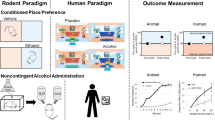
Abstract
Laboratory models, including animal tissues and live animals, have proven useful for discovery of molecular targets of alcohol action as well as for characterization of genetic and environmental factors that influence alcohol's neural actions. Here we consider strengths and weaknesses of laboratory models used in alcohol research and analyze the limitations of using animals to model a complex human disease. We describe targets for the neural actions of alcohol, and we review studies in which animal models were used to examine excessive alcohol drinking and to discover genes that may contribute to risk for alcoholism. Despite some limitations of the laboratory models used in alcohol research, these experimental approaches are likely to contribute to the development of new therapies for alcohol abuse and alcoholism.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Ann Thomson
Similar content being viewed by others
References
Diamond, I. & Gordon, A.S. Cellular and molecular neuroscience of alcoholism. Physiol. Rev. 77, 1–20 (1997).
Schuckit, M.A., Smith, T.L. & Kalmijn, J. The search for genes contributing to the low level of response to alcohol: patterns of findings across studies. Alcohol. Clin. Exp. Res. 28, 1449–1458 (2004).
Oroszi, G. & Goldman, D. Alcoholism: genes and mechanisms. Pharmacogenomics 5, 1037–1048 (2004).
Crabbe, J.C. Genetic contributions to addiction. Annu. Rev. Psychol. 53, 435–462 (2002).
Heinz, A., Schafer, M., Higley, J.D., Krystal, J.H. & Goldman, D. Neurobiological correlates of the disposition and maintenance of alcoholism. Pharmacopsychiatry 36 (Suppl.) S255–S258 (2003).
Weiss, F. et al. Compulsive drug-seeking behavior and relapse. Neuroadaptation, stress, and conditioning factors. Ann. NY Acad. Sci. 937, 1–26 (2001).
Ueno, S. et al. Alcohol actions on GABA(A) receptors: from protein structure to mouse behavior. Alcohol. Clin. Exp. Res. 25, 76S–81S (2001).
Carlezon, W.A., Jr. & Nestler, E.J. Elevated levels of GluR1 in the midbrain: a trigger for sensitization to drugs of abuse? Trends Neurosci. 25, 610–615 (2002).
Krystal, J.H., Petrakis, I.L., Mason, G., Trevisan, L. & D'Souza, D.C. N-methyl-D-aspartate glutamate receptors and alcoholism: reward, dependence, treatment, and vulnerability. Pharmacol. Ther. 99, 79–94 (2003).
Nestler, E.J. Molecular mechanisms of drug addiction. Neuropharmacology 47. Suppl 1, 24–32 (2004).
Gonzales, R.A., Job, M.O. & Doyon, W.M. The role of mesolimbic dopamine in the development and maintenance of ethanol reinforcement. Pharmacol. Ther. 103, 121–146 (2004).
Nestler, E.J. Is there a common molecular pathway for addiction? Nat. Neurosci. 8, 1445–1449 (2005).
Cunningham, C.L. & Phillips, T.J. Genetic basis of ethanol reward. in Molecular Biology of Drug Addiction (ed. Maldonado, R.) 263–294 (Humana, Totowa, New Jersey, 2003).
Crabbe, J.C., Metten, P., Cameron, A.J. & Wahlsten, D. An analysis of the genetics of alcohol intoxication in inbred mice. Neurosci. Biobehav. Rev. 28, 785–802 (2005).
Crabbe, J.C. & Morris, R.G.M. Festina lente: Late night thoughts on high-throughput screening of mouse behavior. Nat. Neurosci. 7, 1175–1179 (2004).
Liu, Y. & Lovinger, D.M. Methods in Alcohol-Related Neuroscience Research (CRC, Washington, D.C., 2000).
Dwyer, D.S. & Bradley, R.J. Chemical properties of alcohols and their protein binding sites. Cell. Mol. Life Sci. 57, 265–275 (2000).
Schuckit, M.A. Drug and Alcohol Abuse (Plenum, New York, 1979).
Simantov, R. & Snyder, S.H. The opiate receptor. Biochem. Soc. Trans. 5, 62–65 (1977).
Carpenter-Hyland, E.P., Woodward, J.J. & Chandler, L.J. Chronic ethanol induces synaptic but not extrasynaptic targeting of NMDA receptors. J. Neurosci. 24, 7859–7868 (2004).
Thomas, M.P. & Morrisett, R.A. Dynamics of NMDAR-mediated neurotoxicity during chronic ethanol exposure and withdrawal. Neuropharmacology 39, 218–226 (2000).
Lovinger, D.M. Alcohols and neurotransmitter gated ion channels: past, present and future. Naunyn Schmiedebergs Arch. Pharmacol. 356, 267–282 (1997).
Grobin, A.C., Matthews, D.B., Devaud, L.L. & Morrow, A.L. The role of GABA(A) receptors in the acute and chronic effects of ethanol. Psychopharmacology (Berl.) 139, 2–19 (1998).
Aguayo, L.G., Peoples, R.W., Yeh, H.H. & Yevenes, G.E. GABA(A) receptors as molecular sites of ethanol action. Direct or indirect actions? Curr. Top. Med. Chem. 2, 869–885 (2002).
Mihic, S.J. et al. Sites of alcohol and volatile anaesthetic action on GABA(A) and glycine receptors. Nature 389, 385–389 (1997).
Sundstrom-Poromaa, I. et al. Hormonally regulated alpha(4)beta(2)delta GABA(A) receptors are a target for alcohol. Nat. Neurosci. 5, 721–722 (2002).
Wallner, M., Hanchar, H.J. & Olsen, R.W. Ethanol enhances alpha 4 beta 3 delta and alpha 6 beta 3 delta gamma-aminobutyric acid type A receptors at low concentrations known to affect humans. Proc. Natl. Acad. Sci. USA 100, 15218–15223 (2003).
Hanchar, H.J., Dodson, P.D., Olsen, R.W., Otis, T.S. & Wallner, M. Alcohol-induced motor impairment caused by increased extrasynaptic GABA(A) receptor activity. Nat. Neurosci. 8, 339–345 (2005).
Wei, W., Faria, L.C. & Mody, I. Low ethanol concentrations selectively augment the tonic inhibition mediated by delta subunit-containing GABAA receptors in hippocampal neurons. J. Neurosci. 24, 8379–8382 (2004).
Korpi, E.R., Kleingoor, C., Kettenmann, H. & Seeburg, P.H. Benzodiazepine-induced motor impairment linked to point mutation in cerebellar GABAA receptor. Nature 361, 356–359 (1993).
Congeddu, E. et al. Molecular characterization of new polymorphisms at the β2, α1, γ2 GABA(A) receptor subunit genes associated to a rat nonpreferring ethanol phenotype. Brain Res. Mol. Brain Res. 110, 289–297 (2003).
Radcliffe, R.A. et al. Behavioral characterization of alcohol-tolerant and alcohol-nontolerant rat lines and an F(2) generation. Behav. Genet. 34, 453–463 (2004).
Homanics, G.E. et al. Gene knockout of the alpha6 subunit of the gamma-aminobutyric acid type A receptor: lack of effect on responses to ethanol, pentobarbital, and general anesthetics. Mol. Pharmacol. 51, 588–596 (1997).
Siggins, G.R., Roberto, M. & Nie, Z. The tipsy terminal: presynaptic effects of ethanol. Pharmacol. Ther. 107, 80–98 (2005).
Ariwodola, O.J. & Weiner, J.L. Ethanol potentiation of GABAergic synaptic transmission may be self-limiting: role of presynaptic GABA(B) receptors. J. Neurosci. 24, 10679–10686 (2004).
Carta, M., Mameli, M. & Valenzuela, C.F. Alcohol enhances GABAergic transmission to cerebellar granule cells via an increase in Golgi cell excitability. J. Neurosci. 24, 3746–3751 (2004).
Nie, Z. et al. Ethanol augments GABAergic transmission in the central amygdala via CRF1 receptors. Science 303, 1512–1514 (2004).
Wolf, F.W. & Heberlein, U. Invertebrate models of drug abuse. J. Neurobiol. 54, 161–178 (2003).
Scholz, H., Franz, M. & Heberlein, U. The hangover gene defines a stress pathway required for ethanol tolerance development. Nature 436, 845–847 (2005).
Rodan, A.R., Kiger, J.A., Jr. & Heberlein, U. Functional dissection of neuroanatomical loci regulating ethanol sensitivity in Drosophila. J. Neurosci. 22, 9490–9501 (2002).
Thiele, T.E. et al. High ethanol consumption and low sensitivity to ethanol-induced sedation in protein kinase A-mutant mice. J. Neurosci. 20, RC75, 1–6 (2000).
Yao, L. et al. βγ dimers mediate synergy of dopamine D2 and adenosine A2 receptor-stimulated PKA signaling and regulate ethanol consumption. Cell 109, 733–743 (2002).
Kim, M.S., Repp, A. & Smith, D.P. LUSH odorant-binding protein mediates chemosensory responses to alcohols in Drosophila melanogaster. Genetics 150, 711–721 (1998).
Kruse, S.W., Zhao, R., Smith, D.P. & Jones, D.N. Structure of a specific alcohol-binding site defined by the odorant binding protein LUSH from Drosophila melanogaster. Nat. Struct. Biol. 10, 694–700 (2003).
Zhou, J.J. et al. Revisiting the odorant-binding protein LUSH of Drosophila melanogaster: evidence for odour recognition and discrimination. FEBS Lett. 558, 23–26 (2004).
Davies, A.G. et al. A central role of the BK potassium channel in behavioral responses to ethanol in C. elegans. Cell 115, 655–666 (2003).
Dopico, A.M., Lemos, J.R. & Treistman, S.N. Ethanol increases the activity of large conductance, Ca(2+)-activated K+ channels in isolated neurohypophysial terminals. Mol. Pharmacol. 49, 40–48 (1996).
Corl, A.B., Rodan, A.R. & Heberlein, U. Insulin signaling in the nervous system regulates ethanol intoxication in Drosophila melanogaster. Nat. Neurosci. 8, 18–19 (2005).
Gerlai, R. Zebra fish: an uncharted behavior genetic model. Behav. Genet. 33, 461–468 (2003).
Lockwood, B., Bjerke, S., Kobayashi, K. & Guo, S. Acute effects of alcohol on larval zebrafish: a genetic system for large-scale screening. Pharmacol. Biochem. Behav. 77, 647–654 (2004).
Carvan, M.J., III, Loucks, E., Weber, D.N. & Williams, F.E. Ethanol effects on the developing zebrafish: neurobehavior and skeletal morphogenesis. Neurotoxicol. Teratol. 26, 757–768 (2004).
Browman, K.E., Crabbe, J.C. & Li, T.K. Genetic strategies in preclinical substance abuse research. in Psychopharmacology: The Fourth Generation of Progress (eds. Bloom, F.E. & Kupfer, D.J.) (Lippincott, Williams & Wilkins, Philadelphia, 2000).
Murphy, J.M. et al. Phenotypic and genotypic characterization of the Indiana University rat lines selectively bred for high and low alcohol preference. Behav. Genet. 32, 363–388 (2002).
McBride, W.J. & Li, T.K. Animal models of alcoholism: neurobiology of high alcohol-drinking behavior in rodents. Crit. Rev. Neurobiol. 12, 339–369 (1998).
Crabbe, J.C. & Phillips, T.J. Pharmacogenetic studies of alcohol self-administration and withdrawal. Psychopharmacology (Berl.) 174, 539–560 (2004).
Soyka, M. & Chick, J. Use of acamprosate and opioid antagonists in the treatment of alcohol dependence: a European perspective. Am. J. Addict. 12 Suppl 1, S69–S80 (2003).
Johnson, B.A. & Ait-Daoud, N. Neuropharmacological treatments for alcoholism: scientific basis and clinical findings. Psychopharmacology (Berl.) 149, 327–344 (2000).
Crabbe, J.C., Belknap, J.K. & Buck, K.J. Genetic animal models of alcohol and drug abuse. Science 264, 1715–1723 (1994).
Shirley, R.L., Walter, N.A., Reilly, M.T., Fehr, C. & Buck, K.J. Mpdz is a quantitative trait gene for drug withdrawal seizures. Nat. Neurosci. 7, 699–700 (2004).
Fehr, C., Shirley, R.L., Belknap, J.K., Crabbe, J.C. & Buck, K.J. Congenic mapping of alcohol and pentobarbital withdrawal liability loci to a <1 centimorgan interval of murine chromosome 4: identification of Mpdz as a candidate gene. J. Neurosci. 22, 3730–3738 (2002).
Crabbe, J.C. Identifying genes affecting addiction risk in animal models. in The Cell Biology of Addiction (eds. Madras, B. et al.) (Cold Spring Harbor Press, in press).
Uhl, G.R. Molecular genetics of substance abuse vulnerability: remarkable recent convergence of genome scan results. Ann. NY Acad. Sci. 1025, 1–13 (2004).
Hood, H.M., Belknap, J.K., Crabbe, J.C. & Buck, K.J. Genomewide search for epistasis in a complex trait: Pentobarbital withdrawal convulsions in mice. Behav. Genet. 31, 93–100 (2001).
Bergeson, S.E. et al. Chromosomal loci influencing chronic alcohol withdrawal severity. Mammal. Genome 14, 454–463 (2003).
Jansen, R.C. & Nap, J.P. Genetical genomics: the added value from segregation. Trends Genet. 17, 388–391 (2001).
Hitzemann, R. et al. On the integration of alcohol-related quantitative trait loci and gene expression analyses. Alcohol. Clin. Exp. Res. 28, 1437–1448 (2004).
Tabakoff, B., Bhave, S.V. & Hoffman, P.L. Selective breeding, quantitative trait locus analysis, and gene arrays identify candidate genes for complex drug-related behaviors. J. Neurosci. 23, 4491–4498 (2003).
Chesler, E.J. et al. Complex trait analysis of gene expression uncovers polygenic and pleiotropic networks that modulate nervous system function. Nat. Genet. 37, 233–242 (2005).
Bystrykh, L. et al. Uncovering regulatory pathways that affect hematopoietic stem cell function using 'genetical genomics'. Nat. Genet. 37, 225–232 (2005).
Hubner, N. et al. Integrated transcriptional profiling and linkage analysis for identification of genes underlying disease. Nat. Genet. 37, 243–253 (2005).
Boehm, S.L., II et al. gamma-Aminobutyric acid A receptor subunit mutant mice: new perspectives on alcohol actions. Biochem. Pharmacol. 68, 1581–1602 (2004).
Sullivan, E.V. & Pfefferbaum, A. Neurocircuitry in alcoholism: a substrate of disruption and repair. Psychopharmacology (Berl) 180, 583–594 (2005).
Anderson, K.G., Schweinsburg, A., Paulus, M.P., Brown, S.A. & Tapert, S. Examining personality and alcohol expectancies using functional magnetic resonance imaging (fMRI) with adolescents. J. Stud. Alcohol 66, 323–331 (2005).
Vivian, J.A. et al. Induction and maintenance of ethanol self-administration in cynomolgus monkeys (Macaca fascicularis): long-term characterization of sex and individual differences. Alcohol. Clin. Exp. Res. 25, 1087–1097 (2001).
Middaugh, L.D., Szumlinski, K.K., Van Patten, Y., Marlowe, A.L. & Kalivas, P.W. Chronic ethanol consumption by C57BL/6 mice promotes tolerance to its interoceptive cues and increases extracellular dopamine, an effect blocked by naltrexone. Alcohol. Clin. Exp. Res. 27, 1892–1900 (2003).
Mittleman, G., Van Brunt, C.L. & Matthews, D.B. Schedule-induced ethanol self-administration in DBA/2J and C57BL/6J mice. Alcohol. Clin. Exp. Res. 27, 918–925 (2003).
Falk, J.L. Production of polydipsia in normal rats by an intermittent food schedule. Science 133, 195–196 (1961).
Sharpe, A.L., Tsivkovskaia, N.O. & Ryabinin, A.E. Ataxia and c-Fos expression in mice drinking ethanol in a limited access session. Alcohol. Clin. Exp. Res. 29, 1419–1426 (2005).
Rodd-Henricks, Z.A. et al. Effects of concurrent access to multiple ethanol concentrations and repeated deprivations on alcohol intake of alcohol-preferring rats. Alcohol. Clin. Exp. Res. 25, 1140–1150 (2001).
Rhodes, J.S., Best, K., Belknap, J.K., Finn, D.A. & Crabbe, J.C. Evaluation of a simple model of ethanol drinking to intoxication in C57BL/6J mice. Physiol. Behav. 84, 53–63 (2005).
Finn, D.A. et al. A procedure to produce high alcohol intake in mice. Psychopharmacology (Berl.) 178, 471–480 (2005).
Cronise, K., Finn, D.A., Metten, P. & Crabbe, J.C. Scheduled access to ethanol results in motor impairment and tolerance in female C57BL/6J mice. Pharmacol. Biochem. Behav. 81, 943–953 (2005).
Fidler, T.L., Clews, T.W. & Cunningham, C.L. Reestablishing an intragastric ethanol self-infusion model in rats. Alcohol. Clin. Exp. Res. (in press).
Lopez, M.F. & Becker, H.C. Effect of pattern and number of chronic ethanol exposures on subsequent voluntary ethanol intake in C57BL/6J mice. Psychopharmacology (Berl.) published online 7 July 2005 (10.1007/s00213-005-0026-3).
Kliethermes, C.L. Anxiety-like behaviors following chronic ethanol exposure. Neurosci. Biobehav. Rev. 28, 837–850 (2005).
Anguelova, M., Benkelfat, C. & Turecki, G. A systematic review of association studies investigating genes coding for serotonin receptors and the serotonin transporter: I. Affective disorders. Mol. Psychiatry 8, 574–591 (2003).
Barr, C.S. et al. Interaction between serotonin transporter gene variation and rearing condition in alcohol preference and consumption in female primates. Arch. Gen. Psychiatry 61, 1146–1152 (2004).
Caspi, A. et al. Influence of life stress on depression: moderation by a polymorphism in the 5-HTT gene. Science 301, 386–389 (2003).
Barr, C.S. et al. Sexual dichotomy of an interaction between early adversity and the serotonin transporter gene promoter variant in rhesus macaques. Proc. Natl. Acad. Sci. USA 101, 12358–12363 (2004).
Hu, X. et al. An expanded evaluation of the relationship of four alleles to the level of response to alcohol and the alcoholism risk. Alcohol. Clin. Exp. Res. 29, 8–16 (2005).
Ozaki, N. et al. Serotonin transporter missense mutation associated with a complex neuropsychiatric phenotype. Mol. Psychiat. 8, 895, 933–936 (2003).
Oswald, L.M. & Wand, G.S. Opioids and alcoholism. Physiol. Behav. 81, 339–358 (2004).
Egli, M. Can experimental paradigms and animal models be used to discover clinically effective medications for alcoholism? Addict. Biol. (in press).
Lovinger, D.M., White, G. & Weight, F.F. Ethanol inhibits NMDA-activated ion current in hippocampal neurons. Science 243, 1721–1724 (1989).
Nagy, J. Renaissance of NMDA receptor antagonists: do they have a role in the pharmacotherapy for alcoholism? IDrugs 7, 339–350 (2004).
Backstrom, P., Bachteler, D., Koch, S., Hyytia, P. & Spanagel, R. mGluR5 antagonist MPEP reduces ethanol-seeking and relapse behavior. Neuropsychopharmacology 29, 921–928 (2004).
Hungund, B.L. & Basavarajappa, B.S. Role of endocannabinoids and cannabinoid CB1 receptors in alcohol-related behaviors. Ann. NY Acad. Sci. 1025, 515–527 (2004).
Di, M.V. & Matias, I. Endocannabinoid control of food intake and energy balance. Nat. Neurosci. 8, 585–589 (2005).
Le Foll, B. & Goldberg, S.R. Cannabinoid CB1 receptor antagonists as promising new medications for drug dependence. J. Pharmacol. Exp. Ther. 312, 875–883 (2005).
Van Gaal, L.F., Rissanen, A.M., Scheen, A.J., Ziegler, O. & Rossner, S. Effects of the cannabinoid-1 receptor blocker rimonabant on weight reduction and cardiovascular risk factors in overweight patients: 1-year experience from the RIO-Europe study. Lancet 365, 1389–1397 (2005).
Acknowledgements
The authors are supported by the US Department of Veterans Affairs (J.C.C.), and the US National Institute on Alcohol Abuse and Alcoholism (AA10760, AA12714 and AA13519 to J.C.C) and the Division of Intramural Clinical and Basic Research (D.M.L.). We thank M. Rutledge-Gorman for help in preparing the manuscript, and G. McClearn for many previous versions of Figure 2.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Lovinger, D., Crabbe, J. Laboratory models of alcoholism: treatment target identification and insight into mechanisms. Nat Neurosci 8, 1471–1480 (2005). https://doi.org/10.1038/nn1581
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn1581
This article is cited by
-
Transcriptome analysis of alcohol-treated microglia reveals downregulation of beta amyloid phagocytosis
Journal of Neuroinflammation (2018)
-
Small molecule modulators of σ2R/Tmem97 reduce alcohol withdrawal-induced behaviors
Neuropsychopharmacology (2018)
-
Paradoxical antidepressant effects of alcohol are related to acid sphingomyelinase and its control of sphingolipid homeostasis
Acta Neuropathologica (2017)
-
Ketamine and MAG Lipase Inhibitor-Dependent Reversal of Evolving Depressive-Like Behavior During Forced Abstinence From Alcohol Drinking
Neuropsychopharmacology (2016)
-
Forced swim stress increases ethanol consumption in C57BL/6J mice with a history of chronic intermittent ethanol exposure
Psychopharmacology (2016)