Abstract
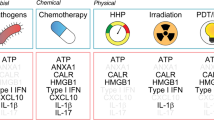
Anthracyclin-treated tumor cells are particularly effective in eliciting an anticancer immune response, whereas other DNA-damaging agents such as etoposide and mitomycin C do not induce immunogenic cell death. Here we show that anthracyclins induce the rapid, preapoptotic translocation of calreticulin (CRT) to the cell surface. Blockade or knockdown of CRT suppressed the phagocytosis of anthracyclin-treated tumor cells by dendritic cells and abolished their immunogenicity in mice. The anthracyclin-induced CRT translocation was mimicked by inhibition of the protein phosphatase 1/GADD34 complex. Administration of recombinant CRT or inhibitors of protein phosphatase 1/GADD34 restored the immunogenicity of cell death elicited by etoposide and mitomycin C, and enhanced their antitumor effects in vivo. These data identify CRT as a key feature determining anticancer immune responses and delineate a possible strategy for immunogenic chemotherapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Steinman, R.M. & Mellman, I. Immunotherapy bewitched, bothered, and bewildered no more. Science 305, 197–200 (2004).
Lake, R.A. & van der Most, R.G. A better way for a cancer cell to die. N. Engl. J. Med. 354, 2503–2504 (2006).
Zitvogel, L., Tesniere, A. & Kroemer, G. Cancer in spite of immunosurveillance: immunoselection and immunosubversion. Nat. Rev. Immunol. 6, 715–727 (2006).
Zitvogel, L., Casares, N., Pequignot, M., Albert, M.L. & Kroemer, G. The immune response against dying tumor cells. Adv. Immunol. 84, 131–179 (2004).
Bellamy, C.O., Malcomson, R.D., Harrison, D.J. & Wyllie, A.H. Cell death in health and disease: the biology and regulation of apoptosis. Semin. Cancer Biol. 6, 3–16 (1995).
Thompson, C.B. Apoptosis in the pathogenesis and treatment of disease. Science 267, 1456–1462 (1995).
Igney, F.H. & Krammer, P.H. Death and anti-death: tumour resistance to apoptosis. Nat. Rev. Cancer 2, 277–288 (2002).
Steinman, R.M., Turley, S., Mellman, I. & Inaba, K. The induction of tolerance by dendritic cells that have captured apoptotic cells. J. Exp. Med. 191, 411–416 (2000).
Liu, K. et al. Immune tolerance after delivery of dying cells to dendritic cells in situ. J. Exp. Med. 196, 1091–1097 (2002).
Kroemer, G. et al. Classification of cell death: recommendations of the Nomenclature Committee on Cell Death. Cell Death Differ. 12, 1463–1467 (2005).
Savill, J. & Fadok, V. Corpse clearance defines the meaning of cell death. Nature 407, 784–788 (2000).
Lauber, K., Blumenthal, S.G., Waibel, M. & Wesselborg, S. Clearance of apoptotic cells: getting rid of the corpses. Mol. Cell 14, 277–287 (2004).
Yoshida, H. et al. Phosphatidylserine-dependent engulfment by macrophages of nuclei from erythroid precursor cells. Nature 437, 754–758 (2005).
Gardai, S.J. et al. Cell-surface calreticulin initiates clearance of viable or apoptotic cells through trans-activation of LRP on the phagocyte. Cell 123, 321–334 (2005).
Henson, P.M. & Hume, D.A. Apoptotic cell removal in development and tissue homeostasis. Trends Immunol. 27, 244–250 (2006).
Hanayama, R. et al. Autoimmune disease and impaired uptake of apoptotic cells in MFG-E8-deficient mice. Science 304, 1147–1150 (2004).
Gaipl, U.S. et al. Inefficient clearance of dying cells and autoreactivity. Curr. Top. Microbiol. Immunol. 305, 161–176 (2006).
Vakkila, J. & Lotze, M.T. Inflammation and necrosis promote tumour growth. Nat. Rev. Immunol. 4, 641–648 (2004).
Casares, N. et al. Caspase-dependent immunogenicity of doxorubicin-induced tumor cell death. J. Exp. Med. 202, 1691–1701 (2005).
Blachere, N.E., Darnell, R.B. & Albert, M.L. Apoptotic cells deliver processed antigen to dendritic cells for cross-presentation. PLoS Biol. 3, e185 (2005).
Bedard, K., Szabo, E., Michalak, M. & Opas, M. Cellular functions of endoplasmic reticulum chaperones calreticulin, calnexin, and ERp57. Int. Rev. Cytol. 245, 91–121 (2005).
Ogden, C.A. et al. C1q and mannose binding lectin engagement of cell surface calreticulin and CD91 initiates macropinocytosis and uptake of apoptotic cells. J. Exp. Med. 194, 781–795 (2001).
Jung, S. et al. In vivo depletion of CD11c(+) dendritic cells abrogates priming of CD8(+) T cells by exogenous cell-associated antigens. Immunity 17, 211–220 (2002).
Zhang, K. & Kaufman, R.J. Signaling the unfolded protein response from the endoplasmic reticulum. J. Biol. Chem. 279, 25935–25938 (2004).
Gupta, V., Ogawa, A.K., Du, X., Houk, K.N. & Armstrong, R.W. A model for binding of structurally diverse natural product inhibitors of protein phosphatases PP1 and PP2A. J. Med. Chem. 40, 3199–3206 (1997).
Boyce, M. et al. A selective inhibitor of eIF2alpha dephosphorylation protects cells from ER stress. Science 307, 935–939 (2005).
Gelebart, P., Opas, M. & Michalak, M. Calreticulin, a Ca2+-binding chaperone of the endoplasmic reticulum. Int. J. Biochem. Cell Biol. 37, 260–266 (2005).
Williams, D.B. Beyond lectins: the calnexin/calreticulin chaperone system of the endoplasmic reticulum. J. Cell Sci. 119, 615–623 (2006).
Kim, S.J., Park, K.M., Kim, N. & Yeom, Y.I. Doxorubicin prevents endoplasmic reticulum stress-induced apoptosis. Biochem. Biophys. Res. Commun. 339, 463–468 (2006).
Hsieh, C.J. et al. Enhancement of vaccinia vaccine potency by linkage of tumor antigen gene to gene encoding calreticulin. Vaccine 22, 3993–4001 (2004).
Cheng, W.F. et al. Sindbis virus replicon particles encoding calreticulin linked to a tumor antigen generate long-term tumor-specific immunity. Cancer Gene Ther. 13, 873–885 (2006).
Basu, S. & Srivastava, P.K. Calreticulin, a peptide-binding chaperone of the endoplasmic reticulum, elicits tumor- and peptide-specific immunity. J. Exp. Med. 189, 797–802 (1999).
Basu, S., Binder, R.J., Suto, R., Anderson, K.M. & Srivastava, P.K. Necrotic but not apoptotic cell death releases heat shock proteins, which deliver a partial maturation signal to dendritic cells and activate the NF-kappa B pathway. Int. Immunol. 12, 1539–1546 (2000).
Zamzami, N. & Kroemer, G. Methods to measure membrane potential and permeability transition in the mitochondria during apoptosis. Methods Mol. Biol. 282, 103–116 (2004).
Culina, S., Lauvau, G., Gubler, B. & van Endert, P.M. Calreticulin promotes folding of functional human leukocyte antigen class I molecules in vitro. J. Biol. Chem. 279, 54210–54215 (2004).
Acknowledgements
We thank P. Aucouturier (Hôpital St. Antoine) for providing the transgenic mice and F. Coutant for help with the phagocytosis experiments. G.K. is supported by the Ligue Contre le Cancer (Equipe Labellisée), the European Commission (RIGHT), Cancéropôle Ile-de-France and Institut National Contre le Cancer (INCa). L.Z. is supported by the Ligue Contre le Cancer and INCa. M.P. is supported by a grant from the Associazione Italiana per la Ricerca sul Cancro and by a Ricerca Corrrente and Finalizzata grant from the Italian Ministry of Health. M.O. received a fellowship from the Lebanese Government (Centre National pour la Recherche Scientifique-L) and Centre National pour la Recherche Scientifique, A.T. from the Fondation pour la Recherche Médicale, L.A. from the Ligue Contre le Cancer, F.G. from INSERM and G.M. from the Association pour la Recherche Clinique et Translationnelle.
Author information
Authors and Affiliations
Contributions
M.O., A.T., F.G., G.M.F., L.A., J.-L. P., M.C., T.P., D.M., N.L. and F.C. performed the in vivo and in vitro experiments. G.M.F. performed mass spectroscopy. N.C. and P.v.E. provided essential reagents. M.P. conducted data analysis. L.Z. and G.K. conceived the study and wrote the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Incidence of tumors after inoculation of dying cells. The data show the actual frequency of tumor-free mice, for the experiment summarized in Fig. 1b. (PDF 475 kb)
Supplementary Fig. 2
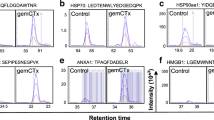
Mass spectroscopic identification and subcellular localization of CRT. (PDF 880 kb)
Supplementary Fig. 3
Kinetics of CRT exposure. CT26 cells were treated with mitoxantrone for the indicated period, followed by immunofluorescence staining with a CRT-specific antibody and cytofluorometric analysis. (PDF 246 kb)
Supplementary Fig. 4
CT26 cells were cultured for different periods with mitoxantrone or doxorubicin and then confronted with DC to measure their phagocytosis (a), as in Fig. 3a or injected into mice, one week before challenge with live cells (b). (PDF 195 kb)
Supplementary Fig. 5
Requirements for CRT-mediated tumor cell immunogenicity. (PDF 316 kb)
Supplementary Fig. 6
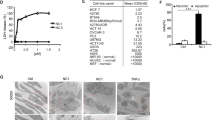
Cells were treated with mitoxantrone or inhibitors of PP1/GADD34, after pre-incubation for 1 h with the indicated inhibitors of protein synthesis (cycloheximide), RNA synthesis (actinomycin D), microtubuli (nocodazol), or the actin cytoskeleton (latrinculin A). (PDF 271 kb)
Supplementary Methods
Generation of bone marrow-derived dendritic cells (DC) (PDF 83 kb)
Rights and permissions
About this article
Cite this article
Obeid, M., Tesniere, A., Ghiringhelli, F. et al. Calreticulin exposure dictates the immunogenicity of cancer cell death. Nat Med 13, 54–61 (2007). https://doi.org/10.1038/nm1523
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1523
This article is cited by
-
The promise and challenges of combination therapies with antibody-drug conjugates in solid tumors
Journal of Hematology & Oncology (2024)
-
Targeting of focal adhesion kinase enhances the immunogenic cell death of PEGylated liposome doxorubicin to optimize therapeutic responses of immune checkpoint blockade
Journal of Experimental & Clinical Cancer Research (2024)
-
Bacteria colonization in tumor microenvironment creates a favorable niche for immunogenic chemotherapy
EMBO Molecular Medicine (2024)
-
Chiral coordination polymer nanowires boost radiation-induced in situ tumor vaccination
Nature Communications (2024)
-
Shikonin improves the effectiveness of PD-1 blockade in colorectal cancer by enhancing immunogenicity via Hsp70 upregulation
Molecular Biology Reports (2024)