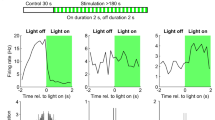
Abstract
A large variety of GABAergic interneurons control information processing in the hippocampal circuits governing the formation of neuronal representations. Whether distinct hippocampal interneuron types contribute differentially to information processing during behavior is not known. We employed a new technique for recording and labeling interneurons and pyramidal cells in drug-free, freely moving rats. Recorded parvalbumin-expressing basket interneurons innervated somata and proximal pyramidal cell dendrites, whereas nitric oxide synthase– and neuropeptide Y–expressing ivy cells provided synaptic and extrasynaptic dendritic modulation. Basket and ivy cells showed distinct spike-timing dynamics, firing at different rates and times during theta and ripple oscillations. Basket, but not ivy, cells changed their firing rates during movement, sleep and quiet wakefulness, suggesting that basket cells coordinate cell assemblies in a behavioral state–contingent manner, whereas persistently firing ivy cells might control network excitability and homeostasis. Different interneuron types provide GABA to specific subcellular domains at defined times and rates, thereby differentially controlling network activity during behavior.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Buzsaki, G. Rhythms of the Brain (Oxford University Press, New York, 2006).
Cardin, J.A. et al. Driving fast-spiking cells induces gamma rhythm and controls sensory responses. Nature 459, 663–667 (2009).
Cobb, S.R., Buhl, E.H., Halasy, K., Paulsen, O. & Somogyi, P. Synchronization of neuronal activity in hippocampus by individual GABAergic interneurons. Nature 378, 75–78 (1995).
Pouille, F. & Scanziani, M. Enforcement of temporal fidelity in pyramidal cells by somatic feed-forward inhibition. Science 293, 1159–1163 (2001).
Buhl, E.H., Halasy, K. & Somogyi, P. Diverse sources of hippocampal unitary inhibitory postsynaptic potentials and the number of synaptic release sites. Nature 368, 823–828 (1994).
Pawelzik, H., Hughes, D.I. & Thomson, A.M. Physiological and morphological diversity of immunocytochemically defined parvalbumin- and cholecystokinin-positive interneurones in CA1 of the adult rat hippocampus. J. Comp. Neurol. 443, 346–367 (2002).
Lamsa, K.P., Heeroma, J.H., Somogyi, P., Rusakov, D.A. & Kullmann, D.M. Anti-Hebbian long-term potentiation in the hippocampal feedback inhibitory circuit. Science 315, 1262–1266 (2007).
Fuchs, E.C. et al. Recruitment of parvalbumin-positive interneurons determines hippocampal function and associated behavior. Neuron 53, 591–604 (2007).
Cossart, R. et al. Interneurons targeting similar layers receive synaptic inputs with similar kinetics. Hippocampus 16, 408–420 (2006).
Glickfeld, L.L. & Scanziani, M. Distinct timing in the activity of cannabinoid-sensitive and cannabinoid-insensitive basket cells. Nat. Neurosci. 9, 807–815 (2006).
Hájos, N. et al. Spike timing of distinct types of GABAergic interneuron during hippocampal gamma oscillations in vitro. J. Neurosci. 24, 9127–9137 (2004).
Gloveli, T. et al. Differential involvement of oriens/pyramidale interneurones in hippocampal network oscillations in vitro. J. Physiol. (Lond.) 562, 131–147 (2005).
Mann, E.O., Suckling, J.M., Hajos, N., Greenfield, S.A. & Paulsen, O. Perisomatic feedback inhibition underlies cholinergically induced fast network oscillations in the rat hippocampus in vitro. Neuron 45, 105–117 (2005).
Gulyás, A.I. et al. Parvalbumin-containing fast-spiking basket cells generate the field potential oscillations induced by cholinergic receptor activation in the hippocampus. J. Neurosci. 30, 15134–15145 (2010).
Klausberger, T. et al. Brain-state- and cell-type-specific firing of hippocampal interneurons in vivo. Nature 421, 844–848 (2003).
Gentet, L.J., Avermann, M., Matyas, F., Staiger, J.F. & Petersen, C.C. Membrane potential dynamics of GABAergic neurons in the barrel cortex of behaving mice. Neuron 65, 422–435 (2010).
Gentet, L.J. et al. Unique functional properties of somatostatin-expressing GABAergic neurons in mouse barrel cortex. Nat. Neurosci. 15, 607–612 (2012).
Csicsvari, J., Hirase, H., Czurko, A., Mamiya, A. & Buzsaki, G. Oscillatory coupling of hippocampal pyramidal cells and interneurons in the behaving Rat. J. Neurosci. 19, 274–287 (1999).
Maurer, A.P., Cowen, S.L., Burke, S.N., Barnes, C.A. & McNaughton, B.L. Phase precession in hippocampal interneurons showing strong functional coupling to individual pyramidal cells. J. Neurosci. 26, 13485–13492 (2006).
Ego–Stengel, V. & Wilson, M.A. Spatial selectivity and theta phase precession in CA1 interneurons. Hippocampus 17, 161–174 (2007).
Royer, S. et al. Control of timing, rate and bursts of hippocampal place cells by dendritic and somatic inhibition. Nat. Neurosci. 15, 769–775 (2012).
Pinault, D. A novel single-cell staining procedure performed in vivo under electrophysiological control: morpho-functional features of juxtacellularly labeled thalamic cells and other central neurons with biocytin or Neurobiotin. J. Neurosci. Methods 65, 113–136 (1996).
Haiss, F., Butovas, S. & Schwarz, C. A miniaturized chronic microelectrode drive for awake behaving head restrained mice and rats. J. Neurosci. Methods 187, 67–72 (2010).
Fazzari, P. et al. Control of cortical GABA circuitry development by Nrg1 and ErbB4 signaling. Nature 464, 1376–1380 (2010).
Fuentealba, P. et al. Ivy cells: a population of nitric oxide–producing, slow-spiking GABAergic neurons and their involvement in hippocampal network activity. Neuron 57, 917–929 (2008).
Szabadics, J. & Soltesz, I. Functional specificity of mossy fiber innervation of GABAergic cells in the hippocampus. J. Neurosci. 29, 4239–4251 (2009).
Tricoire, L. et al. Common origins of hippocampal ivy and nitric oxide synthase–expressing neurogliaform cells. J. Neurosci. 30, 2165–2176 (2010).
Epsztein, J., Brecht, M. & Lee, A.K. Intracellular determinants of hippocampal CA1 place and silent cell activity in a novel environment. Neuron 70, 109–120 (2011).
Harvey, C.D., Collman, F., Dombeck, D.A. & Tank, D.W. Intracellular dynamics of hippocampal place cells during virtual navigation. Nature 461, 941–946 (2009).
O'Keefe, J. Place units in the hippocampus of the freely moving rat. Exp. Neurol. 51, 78–109 (1976).
Oláh, S. et al. Regulation of cortical microcircuits by unitary GABA-mediated volume transmission. Nature 461, 1278–1281 (2009).
Lubenov, E.V. & Siapas, A.G. Hippocampal theta oscillations are travelling waves. Nature 459, 534–539 (2009).
Hartwich, K., Pollak, T. & Klausberger, T. Distinct firing patterns of identified basket and dendrite-targeting interneurons in the prefrontal cortex during hippocampal theta and local spindle oscillations. J. Neurosci. 29, 9563–9574 (2009).
Klausberger, T. & Somogyi, P. Neuronal diversity and temporal dynamics: the unity of hippocampal circuit operations. Science 321, 53–57 (2008).
Ylinen, A. et al. Sharp wave–associated high-frequency oscillation (200 Hz) in the intact hippocampus: network and intracellular mechanisms. J. Neurosci. 15, 30–46 (1995).
Vida, I., Bartos, M. & Jonas, P. Shunting inhibition improves robustness of gamma oscillations in hippocampal interneuron networks by homogenizing firing rates. Neuron 49, 107–117 (2006).
Hu, H., Martina, M. & Jonas, P. Dendritic mechanisms underlying rapid synaptic activation of fast-spiking hippocampal interneurons. Science 327, 52–58 (2010).
Losonczy, A., Zemelman, B., Vaziri, A. & Magee, J. Network mechanisms of theta related neuronal activity in hippocampal CA1 pyramidal neurons. Nat. Neurosci. 13, 967–972 (2010).
Lovett–Barron, M. Regulation of neuronal input transformations by tunable dendritic inhibition. Nat. Neurosci. 15, 423–430 (2012).
Freund, T.F. & Buzsaki, G. Interneurons of the hippocampus. Hippocampus 6, 347–470 (1996).
Foster, D.J. & Wilson, M.A. Reverse replay of behavioral sequences in hippocampal place cells during the awake state. Nature 440, 680–683 (2006).
Diba, K. & Buzsaki, G. Forward and reverse hippocampal place-cell sequences during ripples. Nat. Neurosci. 10, 1241–1242 (2007).
Markwardt, S.J., Dieni, C.V., Wadiche, J.I. & Overstreet-Wadiche, L. Ivy/neurogliaform interneurons coordinate activity in the neurogenic niche. Nat. Neurosci. 14, 1407–1409 (2011).
Vizi, E.S. Role of high-affinity receptors and membrane transporters in nonsynaptic communication and drug action in the central nervous system. Pharm. Rev. 52, 63–89 (2000).
Lee, A.K., Manns, I.D., Sakmann, B. & Brecht, M. Whole-cell recordings in freely moving rats. Neuron 51, 399–407 (2006).
Hacker, S.O., White, C.E. & Black, I.H. A comparison of target-controlled infusion versus volatile inhalant anesthesia for heart rate, respiratory rate, and recovery time in a rat model. Contemp. Top. Lab. Anim. Sci. 44, 7–12 (2005).
Lasztóczi, B., Tukker, J.J., Somogyi, P. & Klausberger, T. Terminal field and firing selectivity of cholecystokinin-expressing interneurons in the hippocampal CA3 area. J. Neurosci. 31, 18073–18093 (2011).
Klausberger, T. et al. Complementary roles of cholecystokinin- and parvalbumin-expressing GABAergic neurons in hippocampal network oscillations. J. Neurosci. 25, 9782–9793 (2005).
Louie, K. & Wilson, M.A. Temporally structured replay of awake hippocampal ensemble activity during rapid eye movement sleep. Neuron 29, 145–156 (2001).
Berens, P. CircStat: a MATLAB toolbox for circular statistics. J. Stat. Softw. 31, 1–21 (2009).
Acknowledgements
The authors thank A. Koroknai for the reconstruction of the dendritic tree of cell D26p, I. Lukacs for the reconstruction of cell TV08k, J. Somogyi for confocal microscopic data acquisition and illustrations, K. Detzner for electron microscopy and immunohistochemistry, R. Hauer and E. Borok for histological processing, T. Sakatani for contributions to microdrive development, B. Micklem for help with the preparation of illustrations, J. O'Neill for help with place cell analysis, Y. Dalezios and G. Dorffner for advice on statistics, and R. Guillery, D. Dupret, M. Capogna, J. Csicsvari and L. Marton for comments on an earlier version of the manuscript. This work was supported in part by grant 242689 of the European Research Council, grant SCIC03 of the Vienna Science and Technology Fund and grant W1205 of the Austrian Science Fund.
Author information
Authors and Affiliations
Contributions
All authors contributed to experiments, analysis and manuscript preparation.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figure 1 and Supplementary Table 1 (PDF 542 kb)
Rights and permissions
About this article
Cite this article
Lapray, D., Lasztoczi, B., Lagler, M. et al. Behavior-dependent specialization of identified hippocampal interneurons. Nat Neurosci 15, 1265–1271 (2012). https://doi.org/10.1038/nn.3176
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3176
This article is cited by
-
A developmental increase of inhibition promotes the emergence of hippocampal ripples
Nature Communications (2024)
-
Associations between in vitro, in vivo and in silico cell classes in mouse primary visual cortex
Nature Communications (2023)
-
Robust Resting-State Dynamics in a Large-Scale Spiking Neural Network Model of Area CA3 in the Mouse Hippocampus
Cognitive Computation (2023)
-
Sex-specific regulation of inhibition and network activity by local aromatase in the mouse hippocampus
Nature Communications (2022)
-
The role of inhibitory circuits in hippocampal memory processing
Nature Reviews Neuroscience (2022)