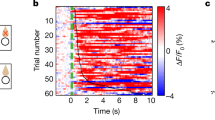
Abstract
In the face of starvation, animals will engage in high-risk behaviors that would normally be considered maladaptive. Starving rodents, for example, will forage in areas that are more susceptible to predators and will also modulate aggressive behavior within a territory of limited or depleted nutrients. The neural basis of these adaptive behaviors likely involves circuits that link innate feeding, aggression and fear. Hypothalamic agouti-related peptide (AgRP)-expressing neurons are critically important for driving feeding and project axons to brain regions implicated in aggression and fear. Using circuit-mapping techniques in mice, we define a disynaptic network originating from a subset of AgRP neurons that project to the medial nucleus of the amygdala and then to the principal bed nucleus of the stria terminalis, which suppresses territorial aggression and reduces contextual fear. We propose that AgRP neurons serve as a master switch capable of coordinating behavioral decisions relative to internal state and environmental cues.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Sih, A. Optimal behavior: can foragers balance two conflicting demands? Science 210, 1041–1043 (1980).
Krebs, J.R. Optimal foraging, predation risk and territory defense. Ardea 68, 83–90 (1980).
Brown, J.L. The evolution of diversity in avian territorial systems. Wilson Bull. 76, 160–169, http://www.jstor.org/stable/4159278 (1964).
Anderson, P.K. Foraging range in mice and voles – the role of risk. Can. J. Zool. 64, 2645–2653 (1986).
Magnhagen, C. Predation risk and foraging in juvenile pink (Oncorhynchus gorbuscha) and chum salmon (O. keta). Can. J. Fish. Aquat. Sci. 45, 592–596 (1988).
Kohler, S.L. & Mcpeek, M.A. Predation risk and the foraging behavior of competing stream insects. Ecology 70, 1811–1825 (1989).
Whitham, J. & Mathis, A. Effects of hunger and predation risk on foraging behavior of graybelly salamanders, Eurycea multiplicata. J. Chem. Ecol. 26, 1659–1665 (2000).
Hahn, T.M., Breininger, J.F., Baskin, D.G. & Schwartz, M.W. Coexpression of Agrp and NPY in fasting-activated hypothalamic neurons. Nat. Neurosci. 1, 271–272 (1998).
Krashes, M.J., Shah, B.P., Koda, S. & Lowell, B.B. Rapid versus delayed stimulation of feeding by the endogenously released AgRP neuron mediators GABA, NPY, and AgRP. Cell Metab. 18, 588–595 (2013).
Luquet, S., Perez, F.A., Hnasko, T.S. & Palmiter, R.D. NPY/AgRP neurons are essential for feeding in adult mice but can be ablated in neonates. Science 310, 683–685 (2005).
Krashes, M.J. et al. Rapid, reversible activation of AgRP neurons drives feeding behavior in mice. J. Clin. Invest. 121, 1424–1428 (2011).
Aponte, Y., Atasoy, D. & Sternson, S.M. AGRP neurons are sufficient to orchestrate feeding behavior rapidly and without training. Nat. Neurosci. 14, 351–355 (2011).
Horvath, T.L., Bechmann, I., Naftolin, F., Kalra, S.P. & Leranth, C. Heterogeneity in the neuropeptide Y-containing neurons of the rat arcuate nucleus: GABAergic and non-GABAergic subpopulations. Brain Res. 756, 283–286 (1997).
Broberger, C., Johansen, J., Johansson, C., Schalling, M. & Hökfelt, T. The neuropeptide Y/agouti gene-related protein (AGRP) brain circuitry in normal, anorectic, and monosodium glutamate-treated mice. Proc. Natl. Acad. Sci. USA 95, 15043–15048 (1998).
Cowley, M.A. et al. Leptin activates anorexigenic POMC neurons through a neural network in the arcuate nucleus. Nature 411, 480–484 (2001).
Padilla, S.L., Carmody, J.S. & Zeltser, L.M. Pomc-expressing progenitors give rise to antagonistic neuronal populations in hypothalamic feeding circuits. Nat. Med. 16, 403–405 (2010).
Betley, J.N., Cao, Z.F., Ritola, K.D. & Sternson, S.M. Parallel, redundant circuit organization for homeostatic control of feeding behavior. Cell 155, 1337–1350 (2013).
Liu, H. et al. Transgenic mice expressing green fluorescent protein under the control of the melanocortin-4 receptor promoter. J. Neurosci. 23, 7143–7154 (2003).
Kishi, T. et al. Expression of melanocortin 4 receptor mRNA in the central nervous system of the rat. J. Comp. Neurol. 457, 213–235 (2003).
Stowers, L., Cameron, P. & Keller, J.A. Ominous odors: olfactory control of instinctive fear and aggression in mice. Curr. Opin. Neurobiol. 23, 339–345 (2013).
Pereno, G.L., Balaszczuk, V. & Beltramino, C.A. Detection of conspecific pheromones elicits fos expression in GABA and calcium-binding cells of the rat vomeronasal system-medial extended amygdala. J. Physiol. Biochem. 67, 71–85 (2011).
Lin, D. et al. Functional identification of an aggression locus in the mouse hypothalamus. Nature 470, 221–226 (2011).
Hong, W., Kim, D.W. & Anderson, D.J. Antagonistic control of social versus repetitive self-grooming behaviors by separable amygdala neuronal subsets. Cell 158, 1348–1361 (2014).
Armbruster, B.N., Li, X., Pausch, M.H., Herlitze, S. & Roth, B.L. Evolving the lock to fit the key to create a family of G protein-coupled receptors potently activated by an inert ligand. Proc. Natl. Acad. Sci. USA 104, 5163–5168 (2007).
Dietrich, M.O., Zimmer, M.R., Bober, J. & Horvath, T.L. Hypothalamic Agrp neurons drive stereotypic behaviors beyond feeding. Cell 160, 1222–1232 (2015).
Laundré, J.W. Ecology. How large predators manage the cost of hunting. Science 346, 33–34 (2014).
Wang, L. & Anderson, D.J. Identification of an aggression-promoting pheromone and its receptor neurons in Drosophila. Nature 463, 227–231 (2010).
Boyden, E.S., Zhang, F., Bamberg, E., Nagel, G. & Deisseroth, K. Millisecond-timescale, genetically targeted optical control of neural activity. Nat. Neurosci. 8, 1263–1268 (2005).
Atasoy, D., Betley, J.N., Su, H.H. & Sternson, S.M. Deconstruction of a neural circuit for hunger. Nature 488, 172–177 (2012).
Liu, J., Garza, J.C., Li, W. & Lu, X.Y. Melanocortin-4 receptor in the medial amygdala regulates emotional stress-induced anxiety-like behaviour, anorexia and corticosterone secretion. Int. J. Neuropsychopharmacol. 16, 105–120 (2013).
King, B.M., Cook, J.T., Rossiter, K.N. & Rollins, B.L. Obesity-inducing amygdala lesions: examination of anterograde degeneration and retrograde transport. Am. J. Physiol. Regul. Integr. Comp. Physiol. 284, R965–R982 (2003).
Xu, P. et al. Estrogen receptor-α in medial amygdala neurons regulates body weight. J. Clin. Invest. 125, 2861–2876 (2015).
Lin, S., Boey, D. & Herzog, H. NPY and Y receptors: lessons from transgenic and knockout models. Neuropeptides 38, 189–200 (2004).
Sanz, E. et al. Cell-type-specific isolation of ribosome-associated mRNA from complex tissues. Proc. Natl. Acad. Sci. USA 106, 13939–13944 (2009).
Heimer-McGinn, V., Murphy, A.C., Kim, J.C., Dymecki, S.M. & Young, P.W. Decreased dendritic spine density as a consequence of tetanus toxin light chain expression in single neurons in vivo. Neurosci. Lett. 555, 36–41 (2013).
Han, S., Soleiman, M.T., Soden, M.E., Zweifel, L.S. & Palmiter, R.D. Elucidating an affective pain circuit that creates a threat memory. Cell 162, 363–374 (2015).
Lo, L. & Anderson, D.J. A Cre-dependent, anterograde transsynaptic viral tracer for mapping output pathways of genetically marked neurons. Neuron 72, 938–950 (2011).
Swanson, L.W. Cerebral hemisphere regulation of motivated behavior. Brain Res. 886, 113–164 (2000).
Canteras, N.S., Simerly, R.B. & Swanson, L.W. Organization of projections from the medial nucleus of the amygdala: a PHAL study in the rat. J. Comp. Neurol. 360, 213–245 (1995).
Niimi, K. et al. Heterogeneous electrophysiological and morphological properties of neurons in the mouse medial amygdala in vitro. Brain Res. 1480, 41–52 (2012).
Bian, X. Physiological and morphological characterization of GABAergic neurons in the medial amygdala. Brain Res. 1509, 8–19 (2013).
Keshavarzi, S., Sullivan, R.K., Ianno, D.J. & Sah, P. Functional properties and projections of neurons in the medial amygdala. J. Neurosci. 34, 8699–8715 (2014).
Cruikshank, S.J., Urabe, H., Nurmikko, A.V. & Connors, B.W. Pathway-specific feedforward circuits between thalamus and neocortex revealed by selective optical stimulation of axons. Neuron 65, 230–245 (2010).
Sun, Q.Q., Akk, G., Huguenard, J.R. & Prince, D.A. Differential regulation of GABA release and neuronal excitability mediated by neuropeptide Y1 and Y2 receptors in rat thalamic neurons. J. Physiol. (Lond.) 531, 81–94 (2001).
Sun, Q.Q., Huguenard, J.R. & Prince, D.A. Neuropeptide Y receptors differentially modulate G-protein-activated inwardly rectifying K+ channels and high-voltage-activated Ca2+ channels in rat thalamic neurons. J. Physiol. (Lond.) 531, 67–79 (2001).
Bosch, M.A., Tonsfeldt, K.J. & Rønnekleiv, O.K. mRNA expression of ion channels in GnRH neurons: subtype-specific regulation by 17β-estradiol. Mol. Cell. Endocrinol. 367, 85–97 (2013).
Behbehani, M.M. Functional characteristics of the midbrain periaqueductal gray. Prog. Neurobiol. 46, 575–605 (1995).
Kennedy, A. et al. Internal states and behavioral decision-making: toward an integration of emotion and cognition. Cold Spring Harb. Symp. Quant. Biol. 79, 199–210 (2014).
Chen, Y., Lin, Y.C., Kuo, T.W. & Knight, Z.A. Sensory detection of food rapidly modulates arcuate feeding circuits. Cell 160, 829–841 (2015).
Betley, J.N. et al. Neurons for hunger and thirst transmit a negative-valence teaching signal. Nature 521, 180–185 (2015).
Sanz, E. et al. Fertility-regulating Kiss1 neurons arise from hypothalamic POMC-expressing progenitors. J. Neurosci. 35, 5549–5556 (2015).
Gore, B.B., Soden, M.E. & Zweifel, L.S. Manipulating gene expression in projection-specific neuronal populations using combinatorial viral approaches. Curr. Protoc. Neurosci. 65, 4.35 (2013).10.1002/0471142301.ns0435s65
Qiu, J. et al. Rapid signaling of estrogen in hypothalamic neurons involves a novel G-protein-coupled estrogen receptor that activates protein kinase C. J. Neurosci. 23, 9529–9540 (2003).
Sparta, D.R. et al. Construction of implantable optical fibers for long-term optogenetic manipulation of neural circuits. Nat. Protoc. 7, 12–23 (2012).
Acknowledgements
We thank K. Kafer and M. Chiang for technical assistance generating the new line of mice and maintaining the mouse colonies. We thank M.A. Patterson and B.C. Jarvie for careful reading of this paper and the entire Palmiter laboratory for discussions and critiques. We thank D.J. Anderson and L. Lo (Caltech) for generously providing H129Δ-fs-TK-TT. We thank E. Strakbein at U.W. Scientific Instruments for the development of tools and apparatus used in this manuscript. This work was supported by funds from the Hilda Preston Davis Foundation (S.L.P.), the US National Institutes of Health (R01DK068098, R01NS038809, M.J.K. and O.K.R.; R01MH094536, L.S.Z.; R01DA024908, R.D.P.), a Marie Sklodowska-Curie award (H2020-MSCA-IF-2014-658352, E.S.), a Ramón y Cajal fellowship (RyC-2012-11873, A.Q.), European Research Council Starting Grant NEUROMITO (ERC-2014-StG-638106, A.Q.) and MINECO Proyectos I+D de Excelencia (SAF2014-57981P, A.Q.).
Author information
Authors and Affiliations
Contributions
S.L.P. designed the study under the guidance of R.D.P.; J.Q. gathered and analyzed electrophysiological data in the laboratories of M.J.K. and O.K.R.; M.E.S. gathered and analyzed electrophysiology data in the laboratory of L.S.Z.; E.S. performed the RiboTag pulldown and quantitative PCR in the laboratory of A.Q.; C.C.N. performed single-cell PCR on harvested cells after whole-cell recordings; R.D.P. generated the Agrpcre and Npy1rcre lines of mice; F.D.B. assisted with behavior experiments and blind scoring; R.D.P., M.J.K., O.K.R. and L.S.Z. provided laboratory space and resources to conduct the experiments; S.L.P. and R.D.P. wrote the manuscript with revisions and input from all contributing authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 AgRP-expressing cells are active in the state of starvation and act to prioritize behavior and physiology to promote food seeking and conserve energy.
(a) Model of AgRP-directed behaviors and physiology. (b) AgRP neurons that project to the Npy1r-expressing cells in the MeA are likely involved in decreasing territorial behaviors to facilitate food seeking in the state of starvation. (c) AgRP neurons that project to the PVH influence feeding but do not influence territorial behavior.
Supplementary Figure 2 Profile of fasted residents.
(a) Fasted animals learn to avoid the shock-associated chamber when the food-challenge assay is performed in the absence of food or food-associated cues (ad lib n = 6; 48-hr Fast n = 6). (b) Territorialized resident animals are significantly heavier than intruders. (c) 24-hr fasted residents display decreased territorial aggression (n = 6). (d) Time distribution of nose-nose interactions and escape behaviors (including rearing and jumping), comparing the ad libitum and fasted state of an individual mouse. (e,f) Average scores: nose-to-nose (e) and escape behavior (f) (n = 9). Error bars represent the mean ± SEM.
Supplementary Figure 3 In the presence of food, AgRP-stimulated animals display little aggression toward an intruding conspecific.
(a) Home-cage aggression of AgRP stimulated animals in the presence of food (n = 7) was significantly less than non-stimulated controls (sal, n = 10; Fig. 1e). (b) Food was presented following 30 sec of exposure to an intruder. The blue bars represent the time that each animal spent engaged with the food during the trial. Stimulated animals spend 47.2 ± 3.4% of the trail engaged with the food and consumed 0.34 ± 0.3 g of food during the 10-min trial.
Supplementary Figure 4 Fluorescent bead targeting and fiber placement. Validation of stereotaxic MeA targeting.
(a) Fluorescent image (left) compared to a matching coronal atlas (right). Scale bar, 200 μm. Fiber placement characterization in the MeA (b) and PVH (c). (b,c) Dapi stain (left), blue circles represent the diameter and ventral position of the fiber relative to the targeted bregma position (middle). Scale bar, 200 μm. Insert (right) is a composite of all fiber tracks identified in the targeted region. Colored circle sizes represent the thickness of the fiber track at the targeted bregma positions: MeA (−1.5 mm), PVH (−0.8 mm)
Supplementary Figure 5 Npy1R is expressed throughout the MeA.
Percent distribution of cells in the rostral, mid and caudal MeA
Supplementary Figure 6 Silencing of Npy1RMeA neurons does not alter the innate response to a threatening environment.
Npy1rCre-expressing cells in the MeA were bilaterally injected with either AAV1-DIO-GFP:TetTox or AAV1-DIO-YFP. Npy1RMeA silencing did not significantly change anxiety assessed by time spent in the exposed arms of an elevated maze. Comparing TetTox, n = 4 to YFP controls, n = 4. Error bars represent the mean ± SEM.
Supplementary Figure 7 Few if any cells in the ARH project to the pBNST; also targeting for experiment Figure 4d.
(a) Retrobead targeting of the pBNST. An image of the fluorescent injection site (left) is paired with a matching coronal atlas image (right). Scale bar, 200 μm. (b) RetroBeads retained in the ARH, scale bar, 100 μm. (c) tdT expression following injection of H129D-fs-TK-TT into the ARH of an AgrpCre animal; scale bar 200 μm. (d) Green RetroBeads injected into the pBNST; scale bar 200 μm. (e) Distribution of aggressive behaviors in: ad libitum mice (left), Npy1RMEA::hM3Dq + CNO (center), and Npy1RMEA::ChR2 with light stimulation of fibers in the pBNST (right).
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 (PDF 2376 kb)
Npy1RMeA activation enhances home-cage aggressive behaviour
Attack behavior demonstrated by CNO-induced activation of Npy1RMeA neurons that express hM3Dq. CNO was delivered by intraperitoneal injection 30 min prior to the test. The intruder test was performed during the dark cycle and recorded with an infrared light. (AVI 14125 kb)
Npy1RMeA activation can evoke violent aggression
Violent attack of an anesthetized intruder demonstrated by CNO-induced activation of Npy1RMeA neurons that express hM3Dq. CNO was delivered by intraperitoneal injection 30 min prior to the test. The intruder test was performed during the dark cycle and recorded with an infrared light. (AVI 17099 kb)
Npy1RMeA → pBNST circuit activation enhances territorial aggression
Nudging behavior demonstrated by light-induced activation of Npy1RMeA neurons that express ChR2 and project to the pBNST. Light was delivered at 10 Hz with 5-ms pulses for 5 s followed by 2 s light-off recovery. The intruder test was performed during the dark cycle and recorded with an infrared light. (AVI 16943 kb)
Rights and permissions
About this article
Cite this article
Padilla, S., Qiu, J., Soden, M. et al. Agouti-related peptide neural circuits mediate adaptive behaviors in the starved state. Nat Neurosci 19, 734–741 (2016). https://doi.org/10.1038/nn.4274
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.4274
This article is cited by
-
Basolateral amygdala neuropeptide Y system modulates binge ethanol consumption
Neuropsychopharmacology (2024)
-
Metabolic shift toward ketosis in asocial cavefish increases social-like affinity
BMC Biology (2023)
-
Activation of neurons in the insular cortex and lateral hypothalamus during food anticipatory period caused by food restriction in mice
The Journal of Physiological Sciences (2023)
-
Modulation of foraging-like behaviors by cholesterol-FGF19 axis
Cell & Bioscience (2023)
-
Defensive responses: behaviour, the brain and the body
Nature Reviews Neuroscience (2023)