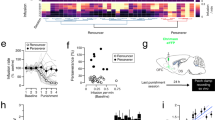
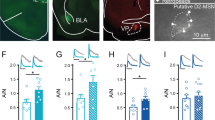
Abstract
Addictive drugs hijack mechanisms of learning and memory that normally underlie reinforcement of natural rewards and induce synaptic plasticity of glutamatergic transmission in the mesolimbic dopamine (DA) system. In the ventral tegmental area (VTA), a single exposure to cocaine efficiently triggers NMDA receptor–dependent synaptic plasticity in DA neurons, whereas plasticity in the nucleus accumbens (NAc) occurs only after repeated injections. Whether these two forms of plasticity are independent or hierarchically organized remains unknown. We combined ex vivo electrophysiology in acute brain slices with behavioral assays modeling drug relapse in mice and found that the duration of the cocaine-evoked synaptic plasticity in the VTA is gated by mGluR1. Overriding mGluR1 in vivo made the potentiation in the VTA persistent. This led to synaptic plasticity in the NAc, which contributes to cocaine-seeking behavior after protracted withdrawal. Impaired mGluR1 function in vulnerable individuals could represent a first step in the recruitment of the neuronal network that underlies drug addiction.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Kalivas, P.W. Glutamate systems in cocaine addiction. Curr. Opin. Pharmacol. 4, 23–29 (2004).
Kauer, J.A. & Malenka, R.C. Synaptic plasticity and addiction. Nat. Rev. Neurosci. 8, 844–858 (2007).
Thomas, M.J., Kalivas, P.W. & Shaham, Y. Neuroplasticity in the mesolimbic dopamine system and cocaine addiction. Br. J. Pharmacol. 154, 327–342 (2008).
Ungless, M.A., Whistler, J.L., Malenka, R.C. & Bonci, A. Single cocaine exposure in vivo induces long-term potentiation in dopamine neurons. Nature 411, 583–587 (2001).
Bellone, C. & Lüscher, C. Cocaine triggered AMPA receptor redistribution is reversed in vivo by mGluR-dependent long-term depression. Nat. Neurosci. 9, 636–641 (2006).
Argilli, E., Sibley, D.R., Malenka, R.C., England, P.M. & Bonci, A. Mechanism and time course of cocaine-induced long-term potentiation in the ventral tegmental area. J. Neurosci. 28, 9092–9100 (2008).
Saal, D., Dong, Y., Bonci, A. & Malenka, R.C. Drugs of abuse and stress trigger a common synaptic adaptation in dopamine neurons. Neuron 37, 577–582 (2003).
Borgland, S.L., Malenka, R.C. & Bonci, A. Acute and chronic cocaine-induced potentiation of synaptic strength in the ventral tegmental area: electrophysiological and behavioral correlates in individual rats. J. Neurosci. 24, 7482–7490 (2004).
Chen, B.T. et al. Cocaine but not natural reward self-administration nor passive cocaine infusion produces persistent LTP in the VTA. Neuron 59, 288–297 (2008).
Kourrich, S., Rothwell, P.E., Klug, J.R. & Thomas, M.J. Cocaine experience controls bidirectional synaptic plasticity in the nucleus accumbens. J. Neurosci. 27, 7921–7928 (2007).
Conrad, K.L. et al. Formation of accumbens GluR2-lacking AMPA receptors mediates incubation of cocaine craving. Nature 454, 118–121 (2008).
Churchill, L., Swanson, C.J., Urbina, M. & Kalivas, P.W. Repeated cocaine alters glutamate receptor subunit levels in the nucleus accumbens and ventral tegmental area of rats that develop behavioral sensitization. J. Neurochem. 72, 2397–2403 (1999).
Boudreau, A.C. & Wolf, M.E. Behavioral sensitization to cocaine is associated with increased AMPA receptor surface expression in the nucleus accumbens. J. Neurosci. 25, 9144–9151 (2005).
Grimm, J.W., Hope, B.T., Wise, R.A. & Shaham, Y. Neuroadaptation. Incubation of cocaine craving after withdrawal. Nature 412, 141–142 (2001).
Ping, A., Xi, J., Prasad, B.M., Wang, M.H. & Kruzich, P.J. Contributions of nucleus accumbens core and shell GluR1 containing AMPA receptors in AMPA- and cocaine-primed reinstatement of cocaine-seeking behavior. Brain Res. 1215, 173–182 (2008).
Kessels, H.W. & Malinow, R. Synaptic AMPA receptor plasticity and behavior. Neuron 61, 340–350 (2009).
Mameli, M., Balland, B., Lujan, R. & Lüscher, C. Rapid synthesis and synaptic insertion of GluR2 for mGluR-LTD in the ventral tegmental area. Science 317, 530–533 (2007).
Mao, L. et al. The scaffold protein Homer1b/c links metabotropic glutamate receptor 5 to extracellular signal–regulated protein kinase cascades in neurons. J. Neurosci. 25, 2741–2752 (2005).
Ronesi, J.A. & Huber, K.M. Homer interactions are necessary for metabotropic glutamate receptor–induced long-term depression and translational activation. J. Neurosci. 28, 543–547 (2008).
Moroni, F. et al. Pharmacological characterization of 1-aminoindan-1,5-dicarboxylic acid, a potent mGluR1 antagonist. J. Pharmacol. Exp. Ther. 281, 721–729 (1997).
Thomas, M.J., Beurrier, C., Bonci, A. & Malenka, R.C. Long-term depression in the nucleus accumbens: a neural correlate of behavioral sensitization to cocaine. Nat. Neurosci. 4, 1217–1223 (2001).
Engblom, D. et al. Glutamate receptors on dopamine neurons control the persistence of cocaine seeking. Neuron 59, 497–508 (2008).
Anderson, S.M. et al. CaMKII: a biochemical bridge linking accumbens dopamine and glutamate systems in cocaine seeking. Nat. Neurosci. 11, 344–353 (2008).
Haber, S.N., Fudge, J.L. & McFarland, N.R. Striatonigrostriatal pathways in primates form an ascending spiral from the shell to the dorsolateral striatum. J. Neurosci. 20, 2369–2382 (2000).
Ikemoto, S. Dopamine reward circuitry: two projection systems from the ventral midbrain to the nucleus accumbens-olfactory tubercle complex. Brain Res. Rev. 56, 27–78 (2007).
Everitt, B.J. & Robbins, T.W. Neural systems of reinforcement for drug addiction: from actions to habits to compulsion. Nat. Neurosci. 8, 1481–1489 (2005).
Lüscher, C. & Bellone, C. Cocaine-evoked synaptic plasticity: a key to addiction? Nat. Neurosci. 11, 737–738 (2008).
Zweifel, L.S., Argilli, E., Bonci, A. & Plamiter, R.D. Role of NMDA receptors in dopamine neurons for plasticity and addictive behaviors. Neuron 59, 486–496 (2008).
Vezina, P. & Queen, A.L. Induction of locomotor sensitization by amphetamine requires the activation of NMDA receptors in the rat ventral tegmental area. Psychopharmacology (Berl.) 151, 184–191 (2000).
Harris, G.C., Wimmer, M., Byrne, R. & Aston-Jones, G. Glutamate-associated plasticity in the ventral tegmental area is necessary for conditioning environmental stimuli with morphine. Neuroscience 129, 841–847 (2004).
Dong, Y. et al. Cocaine-induced potentiation of synaptic strength in dopamine neurons: behavioral correlates in GluRA−/− mice. Proc. Natl. Acad. Sci. USA 101, 14282–14287 (2004).
Mead, A.N., Brown, G., Le Merrer, J. & Stephens, D.N. Effects of deletion of gria1 or gria2 genes encoding glutamatergic AMPA-receptor subunits on place preference conditioning in mice. Psychopharmacology (Berl.) 179, 164–171 (2005).
Carlezon, W.A. Jr & Nestler, E.J. Elevated levels of GluR1 in the midbrain: a trigger for sensitization to drugs of abuse? Trends Neurosci. 25, 610–615 (2002).
Lammel, S. et al. Unique properties of mesoprefrontal neurons within a dual mesocorticolimbic dopamine system. Neuron 57, 760–773 (2008).
Zhao, S. et al. Generation of embryonic stem cells and transgenic mice expressing green fluorescence protein in midbrain dopaminergic neurons. Eur. J. Neurosci. 19, 1133–1140 (2004).
Acknowledgements
We thank members of the Lüscher laboratory, as well as B.J. Everitt, M. Frerking, A. Lüthi, M. Serafin, M. Carta and C.F. Valenzuela for helpful discussions and suggestions regarding the manuscript. R. Sprengel (Max Planck Institute Heidelberg) generated the NR12loxP/loxP mouse line and G. Schütz (Deutsches Krebsforschungszentrum Heidelberg) provided the NR1DAT-CreERT2 mouse line. C.L. is supported by grants from the Swiss National Science Foundation and the Swiss initiative in system biology (SystemsX: neurochoice). R.S. is supported by Nationales Genomforschungsnetz and Deutsche Forschungsgemeinschaft.
Author information
Authors and Affiliations
Contributions
M.M. carried out the electrophysiology experiments. B.H. performed the behavioral experiments. D.E. generated the mutant mice. J.R.P. bred the mice for the behavioral experiments and injected them with tamoxifen. C.L. designed the study with M.M. and C.C. and wrote the manuscript with the help of M.M. and R.S.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–4 (PDF 1831 kb)
Rights and permissions
About this article
Cite this article
Mameli, M., Halbout, B., Creton, C. et al. Cocaine-evoked synaptic plasticity: persistence in the VTA triggers adaptations in the NAc. Nat Neurosci 12, 1036–1041 (2009). https://doi.org/10.1038/nn.2367
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2367
This article is cited by
-
Activation of RMTg projections to the VTA reverse cocaine-induced molecular adaptation in the reward system
Translational Psychiatry (2024)
-
Cocaine induces input and cell-type-specific synaptic plasticity in ventral pallidum-projecting nucleus accumbens medium spiny neurons
Neuropsychopharmacology (2022)
-
A short burst of reward curbs the addictiveness of ketamine
Nature (2022)
-
Dual action of ketamine confines addiction liability
Nature (2022)
-
Ca2+-permeable AMPA receptors set the threshold for retrieval of drug memories
Molecular Psychiatry (2022)