Abstract
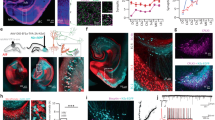
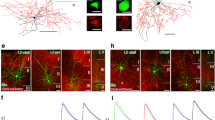
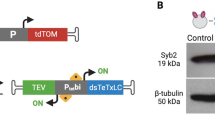
The vast diversity of GABAergic interneurons is believed to endow hippocampal microcircuits with the required flexibility for memory encoding and retrieval. However, dissection of the functional roles of defined interneuron types has been hampered by the lack of cell-specific tools. We identified a precise molecular marker for a population of hippocampal GABAergic interneurons known as oriens lacunosum-moleculare (OLM) cells. By combining transgenic mice and optogenetic tools, we found that OLM cells are important for gating the information flow in CA1, facilitating the transmission of intrahippocampal information (from CA3) while reducing the influence of extrahippocampal inputs (from the entorhinal cortex). Furthermore, we found that OLM cells were interconnected by gap junctions, received direct cholinergic inputs from subcortical afferents and accounted for the effect of nicotine on synaptic plasticity of the Schaffer collateral pathway. Our results suggest that acetylcholine acting through OLM cells can control the mnemonic processes executed by the hippocampus.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
References
Morris, R.G., Garrud, P., Rawlins, J.N. & O'Keefe, J. Place navigation impaired in rats with hippocampal lesions. Nature 297, 681–683 (1982).
Eichenbaum, H. Hippocampus: cognitive processes and neural representations that underlie declarative memory. Neuron 44, 109–120 (2004).
Murray, A.J. et al. Parvalbumin-positive CA1 interneurons are required for spatial working, but not for reference memory. Nat. Neurosci. 14, 297–299 (2011).
Korotkova, T., Fuchs, E.C., Ponomarenko, A., von Engelhardt, J. & Monyer, H. NMDA receptor ablation on parvalbumin-positive interneurons impairs hippocampal synchrony, spatial representations and working memory. Neuron 68, 557–569 (2010).
Freund, T.F. & Buzsaki, G. Interneurons of the hippocampus. Hippocampus 6, 347–470 (1996).
Somogyi, P. & Klausberger, T. Defined types of cortical interneurone structure space and spike timing in the hippocampus. J. Physiol. (Lond.) 562, 9–26 (2005).
Ascoli, G.A. et al. Petilla terminology: nomenclature of features of GABAergic interneurons of the cerebral cortex. Nat. Rev. Neurosci. 9, 557–568 (2008).
Gradinaru, V. et al. Molecular and cellular approaches for diversifying and extending optogenetics. Cell 141, 154–165 (2010).
Maccaferri, G. & McBain, C.J. Passive propagation of LTD to stratum oriens-alveus inhibitory neurons modulates the temporoammonic input to the hippocampal CA1 region. Neuron 15, 137–145 (1995).
Tort, A.B., Rotstein, H.G., Dugladze, T., Gloveli, T. & Kopell, N.J. On the formation of gamma-coherent cell assemblies by oriens lacunosum-moleculare interneurons in the hippocampus. Proc. Natl. Acad. Sci. USA 104, 13490–13495 (2007).
Gloveli, T. et al. Orthogonal arrangement of rhythm-generating microcircuits in the hippocampus. Proc. Natl. Acad. Sci. USA 102, 13295–13300 (2005).
Rotstein, H.G. et al. Slow and fast inhibition and an H-current interact to create a theta rhythm in a model of CA1 interneuron network. J. Neurophysiol. 94, 1509–1518 (2005).
Wulff, P. et al. Hippocampal theta rhythm and its coupling with gamma oscillations require fast inhibition onto parvalbumin-positive interneurons. Proc. Natl. Acad. Sci. USA 106, 3561–3566 (2009).
Nakauchi, S., Brennan, R.J., Boulter, J. & Sumikawa, K. Nicotine gates long-term potentiation in the hippocampal CA1 region via the activation of alpha2* nicotinic ACh receptors. Eur. J. Neurosci. 25, 2666–2681 (2007).
Cutsuridis, V., Cobb, S. & Graham, B.P. Encoding and retrieval in a model of the hippocampal CA1 microcircuit. Hippocampus 20, 423–446 (2010).
Hasselmo, M.E. Neuromodulation: acetylcholine and memory consolidation. Trends Cogn. Sci. 3, 351–359 (1999).
Ishii, K., Wong, J.K. & Sumikawa, K. Comparison of alpha2 nicotinic acetylcholine receptor subunit mRNA expression in the central nervous system of rats and mice. J. Comp. Neurol. 493, 241–260 (2005).
Jia, Y., Yamazaki, Y., Nakauchi, S. & Sumikawa, K. Alpha2 nicotine receptors function as a molecular switch to continuously excite a subset of interneurons in rat hippocampal circuits. Eur. J. Neurosci. 29, 1588–1603 (2009).
Jia, Y., Yamazaki, Y., Nakauchi, S., Ito, K. & Sumikawa, K. Nicotine facilitates long-term potentiation induction in oriens-lacunosum moleculare cells via Ca2+ entry through non-alpha7 nicotinic acetylcholine receptors. Eur. J. Neurosci. 31, 463–476 (2010).
Maccaferri, G. & McBain, C.J. The hyperpolarization-activated current (Ih) and its contribution to pacemaker activity in rat CA1 hippocampal stratum oriens-alveus interneurones. J. Physiol. (Lond.) 497, 119–130 (1996).
Palmer, L.M. & Stuart, G.J. Site of action potential initiation in layer 5 pyramidal neurons. J. Neurosci. 26, 1854–1863 (2006).
Tong, Q., Ye, C.-P., Jones, J.E., Elmquist, J.K. & Lowell, B.B. Synaptic release of GABA by AgRP neurons is required for normal regulation of energy balance. Nat. Neurosci. 11, 998–1000 (2008).
Kee, M.Z., Wuskell, J.P., Loew, L.M., Augustine, G.J. & Sekino, Y. Imaging activity of neuronal populations with new long-wavelength voltage-sensitive dyes. Brain Cell Biol. 36, 157–172 (2008).
Cardin, J.A. et al. Targeted optogenetic stimulation and recording of neurons in vivo using cell type–specific expression of Channelrhodopsin-2. Nat. Protoc. 5, 247–254 (2010).
Maccaferri, G. & Dingledine, R. Control of feedforward dendritic inhibition by NMDA receptor–dependent spike timing in hippocampal interneurons. J. Neurosci. 22, 5462–5472 (2002).
Pouille, F. & Scanziani, M. Enforcement of temporal fidelity in pyramidal cells by somatic feed-forward inhibition. Science 293, 1159–1163 (2001).
Maccaferri, G., Roberts, J.D., Szucs, P., Cottingham, C.A. & Somogyi, P. Cell surface domain specific postsynaptic currents evoked by identified GABAergic neurones in rat hippocampus in vitro. J. Physiol. (Lond.) 524, 91–116 (2000).
Tu, B., Gu, Z., Shen, J.X., Lamb, P.W. & Yakel, J.L. Characterization of a nicotine-sensitive neuronal population in rat entorhinal cortex. J. Neurosci. 29, 10436–10448 (2009).
Blasco-Ibáñez, J.M. & Freund, T.F. Synaptic input of horizontal interneurons in stratum oriens of the hippocampal CA1 subfield: structural basis of feed-back activation. Eur. J. Neurosci. 7, 2170–2180 (1995).
Minneci, F. et al. Signaling properties of stratum oriens interneurons in the hippocampus of transgenic mice expressing EGFP in a subset of somatostatin-containing cells. Hippocampus 17, 538–553 (2007).
Veruki, M.L., Oltedal, L. & Hartveit, E. Electrical coupling and passive membrane properties of AII amacrine cells. J. Neurophysiol. 103, 1456–1466 (2010).
Gulyás, A.I., Gorcs, T.J. & Freund, T.F. Innervation of different peptide-containing neurons in the hippocampus by GABAergic septal afferents. Neuroscience 37, 31–44 (1990).
Gulyás, A.I. et al. Parvalbumin-containing fast-spiking basket cells generate the field potential oscillations induced by cholinergic receptor activation in the hippocampus. J. Neurosci. 30, 15134–15145 (2010).
Klausberger, T. et al. Spike timing of dendrite-targeting bistratified cells during hippocampal network oscillations in vivo. Nat. Neurosci. 7, 41–47 (2004).
Taniguchi, H. et al. A resource of Cre driver lines for genetic targeting of GABAergic neurons in cerebral cortex. Neuron 71, 995–1013 (2011).
Fuchs, E.C. et al. Recruitment of parvalbumin-positive interneurons determines hippocampal function and associated behavior. Neuron 53, 591–604 (2007).
Cardin, J.A. et al. Driving fast-spiking cells induces gamma rhythm and controls sensory responses. Nature 459, 663–667 (2009).
Lovett-Barron, M. et al. Regulation of neuronal input transformations by tunable dendritic inhibition. Nat. Neurosci. 15, 423–430 (2012).
Elfant, D., Pál, B.Z., Emptage, N. & Capogna, M. Specific inhibitory synapses shift the balance from feedforward to feedback inhibition of hippocampal CA1 pyramidal cells. Eur. J. Neurosci. 27, 104–113 (2008).
Kim, S., Guzman, S.J., Hu, H. & Jonas, P. Active dendrites support efficient initiation of dendritic spikes in hippocampal CA3 pyramidal neurons. Nat. Neurosci. 15, 600–606 (2012).
Colom, L.V. Septal networks: relevance to theta rhythm, epilepsy and Alzheimer's disease. J. Neurochem. 96, 609–623 (2006).
Davis, J.A. & Gould, T.J. Associative learning, the hippocampus, and nicotine addiction. Curr. Drug Abuse Rev. 1, 9–19 (2008).
Lynch, M.A. Long-term potentiation and memory. Physiol. Rev. 84, 87–136 (2004).
Dugladze, T. et al. Impaired hippocampal rhythmogenesis in a mouse model of mesial temporal lobe epilepsy. Proc. Natl. Acad. Sci. USA 104, 17530–17535 (2007).
Neymotin, S.A. et al. Ketamine disrupts theta modulation of gamma in a computer model of hippocampus. J. Neurosci. 31, 11733–11743 (2011).
Stanley, E.M., Fadel, J.R. & Mott, D.D. Interneuron loss reduces dendritic inhibition and GABA release in hippocampus of aged rats. Neurobiol. Aging 33, 431 e1–13 (2012).
Franklin, K.B.J. & Paxinos, G. The Mouse Brain in Stereotaxic Coordinates 3rd edn. (Elsevier, 2007).
Marshall, V.M., Allison, J., Templeton, T. & Foote, S.J. Generation of BAC transgenic mice. Methods Mol. Biol. 256, 159–182 (2004).
Leão, R.N., Tan, H.M. & Fisahn, A. Kv7/KCNQ channels control action potential phasing of pyramidal neurons during hippocampal gamma oscillations in vitro. J. Neurosci. 29, 13353–13364 (2009).
Zhou, W.L., Yan, P., Wuskell, J.P., Loew, L.M. & Antic, S.D. Intracellular long-wavelength voltage-sensitive dyes for studying the dynamics of action potentials in axons and thin dendrites. J. Neurosci. Methods 164, 225–239 (2007).
Leão, R.N. et al. A voltage-sensitive dye–based assay for the identification of differentiated neurons derived from embryonic neural stem cell cultures. PLoS ONE 5, e13833 (2010).
Leão, R.N., Colom, L.V., Borgius, L., Kiehn, O. & Fisahn, A. Medial septal dysfunction by Abeta-induced KCNQ channel-block in glutamatergic neurons. Neurobiol. Aging 33, 2046–2061 (2012).
Gezelius, H., Wallen-Mackenzie, A., Enjin, A., Lagerstrom, M. & Kullander, K. Role of glutamate in locomotor rhythm generating neuronal circuitry. J. Physiol. Paris 100, 297–303 (2006).
Enjin, A. et al. Identification of novel spinal cholinergic genetic subtypes disclose Chodl and Pitx2 as markers for fast motor neurons and partition cells. J. Comp. Neurol. 518, 2284–2304 (2010).
Acknowledgements
We thank G. Buzsaki, P. Ascher, B. Lamotte d'Incamps, O. Amaral and E. Hanse for comments on earlier versions of this manuscript. This work was supported by the foundations of Märta och Kjell Beijer, Hållsten and Göran Gustafsson, The Swedish Foundation for International Cooperation in Research and Higher Education, the Brazilian Federal Agency for Support and Evaluation of Graduate Education, the Brazilian National Council of Technological and Scientific Development, the Research Support Agency of the State of Rio Grande do Norte, US National Institutes of Health grant R01EB001963, the Swedish Medical Research Council, the Swedish Brain Foundation, and Uppsala University. K.K. is a Royal Swedish Academy of Sciences Research Fellow supported by a grant from the Knut and Alice Wallenberg Foundation.
Author information
Authors and Affiliations
Contributions
H.G., A. Enjin and K.K. designed and produced the transgenic mice. R.N.L., K.E.L., A.B.L.T., L.M.L. and K.K. designed the experiments. R.N.L., K.E.L., S.M., H.M., K.P. and A. Eriksson performed the experiments. R.N.L., K.E.L., A.B.L.T. and K.K. analyzed data and wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–9 (PDF 2211 kb)
Rights and permissions
About this article
Cite this article
Leão, R., Mikulovic, S., Leão, K. et al. OLM interneurons differentially modulate CA3 and entorhinal inputs to hippocampal CA1 neurons. Nat Neurosci 15, 1524–1530 (2012). https://doi.org/10.1038/nn.3235
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3235
This article is cited by
-
mTORC1-mediated acquisition of reward-related representations by hippocampal somatostatin interneurons
Molecular Brain (2023)
-
Identifying foetal forebrain interneurons as a target for monogenic autism risk factors and the polygenic 16p11.2 microdeletion
BMC Neuroscience (2023)
-
Lipoic acid inhibits cognitive impairment induced by multiple cell phones in young male rats: role of Sirt1 and Atg7 pathway
Scientific Reports (2023)
-
Optogenetics: implications for Alzheimer’s disease research and therapy
Molecular Brain (2022)
-
Stimulation of protein synthesis by optogenetic and chemical induction of excitatory synaptic plasticity in hippocampal somatostatin interneurons
Molecular Brain (2022)