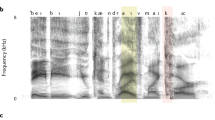
Abstract
Neuronal oscillations are ubiquitous in the brain and may contribute to cognition in several ways: for example, by segregating information and organizing spike timing. Recent data show that delta, theta and gamma oscillations are specifically engaged by the multi-timescale, quasi-rhythmic properties of speech and can track its dynamics. We argue that they are foundational in speech and language processing, 'packaging' incoming information into units of the appropriate temporal granularity. Such stimulus-brain alignment arguably results from auditory and motor tuning throughout the evolution of speech and language and constitutes a natural model system allowing auditory research to make a unique contribution to the issue of how neural oscillatory activity affects human cognition.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Heimbauer, L.A., Beran, M.J. & Owren, M.J. A chimpanzee recognizes synthetic speech with significantly reduced acoustic cues to phonetic content. Curr. Biol. 21, 1210–1214 (2011).
Liberman, A.M. & Mattingly, I.G. The motor theory of speech perception revised. Cognition 21, 1–36 (1985).
Poeppel, D. The analysis of speech in different temporal integration windows: cerebral lateralization as 'asymmetric sampling in time'. Speech Commun. 41, 245–255 (2003).
Shannon, R.V. et al. Speech recognition with primarily temporal cues. Science 270, 303–304 (1995).
Lorenzi, C. et al. Speech perception problems of the hearing impaired reflect inability to use temporal fine structure. Proc. Natl. Acad. Sci. USA 103, 18866–18869 (2006).
Adank, P. & Janse, E. Perceptual learning of time-compressed and natural fast speech. J. Acoust. Soc. Am. 126, 2649–2659 (2009).
Giraud, A.L. et al. Endogenous cortical rhythms determine cerebral specialization for speech perception and production. Neuron 56, 1127–1134 (2007).
Ghitza, O. Linking speech perception and neurophysiology: speech decoding guided by cascaded oscillators locked to the input rhythm. Front. Psychol. 2, 130 (2011).
Ghitza, O. & Greenberg, S. On the possible role of brain rhythms in speech perception: intelligibility of time-compressed speech with periodic and aperiodic insertions of silence. Phonetica 66, 113–126 (2009).
Liégeois-Chauvel, C. et al. Temporal envelope processing in the human left and right auditory cortices. Cereb. Cortex 14, 731–740 (2004).
Ding, N. & Simon, J.Z. Neural representations of complex temporal modulations in the human auditory cortex. J. Neurophysiol. 102, 2731–2743 (2009).
Luo, H. & Poeppel, D. Phase patterns of neuronal responses reliably discriminate speech in human auditory cortex. Neuron 54, 1001–1010 (2007).
Ahissar, E. et al. Speech comprehension is correlated with temporal response patterns recorded from auditory cortex. Proc. Natl. Acad. Sci. USA 98, 13367–13372 (2001).
Abrams, D.A. et al. Right-hemisphere auditory cortex is dominant for coding syllable patterns in speech. J. Neurosci. 28, 3958–3965 (2008).
Nourski, K.V. et al. Temporal envelope of time-compressed speech represented in the human auditory cortex. J. Neurosci. 29, 15564–15574 (2009).
Canolty, R.T. & Knight, R.T. The functional role of cross-frequency coupling. Trends Cogn. Sci. 14, 506–515 (2010).
Schroeder, C.E. & Lakatos, P. Low-frequency neuronal oscillations as instruments of sensory selection. Trends Neurosci. 32, 9–18 (2009).
Atencio, C.A., Sharpee, T.O. & Schreiner, C.E. Cooperative nonlinearities in auditory cortical neurons. Neuron 58, 956–966 (2008).
Sakata, S. & Harris, K.D. Laminar structure of spontaneous and sensory-evoked population activity in auditory cortex. Neuron 64, 404–418 (2009).
Wang, X.J. Neurophysiological and computational principles of cortical rhythms in cognition. Physiol. Rev. 90, 1195–1268 (2010).
Börgers, C., Epstein, S. & Kopell, N.J. Background gamma rhythmicity and attention in cortical local circuits: a computational study. Proc. Natl. Acad. Sci. USA 102, 7002–7007 (2005).
Fries, P., Nikolic, D. & Singer, W. The gamma cycle. Trends Neurosci. 30, 309–316 (2007).
Kayser, C., Logothetis, N.K. & Panzeri, S. Millisecond encoding precision of auditory cortex neurons. Proc. Natl. Acad. Sci. USA 107, 16976–16981 (2010).
Chang, E.F. et al. Categorical speech representation in human superior temporal gyrus. Nat. Neurosci. 13, 1428–1432 (2010).
Rauschecker, J.P. & Scott, S.K. Maps and streams in the auditory cortex: nonhuman primates illuminate human speech processing. Nat. Neurosci. 12, 718–724 (2009).
Kopell, N. et al. Gamma and theta rhythms in biophysical models of hippocampal circuits. in Hippocampal Microcircuits: A Computational Modeller's Resource Book (eds. Cutsuridis, V., Graham, B.P., Cobb, S. & Vida, I.) Ch 15 (Springer, 2011).
Shamir, M. et al. Representation of time-varying stimuli by a network exhibiting oscillations on a faster time scale. PLoS Comput. Biol. 5, e1000370 (2009).
Atencio, C.A. & Schreiner, C.E. Columnar connectivity and laminar processing in cat primary auditory cortex. PLoS ONE 5, e9521 (2010).
Zatorre, R.J., Belin, P. & Penhune, V.B. Structure and function of auditory cortex: music and speech. Trends Cogn. Sci. 6, 37–46 (2002).
Boemio, A. et al. Hierarchical and asymmetric temporal sensitivity in human auditory cortices. Nat. Neurosci. 8, 389–395 (2005).
Jamison, H.L. et al. Hemispheric specialization for processing auditory nonspeech stimuli. Cereb. Cortex 16, 1266–1275 (2006).
Obleser, J., Eisner, F. & Kotz, S.A. Bilateral speech comprehension reflects differential sensitivity to spectral and temporal features. J. Neurosci. 28, 8116–8123 (2008).
Morillon, B. et al. Neurophysiological origin of human brain asymmetry for speech and language. Proc. Natl. Acad. Sci. USA 107, 18688–18693 (2010).
Telkemeyer, S. et al. Sensitivity of newborn auditory cortex to the temporal structure of sounds. J. Neurosci. 29, 14726–14733 (2009).
Hutsler, J. & Galuske, R.A. Hemispheric asymmetries in cerebral cortical networks. Trends Neurosci. 26, 429–435 (2003).
Gireesh, E.D. & Plenz, D. Neuronal avalanches organize as nested theta- and beta/gamma-oscillations during development of cortical layer 2/3. Proc. Natl. Acad. Sci. USA 105, 7576–7581 (2008).
Pagnamenta, A.T. et al. Characterization of a family with rare deletions in CNTNAP5 and DOCK4 suggests novel risk loci for autism and dyslexia. Biol. Psychiatry 68, 320–328 (2010).
Peschansky, V.J. et al. The effect of variation in expression of the candidate dyslexia susceptibility gene homolog Kiaa0319 on neuronal migration and dendritic morphology in the rat. Cereb. Cortex 20, 884–897 (2010).
Wang, Y. et al. Dcdc2 knockout mice display exacerbated developmental disruptions following knockdown of doublecortin. Neuroscience 190, 398–408 (2011).
Goswami, U. A temporal sampling framework for developmental dyslexia. Trends Cogn. Sci. 15, 3–10 (2011).
Ramus, F. & Szenkovits, G. What phonological deficit? Q. J. Exp. Psychol. (Hove) 61, 129–141 (2008).
Ziegler, J.C. et al. Speech-perception-in-noise deficits in dyslexia. Dev. Sci. 12, 732–745 (2009).
Lehongre, K. et al. Altered low-gamma sampling in auditory cortex accounts for the three main facets of dyslexia. Neuron 72, 1080–1090 (2011).
Hickok, G. & Poeppel, D. The cortical organization of speech processing. Nat. Rev. Neurosci. 8, 393–402 (2007).
Holcombe, A.O. Seeing slow and seeing fast: two limits on perception. Trends Cogn. Sci. 13, 216–221 (2009).
Eliades, S.J. & Wang, X. Neural substrates of vocalization feedback monitoring in primate auditory cortex. Nature 453, 1102–1106 (2008).
Chandrasekaran, C. et al. Monkeys and humans share a common computation for face/voice integration. PLoS Comput. Biol. 7, e1002165 (2011).
Schroeder, C.E. et al. Dynamics of active sensing and perceptual selection. Curr. Opin. Neurobiol. 20, 172–176 (2010).
Oberlaender, M. et al. Cell type-specific three-dimensional structure of thalamocortical circuits in a column of rat vibrissal cortex. Cereb. Cortex doi:10.1093/cercor/bhr317 (16 November 2011).
Chi, T., Ru, P. & Shamma, S.A. Multiresolution spectrotemporal analysis of complex sounds. J. Acoust. Soc. Am. 118, 887–906 (2005).
Acknowledgements
We are deeply grateful to C. Liegeois-Chauvel for providing stereotactic EEG data and C.-G. Bénar for related methodological support. In A.-L.G.'s team we thank Y. Beigneux, B. Morillon and L. Arnal, who analyzed these data; A. Hyafil, C. Kapdebon and L. Fontolan, who carried out the computational modeling work; and K. Lehongre and D. Roussillon, who conducted the experiments in human subjects. In D.P.'s team, we thank H. Luo, M. Howard and G. Cogan for pioneering work and many discussions of these issues. We also thank our colleagues O. Ghitza, S. Greenberg, B. Gutkin, V. Wyart, C. Lorenzi, F. Ramus and C. Schroeder for motivating and discussing various aspects of this research. This work is supported by the Centre National de la Recherche Scientifique of France and the European Research Council (A.-L.G.), and US National Institutes of Health grant 2R01 DC05660 (D.P.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Giraud, AL., Poeppel, D. Cortical oscillations and speech processing: emerging computational principles and operations. Nat Neurosci 15, 511–517 (2012). https://doi.org/10.1038/nn.3063
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3063
This article is cited by
-
Auditory-motor synchronization and perception suggest partially distinct time scales in speech and music
Communications Psychology (2024)
-
Developmental delays in cortical auditory temporal processing in a mouse model of Fragile X syndrome
Journal of Neurodevelopmental Disorders (2023)
-
Hearing as adaptive cascaded envelope interpolation
Communications Biology (2023)
-
Biological principles for music and mental health
Translational Psychiatry (2023)
-
High-resolution neural recordings improve the accuracy of speech decoding
Nature Communications (2023)