Abstract
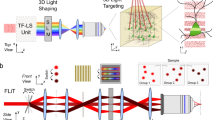
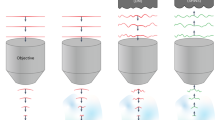
Stimulation of light-sensitive chemical probes has become a powerful tool for the study of dynamic signaling processes in living tissue. Classically, this approach has been constrained by limitations of lens-based and point-scanning illumination systems. Here we describe a microscope configuration that incorporates a nematic liquid-crystal spatial light modulator to generate holographic patterns of illumination. This microscope can produce illumination spots of variable size and number, and in patterns shaped to precisely match user-defined elements in a specimen. Using holographic illumination to photolyze caged glutamate in brain slices, we show that shaped excitation on segments of neuronal dendrites and simultaneous, multispot excitation of different dendrites enables precise spatial and rapid temporal control of glutamate receptor activation. By allowing the excitation volume shape to be tailored precisely, the holographic microscope provides an extremely flexible method for activation of various photosensitive proteins and small molecules.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Tang, C.M. Photolysis of caged neutrotransmitters; theory and procedures for light delivery. in Current Protocols in Neuroscience Unit 6.21 (John Wiley & Sons, Hoboken, NJ, 2006).
DiGregorio, D.A., Rothman, J.S., Nielsen, T.A. & Silver, R.A. Desensitization properties of AMPA receptors at the cerebellar mossy fiber granule cell synapse. J. Neurosci. 27, 8344–8357 (2007).
Matsuzaki, M. et al. Dendritic spine geometry is critical for AMPA receptor expression in hippocampal CA1 pyramidal neurons. Nat. Neurosci. 4, 1086–1092 (2001).
Carter, A.G. & Sabatini, B.L. State-dependent calcium signaling in dendritic spines of striatal medium spiny neurons. Neuron 44, 483–493 (2004).
Shepherd, G.M. & Svoboda, K. Laminar and columnar organization of ascending excitatory projections to layer 2/3 pyramidal neurons in rat barrel cortex. J. Neurosci. 25, 5670–5679 (2005).
Gasparini, S. & Magee, J.C. State-dependent dendritic computation in hippocampal CA1 pyramidal neurons. J. Neurosci. 26, 2088–2100 (2006).
Shoham, S., O'Connor, D.H., Sarkisov, D.V. & Wang, S.S. Rapid neurotransmitter uncaging in spatially defined patterns. Nat. Methods 2, 837–843 (2005).
Judkewitz, B., Roth, A. & Hausser, M. Dendritic enlightenment: using patterned two-photon uncaging to reveal the secrets of the brain's smallest dendrites. Neuron 50, 180–183 (2006).
Curtis, J.E., Koss, B.A. & Grier, D.G. Dynamic holographic optical tweezers. Opt. Commun. 207, 169–175 (2002).
Rodrigo, P.J., Daria, V.R. & Glückstad, J. Four-dimensional optical manipulation of colloidal particles. Appl. Phys. Lett. 86, 074103 (2005).
Leach, J. et al. 3D manipulation of particles into crystal structures using holographic optical tweezers. Opt. Express 12, 220–226 (2004).
Melville, H. et al. Optical trapping of three-dimensional structures using dynamic holograms. Opt. Express 11, 3562–3567 (2003).
Emiliani, V. et al. Wave front engineering for microscopy of living cells. Opt. Express 13, 1395–1405 (2005).
Wulff, K.D. et al. Aberration correction in holographic optical tweezers. Opt. Express 14, 4169–4174 (2006).
Cojoc, D., Di Fabrizio, E., Businaro, L. & Cabrini, S. Spherical-based approach to design diffractive optical elements. Proc. Soc. Photo. Opt. Instrum. Eng. 5227, 123–131 (2003).
Wyrowski, F. & Bryngdahl, O. Iterative Fourier-transform algorithm applied to computer holography. J. Opt. Soc. Am. A 5, 1058 (1988).
Goodman, J.W. Introduction to Fourier Optics (McGraw-Hill, New York, 1968).
Otis, T.S., Wu, Y.C. & Trussell, L.O. Delayed clearance of transmitter and the role of glutamate transporters at synapses with multiple release sites. J. Neurosci. 16, 1634–1644 (1996).
Carter, A.G. & Regehr, W.G. Prolonged synaptic currents and glutamate spillover at the parallel fiber to stellate cell synapse. J. Neurosci. 20, 4423–4434 (2000).
Tanaka, J. et al. Number and density of AMPA receptors in single synapses in immature cerebellum. J. Neurosci. 25, 799–807 (2005).
Megias, M., Emri, Z., Freund, T.F. & Gulyas, A.I. Total number and distribution of inhibitory and excitatory synapses on hippocampal CA1 pyramidal cells. Neuroscience 102, 527–540 (2001).
Andrasfalvy, B.K. & Magee, J.C. Distance-dependent increase in AMPA receptor number in the dendrites of adult hippocampal CA1 pyramidal neurons. J. Neurosci. 21, 9151–9159 (2001).
Silver, R.A., Cull-Candy, S.G. & Takahashi, T. Non-NMDA glutamate receptor occupancy and open probability at a rat cerebellar synapse with single and multiple release sites. J. Physiol. (Lond.) 494, 231–250 (1996).
DiGregorio, D.A., Nusser, Z. & Silver, R.A. Spillover of glutamate onto synaptic AMPA receptors enhances fast transmission at a cerebellar synapse. Neuron 35, 521–533 (2002).
Cathala, L., Holderith, N.B., Nusser, Z., DiGregorio, D.A. & Cull-Candy, S.G. Changes in synaptic structure underlie the developmental speeding of AMPA receptor-mediated EPSCs. Nat. Neurosci. 8, 1310–1318 (2005).
Smith, T.C., Wang, L.Y. & Howe, J.R. Heterogeneous conductance levels of native AMPA receptors. J. Neurosci. 20, 2073–2085 (2000).
Schmitz, C.H.J., Spatz, J.P. & Curtis, J.E. High-precision steering of multiple holographic optical traps. Opt. Express 13, 8678–8685 (2005).
Horneck, L.J. 128 × 128 Deformable Mirror Device. IEEE Trans. Electron. Dev. ED-30, 539–545 (1983).
Reddy, G.D. & Saggau, P. Fast three-dimensional laser scanning scheme using acousto-optic deflectors. J. Biomed. Opt. 10, 064038 (2005).
Gobel, W., Kampa, B.M. & Helmchen, F. Imaging cellular network dynamics in three dimensions using fast 3D laser scanning. Nat. Methods 4, 73–79 (2007).
Morrison, J., Wan, P., Corrie, J.E. & Papageorgiou, G. Mechanisms of photorelease of carboxylic acids from 1-acyl-7-nitroindolines in solutions of varying water content. Photochem. Photobiol. Sci. 1, 960–969 (2002).
Gervasi, N. et al. Dynamics of protein kinase A signaling at the membrane, in the cytosol, and in the nucleus of neurons in mouse brain slices. J. Neurosci. 27, 2744–2750 (2007).
Zhang, F. et al. Multimodal fast optical interrogation of neural circuitry. Nature 446, 633–639 (2007).
Szobota, S. et al. Remote control of neuronal activity with a light-gated glutamate receptor. Neuron 54, 535–545 (2007).
Gray, N.W., Weimer, R.M., Bureau, I. & Svoboda, K. Rapid redistribution of synaptic PSD-95 in the neocortex in vivo. PLoS Biol. 4, e370 (2006).
Acknowledgements
We thank D. Cojoc, E. Ferrari and E. di Fabrizio for collaborating in the development of the software; E. Papagiakoumou for helping to set up the double microscope system; D. Oron (Weizmann Institute of Science) for stimulating discussions and providing the coumarin samples; D. Ogden for stimulating discussions, comments on the manuscript and suggesting the use of 405-nm laser as a source; and A. Silver and A. Marty for critical reading of the manuscript. This work was supported by grants from the European Science Foundation and the Centre Nationale de la Recherche Scientifique through the European Young Investigators program (to V.E.), the European Commission FP6 Specific Targeted Project “Photolysis” LSHM-CT-2007-037765 (to V.E., S.C. and D.A.D.), Fondation Fyssen, the Actions Thématiques et Incitatives sur Programme Jeune Chercheurs, Federation pour la Recherche sur le Cerveau (to D.A.D.), Foundation Bettencourt-Schueller (to V.E. and S.C.), US National Institutes of Health grant NS49238 (to T.S.O.), the Network of European Science Institutes (to D.A.D. and V.E.) and the Alexander von Humboldt Foundation (to C.L.).
Author information
Authors and Affiliations
Contributions
C.L. set up the holographic optical microscope and performed the theoretical simulation for axial beam propagation; C.L., T.S.O. and D.A.D. performed and analyzed the cerebellar brain slice experiments; C.L. and S.C. performed and analyzed the hippocampal brain slice experiments; T.S.O., S.C. and D.A.D. contributed to writing the manuscript. V.D. developed the software. V.E. conceived and supervised the project, and prepared the manuscript.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Notes 1–3, Supplementary Methods (PDF 274 kb)
Rights and permissions
About this article
Cite this article
Lutz, C., Otis, T., DeSars, V. et al. Holographic photolysis of caged neurotransmitters. Nat Methods 5, 821–827 (2008). https://doi.org/10.1038/nmeth.1241
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth.1241
This article is cited by
-
Prominent in vivo influence of single interneurons in the developing barrel cortex
Nature Neuroscience (2023)
-
GABAB receptors constrain glutamate presynaptic release and postsynaptic actions in substantia gelatinosa of rat spinal cord
Brain Structure and Function (2022)
-
Optical vagus nerve modulation of heart and respiration via heart-injected retrograde AAV
Scientific Reports (2021)
-
BiPOLES is an optogenetic tool developed for bidirectional dual-color control of neurons
Nature Communications (2021)