Abstract
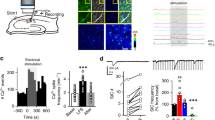
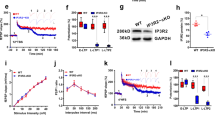
Astrocytes are critical participants in synapse development and function, but their role in synaptic plasticity is unclear. Eph receptors and their ephrin ligands have been suggested to regulate neuron-glia interactions, and EphA4-mediated ephrin reverse signaling is required for synaptic plasticity in the hippocampus. Here we show that long-term potentiation (LTP) at the CA3–CA1 synapse is modulated by EphA4 in the postsynaptic CA1 cell and by ephrin-A3, a ligand of EphA4 that is found in astrocytes. Lack of EphA4 increased the abundance of glial glutamate transporters, and ephrin-A3 modulated transporter currents in astrocytes. Pharmacological inhibition of glial glutamate transporters rescued the LTP defects in EphA4 (Epha4) and ephrin-A3 (Efna3) mutant mice. Transgenic overexpression of ephrin-A3 in astrocytes reduces glutamate transporter levels and produces focal dendritic swellings possibly caused by glutamate excitotoxicity. These results suggest that EphA4/ephrin-A3 signaling is a critical mechanism for astrocytes to regulate synaptic function and plasticity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Accession codes
References
Allen, N.J. & Barres, B.A. Signaling between glia and neurons: focus on synaptic plasticity. Curr. Opin. Neurobiol. 15, 542–548 (2005).
Barres, B.A. The mystery and magic of glia: a perspective on their roles in health and disease. Neuron 60, 430–440 (2008).
Halassa, M.M., Fellin, T. & Haydon, P.G. The tripartite synapse: roles for gliotransmission in health and disease. Trends Mol. Med. 13, 54–63 (2007).
Bergles, D.E. & Jahr, C.E. Glial contribution to glutamate uptake at Schaffer collateral-commissural synapses in the hippocampus. J. Neurosci. 18, 7709–7716 (1998).
Tzingounis, A.V. & Wadiche, J.I. Glutamate transporters: confining runaway excitation by shaping synaptic transmission. Nat. Rev. Neurosci. 8, 935–947 (2007).
Pita-Almenar, J.D., Collado, M.S., Colbert, C.M. & Eskin, A. Different mechanisms exist for the plasticity of glutamate reuptake during early long-term potentiation (LTP) and late LTP. J. Neurosci. 26, 10461–10471 (2006).
Genoud, C. et al. Plasticity of astrocytic coverage and glutamate transporter expression in adult mouse cortex. PLoS Biol. 4, e343 (2006).
Essmann, C.L. et al. Serine phosphorylation of ephrinB2 regulates trafficking of synaptic AMPA receptors. Nat. Neurosci. 11, 1035–1043 (2008).
Klein, R. Bidirectional modulation of synaptic functions by Eph/ephrin signaling. Nat. Neurosci. 12, 15–20 (2009).
Kayser, M.S., Nolt, M.J. & Dalva, M.B. EphB receptors couple dendritic filopodia motility to synapse formation. Neuron 59, 56–69 (2008).
Murai, K.K., Nguyen, L.N., Irie, F., Yamaguchi, Y. & Pasquale, E.B. Control of hippocampal dendritic spine morphology through ephrin-A3/EphA4 signaling. Nat. Neurosci. 6, 153–160 (2003).
Grunwald, I.C. et al. Hippocampal plasticity requires postsynaptic ephrinBs. Nat. Neurosci. 7, 33–40 (2004).
Grunwald, I.C. et al. Kinase-independent requirement of EphB2 receptors in hippocampal synaptic plasticity. Neuron 32, 1027–1040 (2001).
Henderson, J.T. et al. The receptor tyrosine kinase EphB2 regulates NMDA-dependent synaptic function. Neuron 32, 1041–1056 (2001).
Dalva, M.B. et al. EphB receptors interact with NMDA receptors and regulate excitatory synapse formation. Cell 103, 945–956 (2000).
Bouzioukh, F. et al. Tyrosine phosphorylation sites in ephrinB2 are required for hippocampal long-term potentiation but not long-term depression. J. Neurosci. 27, 11279–11288 (2007).
Kullander, K. et al. Kinase-dependent and kinase-independent functions of EphA4 receptors in major axon tract formation in vivo. Neuron 29, 73–84 (2001).
Tsien, J.Z. et al. Subregion- and cell type-restricted gene knockout in mouse brain. Cell 87, 1317–1326 (1996).
Carmona, M.A., Murai, K.K., Wang, L., Roberts, A.J. & Pasquale, E.B. Glial ephrin-A3 regulates hippocampal dendritic spine morphology and glutamate transport. Proc. Natl. Acad. Sci. USA 106, 12524–12529 (2009).
Tremblay, M.E. et al. Localization of EphA4 in axon terminals and dendritic spines of adult rat hippocampus. J. Comp. Neurol. 501, 691–702 (2007).
Albensi, B.C., Oliver, D.R., Toupin, J. & Odero, G. Electrical stimulation protocols for hippocampal synaptic plasticity and neuronal hyper-excitability: are they effective or relevant? Exp. Neurol. 204, 1–13 (2007).
Beart, P.M. & O'Shea, R.D. Transporters for L-glutamate: an update on their molecular pharmacology and pathological involvement. Br. J. Pharmacol. 150, 5–17 (2007).
Liu, G., Choi, S. & Tsien, R.W. Variability of neurotransmitter concentration and nonsaturation of postsynaptic AMPA receptors at synapses in hippocampal cultures and slices. Neuron 22, 395–409 (1999).
Oliet, S.H., Piet, R. & Poulain, D.A. Control of glutamate clearance and synaptic efficacy by glial coverage of neurons. Science 292, 923–926 (2001).
Tsukada, S., Iino, M., Takayasu, Y., Shimamoto, K. & Ozawa, S. Effects of a novel glutamate transporter blocker, (2S, 3S)-3-[3-[4-(trifluoromethyl)benzoylamino]benzyloxy]aspartate (TFB-TBOA), on activities of hippocampal neurons. Neuropharmacology 48, 479–491 (2005).
Nolte, C. et al. GFAP promoter-controlled EGFP-expressing transgenic mice: a tool to visualize astrocytes and astrogliosis in living brain tissue. Glia 33, 72–86 (2001).
Rothstein, J.D. et al. Knockout of glutamate transporters reveals a major role for astroglial transport in excitotoxicity and clearance of glutamate. Neuron 16, 675–686 (1996).
Tanaka, K. et al. Epilepsy and exacerbation of brain injury in mice lacking the glutamate transporter GLT-1. Science 276, 1699–1702 (1997).
Watanabe, T. et al. Amygdala-kindled and pentylenetetrazole-induced seizures in glutamate transporter GLAST-deficient mice. Brain Res. 845, 92–96 (1999).
Greenwood, S.M. & Connolly, C.N. Dendritic and mitochondrial changes during glutamate excitotoxicity. Neuropharmacology 53, 891–898 (2007).
Feng, G. et al. Imaging neuronal subsets in transgenic mice expressing multiple spectral variants of GFP. Neuron 28, 41–51 (2000).
Hasbani, M.J., Schlief, M.L., Fisher, D.A. & Goldberg, M.P. Dendritic spines lost during glutamate receptor activation reemerge at original sites of synaptic contact. J. Neurosci. 21, 2393–2403 (2001).
Lu, Y.M. et al. Mice lacking metabotropic glutamate receptor 5 show impaired learning and reduced CA1 long-term potentiation (LTP) but normal CA3 LTP. J. Neurosci. 17, 5196–5205 (1997).
Mulholland, P.J. et al. Glutamate transporters regulate extrasynaptic NMDA receptor modulation of Kv2.1 potassium channels. J. Neurosci. 28, 8801–8809 (2008).
Wadiche, J.I. & Jahr, C.E. Patterned expression of Purkinje cell glutamate transporters controls synaptic plasticity. Nat. Neurosci. 8, 1329–1334 (2005).
Nikkuni, O., Takayasu, Y., Iino, M., Tanaka, K. & Ozawa, S. Facilitated activation of metabotropic glutamate receptors in cerebellar Purkinje cells in glutamate transporter EAAT4-deficient mice. Neurosci. Res. 59, 296–303 (2007).
Levenson, J. et al. Long-term potentiation and contextual fear conditioning increase neuronal glutamate uptake. Nat. Neurosci. 5, 155–161 (2002).
Nishida, H. & Okabe, S. Direct astrocytic contacts regulate local maturation of dendritic spines. J. Neurosci. 27, 331–340 (2007).
Nestor, M.W., Mok, L.P., Tulapurkar, M.E. & Thompson, S.M. Plasticity of neuron-glial interactions mediated by astrocytic EphARs. J. Neurosci. 27, 12817–12828 (2007).
Diamond, J.S. & Jahr, C.E. Synaptically released glutamate does not overwhelm transporters on hippocampal astrocytes during high-frequency stimulation. J. Neurophysiol. 83, 2835–2843 (2000).
Lehre, K.P. & Rusakov, D.A. Asymmetry of glia near central synapses favors presynaptically directed glutamate escape. Biophys. J. 83, 125–134 (2002).
Morris, R.G. et al. Elements of a neurobiological theory of the hippocampus: the role of activity-dependent synaptic plasticity in memory. Philos. Trans. R. Soc. Lond. B 358, 773–786 (2003).
Tsuda, H. et al. The amyotrophic lateral sclerosis 8 protein VAPB is cleaved, secreted, and acts as a ligand for Eph receptors. Cell 133, 963–977 (2008).
Weiergraber, M. et al. Altered seizure susceptibility in mice lacking the Ca(v)2.3 E-type Ca2+ channel. Epilepsia 47, 839–850 (2006).
Luscher, C., Malenka, R.C. & Nicoll, R.A. Monitoring glutamate release during LTP with glial transporter currents. Neuron 21, 435–441 (1998).
Egea, J. et al. Regulation of EphA 4 kinase activity is required for a subset of axon guidance decisions suggesting a key role for receptor clustering in Eph function. Neuron 47, 515–528 (2005).
Haugeto, O. et al. Brain glutamate transporter proteins form homomultimers. J. Biol. Chem. 271, 27715–27722 (1996).
Kramer, E.R. et al. Absence of Ret signaling in mice causes progressive and late degeneration of the nigrostriatal system. PLoS Biol. 5, e39 (2007).
Stoppini, L., Buchs, P.A. & Muller, D. A simple method for organotypic cultures of nervous tissue. J. Neurosci. Methods 37, 173–182 (1991).
Livak, K.J. & Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-delta delta C(T)) method. Methods 25, 402–408 (2001).
Acknowledgements
We thank M. Bösl and the transgenic core facility for generating transgenic mice; E. Kandel (Columbia University) and F. Kirchhoff (Max Planck Institute of Experimental Medicine, Göttingen) for transgenic mice; M. Klein and O. Gökce for technical help; K. Deininger, C. Erlacher, V. Staiger, V. Stein and M. Traut for scientific input and suggestions; M. Korte, I. Kadow, V. Stein, J. Egea and R. Fonseca for critical comments on the manuscript. S.P. was supported by a postdoctoral fellowship from Fundação para a Ciência e Tecnologia of Portugal, co-funded by Programa Operacional Ciência e Inovação 2010 and Fundo Social Europeu. M.A.C. was supported by a fellowship from Fundación Española para la Ciencia y la Tecnología. This work was in part supported by grants from the European Union (Endotrack), the Deutsche Forschungsgemeinschaft (SPP1172) and the Max-Planck Society (all to R.K.), the Wellcome Trust and the Biotechnology and Biological Sciences Research Council, UK (R.S.), the German National Genome Research Network (NGF N grant 01GR0430) (T.K.), and US National Institutes of Health grant HD025938 (E.B.P.).
Author information
Authors and Affiliations
Contributions
A.F. designed, performed, analyzed most of the electrophysiology experiments and co-wrote the manuscript. S.P. designed, performed, analyzed the biochemical and quantitative anatomical studies and co-wrote the manuscript. S.D.H. and C.R.R. designed, performed and analyzed the astrocyte patch clamp recordings. M.A.C. and E.B.P. provided the Efna3−/− model, gave advice and aided in the interpretation of data. L.B., B.F. and T.K. performed and analyzed the induced seizure experiments. L.G. performed biochemical studies. Y.R. and R.S. provided the CA3-Cre mouse. K.K. provided Epha4lx/+ ES cells. R.K. supervised the project, designed experiments and co-wrote the manuscript. The two first authors, who contributed equally, are listed in alphabetical order.
Corresponding authors
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 (PDF 1182 kb)
Rights and permissions
About this article
Cite this article
Filosa, A., Paixão, S., Honsek, S. et al. Neuron-glia communication via EphA4/ephrin-A3 modulates LTP through glial glutamate transport. Nat Neurosci 12, 1285–1292 (2009). https://doi.org/10.1038/nn.2394
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2394
This article is cited by
-
Neuronal DSCAM regulates the peri-synaptic localization of GLAST in Bergmann glia for functional synapse formation
Nature Communications (2024)
-
Probing pathways by which rhynchophylline modifies sleep using spatial transcriptomics
Biology Direct (2023)
-
Role of spinal astrocytes through the perisynaptic astrocytic process in pathological pain
Molecular Brain (2023)
-
EphrinA4 mimetic peptide impairs fear conditioning memory reconsolidation in lateral amygdala
Scientific Reports (2022)
-
EphA4 Obstructs Spinal Cord Neuron Regeneration by Promoting Excessive Activation of Astrocytes
Cellular and Molecular Neurobiology (2022)