Abstract
Maintaining energy balance is of paramount importance for metabolic health and survival. It is achieved through the coordinated regulation of neuronal circuits that control a wide range of physiological processes affecting energy intake and expenditure, such as feeding, metabolic rate, locomotor activity, arousal, growth and reproduction. Neuronal populations distributed throughout the CNS but highly enriched in the mediobasal hypothalamus, sense hormonal, nutrient and neuronal signals of systemic energy status and relay this information to secondary neurons that integrate the information and regulate distinct physiological parameters in a manner that promotes energy homeostasis. To achieve this, it is critical that neuronal circuits provide information about short-term changes in nutrient availability in the larger context of long-term energy status. For example, the same signals lead to different cellular and physiological responses if delivered under fasted versus fed conditions. Thus, there is a clear need to have mechanisms that rapidly and reversibly adjust responsiveness of hypothalamic circuits to acute changes in nutrient availability.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Feldman, D.E. Synaptic mechanisms for plasticity in neocortex. Annu. Rev. Neurosci. 32, 33–55 (2009).
Turrigiano, G.G. & Nelson, S.B. Homeostatic plasticity in the developing nervous system. Nat. Rev. Neurosci. 5, 97–107 (2004).
Zhang, W. & Linden, D.J. The other side of the engram: experience-driven changes in neuronal intrinsic excitability. Nat. Rev. Neurosci. 4, 885–900 (2003).
Horvath, T.L. & Diano, S. The floating blueprint of hypothalamic feeding circuits. Nat. Rev. Neurosci. 5, 662–667 (2004).
Horvath, T.L. The hardship of obesity: a soft-wired hypothalamus. Nat. Neurosci. 8, 561–565 (2005).
Horvath, T.L. Synaptic plasticity in energy balance regulation. Obesity (Silver Spring) 14 (suppl. 5), 228S–233S (2006).
Pinto, S. et al. Rapid rewiring of arcuate nucleus feeding circuits by leptin. Science 304, 110–115 (2004).
Saper, C.B., Chou, T.C. & Elmquist, J.K. The need to feed: homeostatic and hedonic control of eating. Neuron 36, 199–211 (2002).
Schwartz, M.W. & Porte, D. Jr. Diabetes, obesity, and the brain. Science 307, 375–379 (2005).
Mountjoy, K.G., Mortrud, M.T., Low, M.J., Simerly, R.B. & Cone, R.D. Localization of the melanocortin-4 receptor (MC4-R) in neuroendocrine and autonomic control circuits in the brain. Mol. Endocrinol. 8, 1298–1308 (1994).
Ollmann, M.M. et al. Antagonism of central melanocortin receptors in vitro and in vivo by agouti-related protein. Science 278, 135–138 (1997).
Fan, W., Boston, B.A., Kesterson, R.A., Hruby, V.J. & Cone, R.D. Role of melanocortinergic neurons in feeding and the agouti obesity syndrome. Nature 385, 165–168 (1997).
Bagnol, D. et al. Anatomy of an endogenous antagonist: relationship between Agouti-related protein and proopiomelanocortin in brain. J. Neurosci. 19, RC26 (1999).
Mountjoy, K.G., Wu, C.S., Cornish, J. & Callon, K.E. alpha-MSH and desacetyl-alpha-MSH signaling through melanocortin receptors. Ann. NY Acad. Sci. 994, 58–65 (2003).
Smith, M.A. et al. Melanocortins and agouti-related protein modulate the excitability of two arcuate nucleus neuron populations by alteration of resting potassium conductances. J. Physiol. (Lond.) 578, 425–438 (2007).
Vaisse, C., Clement, K., Guy-Grand, B. & Froguel, P. A frameshift mutation in human MC4R is associated with a dominant form of obesity. Nat. Genet. 20, 113–114 (1998).
Yeo, G.S. et al. A frameshift mutation in MC4R associated with dominantly inherited human obesity. Nat. Genet. 20, 111–112 (1998).
Huszar, D. et al. Targeted disruption of the melanocortin-4 receptor results in obesity in mice. Cell 88, 131–141 (1997).
Cone, R.D. et al. The arcuate nucleus as a conduit for diverse signals relevant to energy homeostasis. Int. J. Obes. Relat. Metab. Disord. 25 (suppl. 5), S63–S67 (2001).
Mizuno, T.M. et al. Hypothalamic pro-opiomelanocortin mRNA is reduced by fasting and [corrected] in ob/ob and db/db mice, but is stimulated by leptin. Diabetes 47, 294–297 (1998).
Cowley, M.A. et al. Leptin activates anorexigenic POMC neurons through a neural network in the arcuate nucleus. Nature 411, 480–484 (2001).
Cowley, M.A. et al. Electrophysiological actions of peripheral hormones on melanocortin neurons. Ann. NY Acad. Sci. 994, 175–186 (2003).
Takahashi, K.A. & Cone, R.D. Fasting induces a large, leptin-dependent increase in the intrinsic action potential frequency of orexigenic arcuate nucleus neuropeptide Y/Agouti-related protein neurons. Endocrinology 146, 1043–1047 (2005).
Elias, C.F. et al. Leptin differentially regulates NPY and POMC neurons projecting to the lateral hypothalamic area. Neuron 23, 775–786 (1999).
Glaum, S.R. et al. Leptin, the obese gene product, rapidly modulates synaptic transmission in the hypothalamus. Mol. Pharmacol. 50, 230–235 (1996).
Cowley, M.A. et al. The distribution and mechanism of action of ghrelin in the CNS demonstrates a novel hypothalamic circuit regulating energy homeostasis. Neuron 37, 649–661 (2003).
van den Top, M., Lee, K., Whyment, A.D., Blanks, A.M. & Spanswick, D. Orexigen-sensitive NPY/AgRP pacemaker neurons in the hypothalamic arcuate nucleus. Nat. Neurosci. 7, 493–494 (2004).
Vong, L. et al. Leptin action on GABAergic neurons prevents obesity and reduces inhibitory tone to POMC neurons. Neuron 71, 142–154 (2011).
Horvath, T.L., Naftolin, F., Kalra, S.P. & Leranth, C. Neuropeptide Y innervation of beta-endorphin–containing cells in the rat mediobasal hypothalamus: a light and electron microscopic double immunostaining analysis. Endocrinology 131, 2461–2467 (1992).
Roseberry, A.G., Liu, H., Jackson, A.C., Cai, X. & Friedman, J.M. Neuropeptide Y–mediated inhibition of proopiomelanocortin neurons in the arcuate nucleus shows enhanced desensitization in ob/ob mice. Neuron 41, 711–722 (2004).
Baskin, D.G. et al. Increased expression of mRNA for the long form of the leptin receptor in the hypothalamus is associated with leptin hypersensitivity and fasting. Diabetes 47, 538–543 (1998).
Baskin, D.G., Breininger, J.F., Bonigut, S. & Miller, M.A. Leptin binding in the arcuate nucleus is increased during fasting. Brain Res. 828, 154–158 (1999).
Luckman, S.M., Rosenzweig, I. & Dickson, S.L. Activation of arcuate nucleus neurons by systemic administration of leptin and growth hormone–releasing peptide-6 in normal and fasted rats. Neuroendocrinology 70, 93–100 (1999).
Gao, Q. et al. Anorectic estrogen mimics leptin′s effect on the rewiring of melanocortin cells and Stat3 signaling in obese animals. Nat. Med. 13, 89–94 (2007).
Gyengesi, E. et al. Corticosterone regulates synaptic input organization of POMC and NPY/AgRP neurons in adult mice. Endocrinology 151, 5395–5402 (2010).
Qiu, J., Fang, Y., Ronnekleiv, O.K. & Kelly, M.J. Leptin excites proopiomelanocortin neurons via activation of TRPC channels. J. Neurosci. 30, 1560–1565 (2010).
Padilla, S.L., Reef, D. & Zeltser, L.M. Defining POMC neurons using transgenic reagents: impact of transient Pomc expression in diverse immature neuronal populations. Endocrinology 153, 1219–1231 (2012).
Münzberg, H., Huo, L., Nillni, E.A., Hollenberg, A.N. & Bjorbaek, C. Role of signal transducer and activator of transcription 3 in regulation of hypothalamic proopiomelanocortin gene expression by leptin. Endocrinology 144, 2121–2131 (2003).
Williams, K.W. et al. Segregation of acute leptin and insulin effects in distinct populations of arcuate proopiomelanocortin neurons. J. Neurosci. 30, 2472–2479 (2010).
Hentges, S.T., Otero-Corchon, V., Pennock, R.L., King, C.M. & Low, M.J. Proopiomelanocortin expression in both GABA and glutamate neurons. J. Neurosci. 29, 13684–13690 (2009).
Fioramonti, X. et al. Characterization of glucosensing neuron subpopulations in the arcuate nucleus: integration in neuropeptide Y and pro-opio melanocortin networks? Diabetes 56, 1219–1227 (2007).
Padilla, S.L., Carmody, J.S. & Zeltser, L.M. Pomc-expressing progenitors give rise to antagonistic neuronal populations in hypothalamic feeding circuits. Nat. Med. 16, 403–405 (2010).
Sohn, J.W. et al. Serotonin 2C receptor activates a distinct population of arcuate pro-opiomelanocortin neurons via TRPC channels. Neuron 71, 488–497 (2011).
King, C.M. & Hentges, S.T. Relative number and distribution of murine hypothalamic proopiomelanocortin neurons innervating distinct target sites. PLoS ONE 6, e25864 (2011).
Sternson, S.M., Shepherd, G.M. & Friedman, J.M. Topographic mapping of VMH → arcuate nucleus microcircuits and their reorganization by fasting. Nat. Neurosci. 8, 1356–1363 (2005).
Yang, Y., Atasoy, D., Su, H.H. & Sternson, S.M. Hunger states switch a flip-flop memory circuit via a synaptic AMPK-dependent positive feedback loop. Cell 146, 992–1003 (2011).
Liu, T. et al. Fasting activation of AgRP neurons requires NMDA receptors and involves spinogenesis and increased excitatory tone. Neuron 73, 511–522 (2012).
Holtmaat, A. & Svoboda, K. Experience-dependent structural synaptic plasticity in the mammalian brain. Nat. Rev. Neurosci. 10, 647–658 (2009).
Luquet, S., Perez, F.A., Hnasko, T.S. & Palmiter, R.D. NPY/AgRP neurons are essential for feeding in adult mice but can be ablated in neonates. Science 310, 683–685 (2005).
Dietrich, M.O. et al. Agrp neurons mediate Sirt1's action on the melanocortin system and energy balance: roles for Sirt1 in neuronal firing and synaptic plasticity. J. Neurosci. 30, 11815–11825 (2010).
Andrews, Z.B. et al. UCP2 mediates ghrelin's action on NPY/AgRP neurons by lowering free radicals. Nature 454, 846–851 (2008).
Atasoy, D., Betley, J.N., Su, H.H. & Sternson, S.M. Deconstruction of a neural circuit for hunger. Nature 488, 172–177 (2012).
Xu, Y., O'Brien, W.G. III, Lee, C.C., Myers, M.G. Jr. & Tong, Q. Role of GABA release from leptin receptor–expressing neurons in body weight regulation. Endocrinology 153, 2223–2233 (2012).
Münzberg, H. et al. Appropriate inhibition of orexigenic hypothalamic arcuate nucleus neurons independently of leptin receptor/STAT3 signaling. J. Neurosci. 27, 69–74 (2007).
Diano, S. et al. Ghrelin controls hippocampal spine synapse density and memory performance. Nat. Neurosci. 9, 381–388 (2006).
Kohno, D., Sone, H., Minokoshi, Y. & Yada, T. Ghrelin raises [Ca2+]i via AMPK in hypothalamic arcuate nucleus NPY neurons. Biochem. Biophys. Res. Commun. 366, 388–392 (2008).
Spanswick, D., Smith, M.A., Groppi, V.E., Logan, S.D. & Ashford, M.L. Leptin inhibits hypothalamic neurons by activation of ATP-sensitive potassium channels. Nature 390, 521–525 (1997).
Atasoy, D., Aponte, Y., Su, H.H. & Sternson, S.M.A. FLEX switch targets Channelrhodopsin-2 to multiple cell types for imaging and long-range circuit mapping. J. Neurosci. 28, 7025–7030 (2008).
Betley, J.N. et al. Stringent specificity in the construction of a GABAergic presynaptic inhibitory circuit. Cell 139, 161–174 (2009).
Facchinetti, F. et al. Structural, neurochemical and behavioural consequences of neonatal blockade of NMDA receptor through chronic treatment with CGP 39551 or MK-801. Brain Res. Dev. Brain Res. 74, 219–224 (1993).
Herlenius, E. & Lagercrantz, H. Development of neurotransmitter systems during critical periods. Exp. Neurol. 190 (suppl. 1), 8–21 (2004).
Matsumoto, A. & Arai, Y. Morphologic evidence for intranuclear circuits in the hypothalamic arcuate nucleus. Exp. Neurol. 59, 404–412 (1978).
Horvath, T.L. & Gao, X.B. Input organization and plasticity of hypocretin neurons: possible clues to obesity's association with insomnia. Cell Metab. 1, 279–286 (2005).
Chemelli, R.M. et al. Narcolepsy in orexin knockout mice: molecular genetics of sleep regulation. Cell 98, 437–451 (1999).
Horvath, T.L., Diano, S. & van den Pol, A.N. Synaptic interaction between hypocretin (orexin) and neuropeptide Y cells in the rodent and primate hypothalamus: a novel circuit implicated in metabolic and endocrine regulations. J. Neurosci. 19, 1072–1087 (1999).
Sakurai, T. et al. Orexins and orexin receptors: a family of hypothalamic neuropeptides and G protein–coupled receptors that regulate feeding behavior. Cell 92, 573–585 (1998).
Appelbaum, L. et al. Circadian and homeostatic regulation of structural synaptic plasticity in hypocretin neurons. Neuron 68, 87–98 (2010).
Jo, Y.H., Chen, Y.J., Chua, S.C. Jr., Talmage, D.A. & Role, L.W. Integration of endocannabinoid and leptin signaling in an appetite-related neural circuit. Neuron 48, 1055–1066 (2005).
Crosby, K.M., Inoue, W., Pittman, Q.J. & Bains, J.S. Endocannabinoids gate state-dependent plasticity of synaptic inhibition in feeding circuits. Neuron 71, 529–541 (2011).
Naleid, A.M., Grace, M.K., Cummings, D.E. & Levine, A.S. Ghrelin induces feeding in the mesolimbic reward pathway between the ventral tegmental area and the nucleus accumbens. Peptides 26, 2274–2279 (2005).
Abizaid, A., Gao, Q. & Horvath, T.L. Thoughts for food: brain mechanisms and peripheral energy balance. Neuron 51, 691–702 (2006).
Korotkova, T.M., Brown, R.E., Sergeeva, O.A., Ponomarenko, A.A. & Haas, H.L. Effects of arousal- and feeding-related neuropeptides on dopaminergic and GABAergic neurons in the ventral tegmental area of the rat. Eur. J. Neurosci. 23, 2677–2685 (2006).
Dietrich, M.O. et al. AgRP neurons regulate development of dopamine neuronal plasticity and nonfood-associated behaviors. Nat. Neurosci. 15, 1108–1110 (2012).
Lim, B.K., Huang, K.W., Grueter, B.A., Rothwell, P.E. & Malenka, R.C. Anhedonia requires MC4R-mediated synaptic adaptations in nucleus accumbens. Nature 487, 183–189 (2012).
Liu, J. et al. The melanocortinergic pathway is rapidly recruited by emotional stress and contributes to stress-induced anorexia and anxiety-like behavior. Endocrinology 148, 5531–5540 (2007).
Halaas, J.L. et al. Physiological response to long-term peripheral and central leptin infusion in lean and obese mice. Proc. Natl. Acad. Sci. USA 94, 8878–8883 (1997).
Enriori, P.J. et al. Diet-induced obesity causes severe but reversible leptin resistance in arcuate melanocortin neurons. Cell Metab. 5, 181–194 (2007).
Myers, M.G. Jr., Leibel, R.L., Seeley, R.J. & Schwartz, M.W. Obesity and leptin resistance: distinguishing cause from effect. Trends Endocrinol. Metab. 21, 643–651 (2010).
Horvath, T.L. et al. Synaptic input organization of the melanocortin system predicts diet-induced hypothalamic reactive gliosis and obesity. Proc. Natl. Acad. Sci. USA 107, 14875–14880 (2010).
Diano, S. et al. Peroxisome proliferation-associated control of reactive oxygen species sets melanocortin tone and feeding in diet-induced obesity. Nat. Med. 17, 1121–1127 (2011).
Acknowledgements
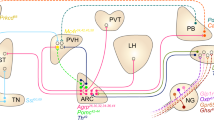
The authors are grateful to C.-X. Yi for help generating Figure 1.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Zeltser, L., Seeley, R. & Tschöp, M. Synaptic plasticity in neuronal circuits regulating energy balance. Nat Neurosci 15, 1336–1342 (2012). https://doi.org/10.1038/nn.3219
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3219
This article is cited by
-
Increased intrinsic and synaptic excitability of hypothalamic POMC neurons underlies chronic stress-induced behavioral deficits
Molecular Psychiatry (2023)
-
Mass spectrometry imaging of mice brain lipid profile changes over time under high fat diet
Scientific Reports (2021)
-
AgRP neurons trigger long-term potentiation and facilitate food seeking
Translational Psychiatry (2021)
-
Chronic unpredictable stress induces depression-related behaviors by suppressing AgRP neuron activity
Molecular Psychiatry (2021)
-
An excitatory ventromedial hypothalamus to paraventricular thalamus circuit that suppresses food intake
Nature Communications (2020)