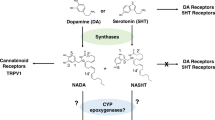
Abstract
Augmentation of endogenous cannabinoid (eCB) signaling represents an emerging approach to the treatment of affective disorders. Cyclooxygenase-2 (COX-2) oxygenates arachidonic acid to form prostaglandins, but also inactivates eCBs in vitro. However, the viability of COX-2 as a therapeutic target for in vivo eCB augmentation has not been explored. Using medicinal chemistry and in vivo analytical and behavioral pharmacological approaches, we found that COX-2 is important for the regulation of eCB levels in vivo. We used a pharmacological strategy involving substrate-selective inhibition of COX-2 to augment eCB signaling without affecting related non-eCB lipids or prostaglandin synthesis. Behaviorally, substrate-selective inhibition of COX-2 reduced anxiety-like behaviors in mice via increased eCB signaling. Our data suggest a key role for COX-2 in the regulation of eCB signaling and indicate that substrate-selective pharmacology represents a viable approach for eCB augmentation with broad therapeutic potential.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Change history
11 August 2013
In the version of this article initially published online, red and blue were switched in the Figure 1h key. The error has been corrected for the print, PDF and HTML versions of this article.
References
Di Marzo, V. Endocannabinoids: synthesis and degradation. Rev. Physiol. Biochem. Pharmacol. 160, 1–24 (2008).
Piomelli, D. The molecular logic of endocannabinoid signaling. Nat. Rev. Neurosci. 4, 873–884 (2003).
Ahn, K., McKinney, M.K. & Cravatt, B.F. Enzymatic pathways that regulate endocannabinoid signaling in the nervous system. Chem. Rev. 108, 1687–1707 (2008).
Kunos, G. Understanding metabolic homeostasis and imbalance: what is the role of the endocannabinoid system? Am. J. Med. 120, S18–S24 (2007).
Di Marzo, V., Piscitelli, F. & Mechoulam, R. Cannabinoids and endocannabinoids in metabolic disorders with focus on diabetes. Handb. Exp. Pharmacol. 203, 75–104 (2011).
Guindon, J. & Hohmann, A.G. The endocannabinoid system and pain. CNS Neurol. Disord. Drug Targets 8, 403–421 (2009).
Hohmann, A.G. & Suplita, R.L. II. Endocannabinoid mechanisms of pain modulation. AAPS J. 8, E693–E708 (2006).
Lichtman, A.H., Varvel, S.A. & Martin, B.R. Endocannabinoids in cognition and dependence. Prostaglandins Leukot. Essent. Fatty Acids 66, 269–285 (2002).
Patel, S. & Hillard, C.J. Adaptations in endocannabinoid signaling in response to repeated homotypic stress: a novel mechanism for stress habituation. Eur. J. Neurosci. 27, 2821–2829 (2008).
Lutz, B. Endocannabinoid signals in the control of emotion. Curr. Opin. Pharmacol. 9, 46–52 (2009).
Centonze, D., Finazzi-Agro, A., Bernardi, G. & Maccarrone, M. The endocannabinoid system in targeting inflammatory neurodegenerative diseases. Trends Pharmacol. Sci. 28, 180–187 (2007).
Bisogno, T. & Di Marzo, V. Cannabinoid receptors and endocannabinoids: role in neuroinflammatory and neurodegenerative disorders. CNS Neurol. Disord. Drug Targets 9, 564–573 (2010).
Bifulco, M., Laezza, C., Gazzerro, P. & Pentimalli, F. Endocannabinoids as emerging suppressors of angiogenesis and tumor invasion. Oncol. Rep. (review) 17, 813–816 (2007).
López-Rodriguez, M.L., Viso, A., Ortega-Gutierrez, S. & Diaz-Laviada, I. Involvement of cannabinoids in cellular proliferation. Mini Rev. Med. Chem. 5, 97–106 (2005).
Tam, J. et al. Involvement of neuronal cannabinoid receptor CB1 in regulation of bone mass and bone remodeling. Mol. Pharmacol. 70, 786–792 (2006).
Cravatt, B.F. et al. Molecular characterization of an enzyme that degrades neuromodulatory fatty-acid amides. Nature 384, 83–87 (1996).
Long, J.Z. et al. Selective blockade of 2-arachidonoylglycerol hydrolysis produces cannabinoid behavioral effects. Nat. Chem. Biol. 5, 37–44 (2009).
Mangieri, R.A. & Piomelli, D. Enhancement of endocannabinoid signaling and the pharmacotherapy of depression. Pharmacol. Res. 56, 360–366 (2007).
Sciolino, N.R., Zhou, W. & Hohmann, A.G. Enhancement of endocannabinoid signaling with JZL184, an inhibitor of the 2-arachidonoylglycerol hydrolyzing enzyme monoacylglycerol lipase, produces anxiolytic effects under conditions of high environmental aversiveness in rats. Pharmacol. Res. 64, 226–234 (2011).
Sumislawski, J.J., Ramikie, T.S. & Patel, S. Reversible gating of endocannabinoid plasticity in the amygdala by chronic stress: a potential role for monoacylglycerol lipase inhibition in the prevention of stress-induced behavioral adaptation. Neuropsychopharmacology 36, 2750–2761 (2011).
Naidu, P.S. et al. Evaluation of fatty acid amide hydrolase inhibition in murine models of emotionality. Psychopharmacology (Berl.) 192, 61–70 (2007).
Kathuria, S. et al. Modulation of anxiety through blockade of anandamide hydrolysis. Nat. Med. 9, 76–81 (2003).
Gaetani, S. et al. The endocannabinoid system as a target for novel anxiolytic and antidepressant drugs. Int. Rev. Neurobiol. 85, 57–72 (2009).
Makriyannis, A., Mechoulam, R. & Piomelli, D. Therapeutic opportunities through modulation of the endocannabinoid system. Neuropharmacology 48, 1068–1071 (2005).
Di Marzo, V. The endocannabinoid system: its general strategy of action, tools for its pharmacological manipulation and potential therapeutic exploitation. Pharmacol Res. 60, 77–84 (2009).
Piomelli, D. The endocannabinoid system: a drug discovery perspective. Curr. Opin. Investig. Drugs 6, 672–679 (2005).
Kozak, K.R., Rowlinson, S.W. & Marnett, L.J. Oxygenation of the endocannabinoid, 2-arachidonylglycerol, to glyceryl prostaglandins by cyclooxygenase-2. J. Biol. Chem. 275, 33744–33749 (2000).
Rouzer, C.A. & Marnett, L.J. Non-redundant functions of cyclooxygenases: oxygenation of endocannabinoids. J. Biol. Chem. 283, 8065–8069 (2008).
Kim, J. & Alger, B.E. Inhibition of cyclooxygenase-2 potentiates retrograde endocannabinoid effects in hippocampus. Nat. Neurosci. 7, 697–698 (2004).
Glaser, S.T. & Kaczocha, M. Cyclooxygenase-2 mediates anandamide metabolism in the mouse brain. J. Pharmacol. Exp. Ther. 335, 380–388 (2010).
Staniaszek, L.E., Norris, L.M., Kendall, D.A., Barrett, D.A. & Chapman, V. Effects of COX-2 inhibition on spinal nociception: the role of endocannabinoids. Br. J. Pharmacol. 160, 669–676 (2010).
Bishay, P. et al. R-flurbiprofen reduces neuropathic pain in rodents by restoring endogenous cannabinoids. PLoS ONE 5, e10628 (2010).
Duggan, K.C. et al. (R)-Profens are substrate-selective inhibitors of endocannabinoid oxygenation by COX-2. Nat. Chem. Biol. 7, 803–809 (2011).
Prusakiewicz, J.J., Duggan, K.C., Rouzer, C.A. & Marnett, L.J. Differential sensitivity and mechanism of inhibition of COX-2 oxygenation of arachidonic acid and 2-arachidonoylglycerol by ibuprofen and mefenamic acid. Biochemistry 48, 7353–7355 (2009).
Kalgutkar, A.S. et al. Biochemically based design of cyclooxygenase-2 (COX-2) inhibitors: facile conversion of nonsteroidal antiinflammatory drugs to potent and highly selective COX-2 inhibitors. Proc. Natl. Acad. Sci. USA 97, 925–930 (2000).
Long, J.Z. et al. Dual blockade of FAAH and MAGL identifies behavioral processes regulated by endocannabinoid crosstalk in vivo. Proc. Natl. Acad. Sci. USA 106, 20270–20275 (2009).
Kinsey, S.G., O'Neal, S.T., Long, J.Z., Cravatt, B.F. & Lichtman, A.H. Inhibition of endocannabinoid catabolic enzymes elicits anxiolytic-like effects in the marble burying assay. Pharmacol. Biochem. Behav. 98, 21–27 (2011).
Rossi, S. et al. Preservation of striatal cannabinoid CB1 receptor function correlates with the antianxiety effects of fatty acid amide hydrolase inhibition. Mol. Pharmacol. 78, 260–268 (2010).
Moreira, F.A., Kaiser, N., Monory, K. & Lutz, B. Reduced anxiety-like behavior induced by genetic and pharmacological inhibition of the endocannabinoid-degrading enzyme fatty acid amide hydrolase (FAAH) is mediated by CB1 receptors. Neuropharmacology 54, 141–150 (2008).
Fride, E., Perchuk, A., Hall, F.S., Uhl, G.R. & Onaivi, E.S. Behavioral methods in cannabinoid research. Methods Mol. Med. 123, 269–290 (2006).
O'Sullivan, S.E. Cannabinoids go nuclear: evidence for activation of peroxisome proliferator-activated receptors. Br. J. Pharmacol. 152, 576–582 (2007).
Cristino, L. et al. Immunohistochemical localization of anabolic and catabolic enzymes for anandamide and other putative endovanilloids in the hippocampus and cerebellar cortex of the mouse brain. Neuroscience 151, 955–968 (2008).
Patel, S. & Hillard, C.J. Pharmacological evaluation of cannabinoid receptor ligands in a mouse model of anxiety: further evidence for an anxiolytic role for endogenous cannabinoid signaling. J. Pharmacol. Exp. Ther. 318, 304–311 (2006).
Müller, N. & Schwarz, M.J. COX-2 inhibition in schizophrenia and major depression. Curr. Pharm. Des. 14, 1452–1465 (2008).
Patrignani, P., Tacconelli, S., Bruno, A., Sostres, C. & Lanas, A. Managing the adverse effects of nonsteroidal anti-inflammatory drugs. Expert Rev. Clin. Pharmacol. 4, 605–621 (2011).
Yu, Y. et al. Vascular COX-2 modulates blood pressure and thrombosis in mice. Sci. Transl. Med. 4, 132ra154 (2012).
Kalgutkar, A.S., Marnett, A.B., Crews, B.C., Remmel, R.P. & Marnett, L.J. Ester and amide derivatives of the nonsteroidal antiinflammatory drug, indomethacin, as selective cyclooxygenase-2 inhibitors. J. Med. Chem. 43, 2860–2870 (2000).
Rowlinson, S.W., Crews, B.C., Lanzo, C.A. & Marnett, L.J. The binding of arachidonic acid in the cyclooxygenase active site of mouse prostaglandin endoperoxide synthase-2 (COX-2) - A putative L-shaped binding conformation utilizing the top channel region. J. Biol. Chem. 274, 23305–23310 (1999).
Rouzer, C.A. & Marnett, L.J. Glyceryl prostaglandin synthesis in lipopolysaccharide-treated RAW264.7 cells is augmented by interferon-gamma and granulocytemacrophage colony stimulating factor. Prostag. Oth. Lipid M. 79, 146–147 (2006).
Blankman, J.L., Simon, G.M. & Cravatt, B.F. A comprehensive profile of brain enzymes that hydrolyze the endocannabinoid 2-arachidonoylglycerol. Chem. Biol. 14, 1347–1356 (2007).
Ahn, K. et al. Discovery and characterization of a highly selective FAAH inhibitor that reduces inflammatory pain. Chem. Biol. 16, 411–420 (2009).
Pedicord, D.L. et al. Molecular characterization and identification of surrogate substrates for diacylglycerol lipase alpha. Biochem. Biophys. Res. Commun. 411, 809–814 (2011).
Uddin, M.J. et al. Fluorinated COX-2 inhibitors as agents in PET imaging of inflammation and cancer. Cancer Prev. Res. (Phila.) 4, 1536–1545 (2011).
Pan, B., Hillard, C.J. & Liu, Q.S. D-2 dopamine receptor activation facilitates endocannabinoid-mediated long-term synaptic depression of GABAergic synaptic transmission in midbrain dopamine neurons via cAMP-protein kinase A signaling. J. Neurosci. 28, 14018–14030 (2008).
Patel, S., Kingsley, P.J., Mackie, K., Marnett, L.J. & Winder, D.G. Repeated homotypic stress elevates 2-arachidonoylglycerol levels and enhances short-term endocannabinoid signaling at inhibitory synapses in basolateral amygdala. Neuropsychopharmacology 34, 2699–2709 (2009).
Kingsley, P.J. & Marnett, L.J. LC-MS-MS analysis of neutral eicosanoids. Methods Enzymol. 433, 91–112 (2007).
Acknowledgements
We thank K. Masuda, M. Brown, R. Stevens and B. Cravatt (Scripps Research Institute) for FAAH knockout mice. This work was supported by US National Institutes of Health grants MH090412 and MH100096 (S.P.), CA89450, GM15431, NS064278 (L.J.M.), DA031572 (D.J.H.), HL96967, HL109199 (J.R.), NS078291 (R.J.C.) and T32-MH065215 (B.C.S.), and the A.B. Hancock Jr. Memorial Laboratory for Cancer Research. Analytical studies were conducted in the Vanderbilt University Mass Spectrometry Core facility and all behavioral experiments were conducted in the Vanderbilt University Medical Center Mouse Neurobehavioral Core facility. The content of this article is solely the responsibility of the authors and does not necessarily represent the official views of the US National Institutes of Health.
Author information
Authors and Affiliations
Contributions
LM-4131 was synthesized and characterized in vitro by D.J.H. in the laboratory of L.J.M. In vivo biochemical and behavioral experiments were designed, executed and analyzed by D.J.H., N.D.H. and S.P. in the laboratory of S.P. Analysis of LM-4131 hydrolysis was performed by D.J.H. and P.J.K. in the laboratory of L.J.M. Analysis of LM-4131 inhibition of MAGL and DAGL was performed by D.J.H. and B.C.S. in the laboratory of R.J.C. J.G.-G. completed cannabinoid tetrad behavioral studies in the laboratory of S.P. Ptgs2−/− mice were bred and genotyped by N.B. in the laboratory of J.R. The manuscript was written by D.J.H., L.J.M. and S.P. and edited by all authors.
Corresponding authors
Ethics declarations
Competing interests
D.J.H., L.J.M. and S.P. have submitted a patent application entitled “Compositions and Methods for Substrate-Selective Inhibition of Endocannabinoid Oxygenation”, which includes the compound LM-4131.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 and Supplementary Table 1 (PDF 2606 kb)
Rights and permissions
About this article
Cite this article
Hermanson, D., Hartley, N., Gamble-George, J. et al. Substrate-selective COX-2 inhibition decreases anxiety via endocannabinoid activation. Nat Neurosci 16, 1291–1298 (2013). https://doi.org/10.1038/nn.3480
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3480
This article is cited by
-
Assessment of NSAIDs as potential inhibitors of the fatty acid amide hydrolase I (FAAH-1) using three different primary fatty acid amide substrates in vitro
BMC Pharmacology and Toxicology (2022)
-
The endocannabinoid system in humans: significant associations between anandamide, brain function during reward feedback and a personality measure of reward dependence
Neuropsychopharmacology (2021)
-
The endocannabinoid hydrolase FAAH is an allosteric enzyme
Scientific Reports (2020)
-
Targeting the endocannabinoid system: a predictive, preventive, and personalized medicine-directed approach to the management of brain pathologies
EPMA Journal (2020)
-
Integrating endocannabinoid signaling in the regulation of anxiety and depression
Acta Pharmacologica Sinica (2019)