurosci. 18, 4063–4075 (1998).
Moreno, A.P., de Carvalho, A.C., Verselis, V., Eghbali, B. & Spray, D.C. Voltage-dependent gap junction channels are formed by connexin32, the major gap junction protein of rat liver. Biophys. J. 59, 920–925 (1991).
Eghbali, B., Kessler, J.A. & Spray, D.C. Expression of gap junction channels in communication-incompetent cells after stable transfection with cDNA encoding connexin 32. Proc. Natl. Acad. Sci. USA 87, 1328–1331 (1990).
Fishman, G.I., Spray, D.C. & Leinwand, L.A. Molecular characterization and functional expression of the human cardiac gap junction channel. J. Cell Biol. 111, 589–598 (1990).
Veenstra, R.D., Wang, H.Z., Westphale, E.M. & Beyer, E.C. Multiple connexins confer distinct regulatory and conductance properties of gap junctions in developing heart. Circ. Res. 71, 1277–1283 (1992).
Merickel, M. Design of a single electrode voltage clamp. J. Neurosci. Methods 2, 87–96 (1980).
White, R.L., Spray, D.C., Campos de Carvalho, A.C., Wittenberg, B.A. & Bennett, M.V. Some electrical and pharmacological properties of gap junctions between adult ventricular myocytes. Am. J. Physiol. 249, C447–C455 (1985).
Ek-Vitorin, J.F. & Burt, J.M. Quantification of gap junction selectivity. Am. J. Physiol. Cell Physiol. 289, C1535–C1546 (2005).
Neher, E. & Sakmann, B. Single-channel currents recorded from membrane of denervated frog muscle fibres. Nature 260, 799–802 (1976).
Sakmann, B. & Neher, E. Single-Channel Recording (Plenum Press, New York, 1995).
Bukauskas, F.F., Elfgang, C., Willecke, K. & Weingart, R. Biophysical properties of gap junction channels formed by mouse connexin40 in induced pairs of transfected human HeLa cells. Biophys. J. 68, 2289–2298 (1995).
Moreno, A.P., Rook, M.B., Fishman, G.I. & Spray, D.C. Gap junction channels: distinct voltage-sensitive and -insensitive conductance states. Biophys. J. 67, 113–119 (1994).
Srinivas, M. et al. Voltage dependence of macroscopic and unitary currents of gap junction channels formed by mouse connexin50 expressed in rat neuroblastoma cells. J. Physiol. 517, 673–689 (1999).
Bukauskas, F.F. & Peracchia, C. Two distinct gating mechanisms in gap junction channels: CO2-sensitive and voltage-sensitive. Biophys. J. 72, 2137–2142 (1997).
Hopperstad, M.G., Srinivas, M. & Spray, D.C. Properties of gap junction channels formed by Cx46 alone and in combination with Cx50. Biophys. J. 79, 1954–1966 (2000).
Trexler, E.B., Bennett, M.V., Bargiello, T.A. & Verselis, V.K. Voltage gating and permeation in a gap junction hemichannel. Proc. Natl. Acad. Sci. USA 93, 5836–5841 (1996).
Bukauskas, F.F. & Verselis, V.K. Gap junction channel gating. Biochim. Biophys. Acta 1662, 42–60 (2004).
Brink, P.R. et al. Evidence for heteromeric gap junction channels formed from rat connexin43 and human connexin37. Am. J. Physiol. 273, C1386–C1396 (1997).
Reed, K.E. et al. Molecular cloning and functional expression of human connexin37, an endothelial cell gap junction protein. J. Clin. Invest. 91, 997–1004 (1993).
Srinivas, M. et al. Functional properties of channels formed by the neuronal gap junction protein connexin36. J. Neurosci. 19, 9848–9855 (1999).
Teubner, B. et al. Functional expression of the murine connexin 36 gene coding for a neuron-specific gap junctional protein. J. Membr. Biol. 176, 249–262 (2000).
Harris, A.L. Emerging issues of connexin channels: biophysics fills the gap. Q. Rev. Biophys. 34, 325–472 (2001).
Verselis, V.K., Ginter, C.S. & Bargiello, T.A. Opposite voltage gating polarities of two closely related connexins. Nature 368, 348–351 (1994).
Revilla, A., Castro, C. & Barrio, L.C. Molecular dissection of transjunctional voltage dependence in the connexin-32 and connexin-43 junctions. Biophys. J. 77, 1374–1383 (1999).
Moreno, A.P., et al. Role of the carboxyl terminal of connexin-43 in transjunctional fast voltage gating. Circ. Res. 90, 450–457 (2002).
Duffy, H.S. et al. pH-dependent intramolecular binding and structure involving Cx43 cytoplasmic domains. J. Biol. Chem. 277, 36706–36714 (2002).
Ek-Vitorin, J.F. et al. PH regulation of connexin43: molecular analysis of the gating particle. Biophys. J. 71, 1273–1284 (1996).
Cruikshank, S.J. et al. Potent block of Cx36 and Cx50 gap junction channels by mefloquine. Proc. Natl. Acad. Sci. USA 101, 12364–12369 (2004).
Srinivas, M., Hopperstad, M.G. & Spray, D.C. Quinine blocks specific gap junction channel subtypes. Proc. Natl. Acad. Sci. USA 98, 10942–10947 (2001).
Bai, D., del Corsso, C., Srinivas, M. & Spray, D.C. Block of specific gap junction channel subtypes by 2–aminoethoxydiphenyl borate (2-APB). J. Pharmacol. Exp. Therapeut. published online 19 September 2006 (PMID: 16985167).
Contreras, J.E., Saez, J.C., Bukauskas, F.F. & Bennett, M.V. Gating and regulation of connexin 43 (Cx43) hemichannels. Proc. Natl. Acad. Sci. USA 100, 11388–11393 (2003).
Eckert, R., Dunina-Barkovskaya, A. & Hulser, D.F. Biophysical characterization of gap-junction channels in HeLa cells. Pflugers Arch. 424, 335–342 (1993).
Acknowledgements
We gratefully acknowledge the many talented graduate students and post doctoral fellows who have worked with us over the years since voltage clamping was first applied to gap junctions. Our research has been supported by grants from the National Institutes of Health (NIH) to D.C.S. and colleagues as well as foreign grants to investigators visiting the laboratory from other countries.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
del Corsso, C., Srinivas, M., Urban-Maldonado, M. et al. Transfection of mammalian cells with connexins and measurement of voltage sensitivity of their gap junctions. Nat Protoc 1, 1799–1809 (2006). https://doi.org/10.1038/nprot.2006.266
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2006.266
This article is cited by
-
Pannexin1-Mediated ATP Release Provides Signal Transmission Between Neuro2A Cells
Neurochemical Research (2012)
-
Engineering biosynthetic excitable tissues from unexcitable cells for electrophysiological and cell therapy studies
Nature Communications (2011)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.