Abstract
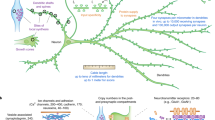
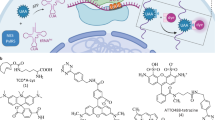
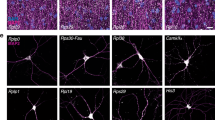
Protein translation has been implicated in different forms of synaptic plasticity, but direct in situ visualization of new proteins is limited to one or two proteins at a time. Here we describe a metabolic labeling approach based on incorporation of noncanonical amino acids into proteins followed by chemoselective fluorescence tagging by means of 'click chemistry'. After a brief incubation with azidohomoalanine or homopropargylglycine, a robust fluorescent signal was detected in somata and dendrites. Pulse-chase application of azidohomoalanine and homopropargylglycine allowed visualization of proteins synthesized in two sequential time periods. This technique can be used to detect changes in protein synthesis and to evaluate the fate of proteins synthesized in different cellular compartments. Moreover, using strain-promoted cycloaddition, we explored the dynamics of newly synthesized membrane proteins using single-particle tracking and quantum dots. The newly synthesized proteins showed a broad range of diffusive behaviors, as would be expected for a pool of labeled proteins that is heterogeneous.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Sutton, M.A. & Schuman, E.M. Dendritic protein synthesis, synaptic plasticity, and memory. Cell 127, 49–58 (2006).
Nguyen, P.V., Abel, T. & Kandel, E.R. Requirement of a critical period of transcription for induction of a late phase of LTP. Science 265, 1104–1107 (1994).
Bito, H., Deisseroth, K. & Tsien, R.W. CREB phosphorylation and dephosphorylation: a Ca2+- and stimulus duration-dependent switch for hippocampal gene expression. Cell 87, 1203–1214 (1996).
Casadio, A. et al. A transient, neuron-wide form of CREB-mediated long-term facilitation can be stabilized at specific synapses by local protein synthesis. Cell 99, 221–237 (1999).
Kang, H. & Schuman, E.M. A requirement for local protein synthesis in neurotrophin-induced hippocampal synaptic plasticity. Science 273, 1402–1406 (1996).
Martin, K.C. et al. Synapse-specific, long-term facilitation of Aplysia sensory to motor synapses: a function for local protein synthesis in memory storage. Cell 91, 927–938 (1997).
Weiler, I.J., Wang, X. & Greenough, W.T. Synapse-activated protein synthesis as a possible mechanism of plastic neural change. Prog. Brain Res. 100, 189–194 (1994).
Steward, O. mRNA localization in neurons: a multipurpose mechanism? Neuron 18, 9–12 (1997).
Liao, L. et al. BDNF induces widespread changes in synaptic protein content and up-regulates components of the translation machinery: an analysis using high-throughput proteomics. J. Proteome Res. 6, 1059–1071 (2007).
Manadas, B. et al. BDNF-induced changes in the expression of the translation machinery in hippocampal neurons: protein levels and dendritic mRNA. J. Proteome Res. 8, 4536–4552 (2009).
Takei, N. et al. Brain-derived neurotrophic factor induces mammalian target of rapamycin-dependent local activation of translation machinery and protein synthesis in neuronal dendrites. J. Neurosci. 24, 9760–9769 (2004).
Aakalu, G., Smith, W.B., Nguyen, N., Jiang, C. & Schuman, E.M. Dynamic visualization of local protein synthesis in hippocampal neurons. Neuron 30, 489–502 (2001).
Macchi, P. et al. A GFP-based system to uncouple mRNA transport from translation in a single living neuron. Mol. Biol. Cell 14, 1570–1582 (2003).
Wang, D.O. et al. Synapse- and stimulus-specific local translation during long-term neuronal plasticity. Science 324, 1536–1540 (2009).
Lin, M.Z., Glenn, J.S. & Tsien, R.Y. A drug-controllable tag for visualizing newly synthesized proteins in cells and whole animals. Proc. Natl. Acad. Sci. USA 105, 7744–7749 (2008).
Kiick, K.L., Saxon, E., Tirrell, D.A. & Bertozzi, C.R. Incorporation of azides into recombinant proteins for chemoselective modification by the Staudinger ligation. Proc. Natl. Acad. Sci. USA 99, 19–24 (2002).
Link, A.J., Mock, M.L. & Tirrell, D.A. Non-canonical amino acids in protein engineering. Curr. Opin. Biotechnol. 14, 603–609 (2003).
Link, A.J. & Tirrell, D.A. Cell surface labeling of Escherichia coli via copper(I)-catalyzed [3+2] cycloaddition. J. Am. Chem. Soc. 125, 11164–11165 (2003).
Zhang, Z. et al. A new strategy for the site-specific modification of proteins in vivo. Biochemistry 42, 6735–6746 (2003).
Beatty, K.E. & Tirrell, D.A. Two-color labeling of temporally defined protein populations in mammalian cells. Bioorg. Med. Chem. Lett. 18, 5995–5999 (2008).
Dieterich, D.C. et al. Labeling, detection and identification of newly synthesized proteomes with bioorthogonal non-canonical amino-acid tagging. Nat. Protoc. 2, 532–540 (2007).
Dieterich, D.C., Link, A.J., Graumann, J., Tirrell, D.A. & Schuman, E.M. Selective identification of newly synthesized proteins in mammalian cells using bioorthogonal noncanonical amino acid tagging (BONCAT). Proc. Natl. Acad. Sci. USA 103, 9482–9487 (2006).
Rostovtsev, V.V., Green, L.G., Fokin, V.V. & Sharpless, K.B. A stepwise Huisgen cycloaddition process: copper(I)-catalyzed regioselective “ligation” of azides and terminal alkynes. Angew. Chem. Int. Edn. Engl. 41, 2596–2599 (2002).
Whitaker, J.E., Haugland, R.P., Ryan, D., Hewitt, P.C. & Prendergast, F.G. Fluorescent rhodol derivatives: versatile, photostable labels and tracers. Anal. Biochem. 207, 267–279 (1992).
Lewin, G.R. & Barde, Y.A. Physiology of the neurotrophins. Annu. Rev. Neurosci. 19, 289–317 (1996).
Alcor, D., Gouzer, G. & Triller, A. Single-particle tracking methods for the study of membrane receptors dynamics. Eur. J. Neurosci. 30, 987–997 (2009).
Baskin, J.M. et al. Copper-free click chemistry for dynamic in vivo imaging. Proc. Natl. Acad. Sci. USA 104, 16793–16797 (2007).
Dahan, M. et al. Diffusion dynamics of glycine receptors revealed by single-quantum dot tracking. Science 302, 442–445 (2003).
Renner, M., Choquet, D. & Triller, A. Control of the postsynaptic membrane viscosity. J. Neurosci. 29, 2926–2937 (2009).
Sergé, A., Fourgeaud, L., Hemar, A. & Choquet, D. Receptor activation and Homer differentially control the lateral mobility of metabotropic glutamate receptor 5 in the neuronal membrane. J. Neurosci. 22, 3910–3920 (2002).
Renner, M. et al. Deleterious effects of amyloid beta oligomers acting as an extracellular scaffold for mGluR5. Neuron (in the press).
Renner, M.L., Cognet, L., Lounis, B., Triller, A. & Choquet, D. The excitatory postsynaptic density is a size exclusion diffusion environment. Neuropharmacology 56, 30–36 (2009).
Ouyang, Y., Kantor, D., Harris, K.M., Schuman, E.M. & Kennedy, M.B. Visualization of the distribution of autophosphorylated calcium/calmodulin-dependent protein kinase II after tetanic stimulation in the CA1 area of the hippocampus. J. Neurosci. 17, 5416–5427 (1997).
Waung, M.W., Pfeiffer, B.E., Nosyreva, E.D., Ronesi, J.A. & Huber, K.M. Rapid translation of Arc/Arg3.1 selectively mediates mGluR-dependent LTD through persistent increases in AMPAR endocytosis rate. Neuron 59, 84–97 (2008).
Ouyang, Y., Rosenstein, A., Kreiman, G., Schuman, E.M. & Kennedy, M.B. Tetanic stimulation leads to increased accumulation of Ca2+/calmodulin-dependent protein kinase II via dendritic protein synthesis in hippocampal neurons. J. Neurosci. 19, 7823–7833 (1999).
Villareal, G., Li, Q., Cai, D. & Glanzman, D.L. The role of rapid, local, postsynaptic protein synthesis in learning-related synaptic facilitation in Aplysia. Curr. Biol. 17, 2073–2080 (2007).
Beatty, K.E. et al. Fluorescence visualization of newly synthesized proteins in mammalian cells. Angew. Chem. Int. Edn. Engl. 45, 7364–7367 (2006).
Agard, N.J., Prescher, J.A. & Bertozzi, C.R. A strain-promoted [3 + 2] azidealkyne cycloaddition for covalent modification of biomolecules in living systems. J. Am. Chem. Soc. 126, 15046–15047 (2004).
Taylor, A.M., Dieterich, D.C., Ito, H.T., Kim, S.A. & Schuman, E.M. Microfluidic local perfusion chambers for the visualization and manipulation of synapses. Neuron 66, 57–68 (2010).
Frey, U. & Morris, R.G. Synaptic tagging and long-term potentiation. Nature 385, 533–536 (1997).
Huang, T., McDonough, C.B. & Abel, T. Compartmentalized PKA signaling events are required for synaptic tagging and capture during hippocampal late-phase long-term potentiation. Eur. J. Cell Biol. 85, 635–642 (2006).
Sajikumar, S., Navakkode, S. & Frey, J.U. Identification of compartment- and process-specific molecules required for “synaptic tagging” during long-term potentiation and long-term depression in hippocampal CA1. J. Neurosci. 27, 5068–5080 (2007).
Ngo, J.T. et al. Cell-selective metabolic labeling of proteins. Nat. Chem. Biol. 5, 715–717 (2009).
Flexner, J.B., Flexner, L.B. & Stellar, E. Memory in mice as affected by intracerebral puromycin. Science 141, 57–59 (1963).
Agranoff, B.W. & Klinger, P.D. Puromycin effect on memory fixation in the goldfish. Science 146, 952–953 (1964).
Renner, M., Specht, C.G. & Triller, A. Molecular dynamics of postsynaptic receptors and scaffold proteins. Curr. Opin. Neurobiol. 18, 532–540 (2008).
Wang, Q. et al. Bioconjugation by copper(I)-catalyzed azide–alkyne [3 + 2] cycloaddition. J. Am. Chem. Soc. 125, 3192–3193 (2003).
Gogolla, N., Galimberti, I., DePaola, V. & Caroni, P. Long-term live imaging of neuronal circuits in organotypic hippocampal slice cultures. Nat. Protoc. 1, 1223–1226 (2006).
Bannai, H., Levi, S., Schweizer, C., Dahan, M. & Triller, A. Imaging the lateral diffusion of membrane molecules with quantum dots. Nat. Protoc. 1, 2628–2634 (2007).
Acknowledgements
We thank L. Chen for making beautiful cultured hippocampal neurons. We thank A.J. Link for discussions and help with the tag syntheses. We are grateful to O. Kobler for help with Imaris software. We are extremely grateful to both C. Bertozzi and J. Baskin for providing the difluorinated cyclooctyne-biotin and advising on its use. This work was supported by the German Academy for Natural Scientists Leopoldina (D.C.D.), the US National Institutes of Health (E.M.S. and D.A.T.), the Howard Hughes Medical Institute (E.M.S.), the Ministère de l'Enseignement Supérieur et de la Recherche (G.G.) and the Nationale de la Recherche MorphoSynDiff–INSERM (A.T.).
Author information
Authors and Affiliations
Contributions
D.C.D., J.J.L.H., G.G. and E.M.S. performed experiments; D.C.D., G.G., A.T. and E.M.S. designed experiments; D.C.D., J.J.L.H., I.Y.S., G.G. and E.M.S. analyzed data; D.C.D., G.G., A.T. and E.M.S. wrote the paper; J.T.N. and D.A.T. provided reagents.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 (PDF 11714 kb)
Rights and permissions
About this article
Cite this article
Dieterich, D., Hodas, J., Gouzer, G. et al. In situ visualization and dynamics of newly synthesized proteins in rat hippocampal neurons. Nat Neurosci 13, 897–905 (2010). https://doi.org/10.1038/nn.2580
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2580
This article is cited by
-
A mutant methionyl-tRNA synthetase-based toolkit to assess induced-mesenchymal stromal cell secretome in mixed-culture disease models
Stem Cell Research & Therapy (2023)
-
THRONCAT: metabolic labeling of newly synthesized proteins using a bioorthogonal threonine analog
Nature Communications (2023)
-
Rapid metabolic reprogramming mediated by the AMP-activated protein kinase during the lytic cycle of Toxoplasma gondii
Nature Communications (2023)
-
A novel splice variant of Elp3/Kat9 regulates mitochondrial tRNA modification and function
Scientific Reports (2022)
-
Metabolic labeling of secreted matrix to investigate cell–material interactions in tissue engineering and mechanobiology
Nature Protocols (2022)