Abstract
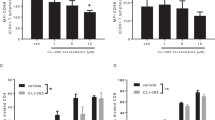
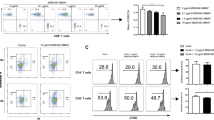
A major goal of immunotherapy for autoimmune diseases and transplantation is induction of regulatory T cells that mediate immunologic tolerance. The mucosal immune system is unique, as tolerance is preferentially induced after exposure to antigen, and induction of regulatory T cells is a primary mechanism of oral tolerance. Parenteral administration of CD3-specific monoclonal antibody is an approved therapy for transplantation in humans and is effective in autoimmune diabetes. We found that orally administered CD3-specific antibody is biologically active in the gut and suppresses autoimmune encephalomyelitis both before induction of disease and at the height of disease. Orally administered CD3-specific antibody induces CD4+CD25−LAP+ regulatory T cells that contain latency-associated peptide (LAP) on their surface and that function in vitro and in vivo through a TGF-β–dependent mechanism. These findings identify a new immunologic approach that is widely applicable for the treatment of human autoimmune conditions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Herold, K.C. et al. Prevention of autoimmune diabetes with nonactivating anti-CD3 monoclonal antibody. Diabetes 41, 385–391 (1992).
Chatenoud, L., Thervet, E., Primo, J. & Bach, J.F. Anti-CD3 antibody induces long-term remission of overt autoimmunity in nonobese diabetic mice. Proc. Natl. Acad. Sci. USA 91, 123–127 (1994).
Chatenoud, L., Primo, J. & Bach, J.F. CD3 antibody-induced dominant self tolerance in overtly diabetic NOD mice. J. Immunol. 158, 2947–2954 (1997).
Mottram, P.L., Murray-Segal, L.J., Han, W., Maguire, J. & Stein-Oakley, A.N. Remission and pancreas isograft survival in recent onset diabetic NOD mice after treatment with low-dose anti-CD3 monoclonal antibodies. Transpl. Immunol. 10, 63–72 (2002).
von Herrath, M.G., Coon, B., Wolfe, T. & Chatenoud, L. Nonmitogenic CD3 antibody reverses virally induced (rat insulin promoter-lymphocytic choriomeningitis virus) autoimmune diabetes without impeding viral clearance. J. Immunol. 168, 933–941 (2002).
Belghith, M. et al. TGF-β-dependent mechanisms mediate restoration of self-tolerance induced by antibodies to CD3 in overt autoimmune diabetes. Nat. Med. 9, 1202–1208 (2003).
Tran, G.T. et al. Reversal of experimental allergic encephalomyelitis with non-mitogenic, non-depleting anti-CD3 mAb therapy with a preferential effect on T(h)1 cells that is augmented by IL-4. Int. Immunol. 13, 1109–1120 (2001).
Kohm, A.P. et al. Treatment with nonmitogenic anti-CD3 monoclonal antibody induces CD4+ T cell unresponsiveness and functional reversal of established experimental autoimmune encephalomyelitis. J. Immunol. 174, 4525–4534 (2005).
Herold, K.C. et al. Anti-CD3 monoclonal antibody in new-onset type 1 diabetes mellitus. N. Engl. J. Med. 346, 1692–1698 (2002).
Herold, K.C. et al. Activation of human T cells by FcR nonbinding anti-CD3 mAb, hOKT3gamma1(Ala-Ala). J. Clin. Invest. 111, 409–418 (2003).
Keymeulen, B. et al. Insulin needs after CD3-antibody therapy in new-onset type 1 diabetes. N. Engl. J. Med. 352, 2598–2608 (2005).
Bisikirska, B., Colgan, J., Luban, J., Bluestone, J.A. & Herold, K.C. TCR stimulation with modified anti-CD3 mAb expands CD8+ T cell population and induces CD8+CD25+ Tregs. J. Clin. Invest. 115, 2904–2913 (2005).
Herold, K.C. et al. A single course of anti-CD3 monoclonal antibody hOKT3gamma1(Ala-Ala) results in improvement in C-peptide responses and clinical parameters for at least 2 years after onset of type 1 diabetes. Diabetes 54, 1763–1769 (2005).
Utset, T.O. et al. Modified anti-CD3 therapy in psoriatic arthritis: a phase I/II clinical trial. J. Rheumatol. 29, 1907–1913 (2002).
Chatenoud, L. et al. Human in vivo antigenic modulation induced by the anti-T cell OKT3 monoclonal antibody. Eur. J. Immunol. 12, 979–982 (1982).
Chatenoud, L. et al. Restriction of the human in vivo immune response against the mouse monoclonal antibody OKT3. J. Immunol. 137, 830–838 (1986).
Chatenoud, L. CD3-specific antibody-induced active tolerance: from bench to bedside. Nat. Rev. Immunol. 3, 123–132 (2003).
Faria, A.M. & Weiner, H.L. Oral tolerance. Immunol. Rev. 206, 232–259 (2005).
Zhang, Z.J., Davidson, L., Eisenbarth, G. & Weiner, H.L. Suppression of diabetes in NOD mice by oral administration of porcine insulin. Proc. Natl. Acad. Sci. USA 88, 10252–10256 (1991).
Maron, R. et al. Mucosal administration of heat shock protein-65 decreases atherosclerosis and inflammation in aortic arch of low-density lipoprotein receptor-deficient mice. Circulation 106, 1708–1715 (2002).
Frenkel, D. et al. Nasal vaccination with myelin oligodendrocyte glycoprotein reduces stroke size by inducing IL-10-producing CD4+ T cells. J. Immunol. 171, 6549–6555 (2003).
Miller, A., Lider, O., Roberts, A.B., Sporn, M.B. & Weiner, H.L. Suppressor T-cells generated by oral tolerization to myelin basic protein suppress both in vitro and in vivo immune responses by the release of TGF-β following antigen-specific triggering. Proc. Natl. Acad. Sci. USA 89, 421–425 (1992).
Chen, Y., Kuchroo, V.K., Inobe, J., Hafler, D.A. & Weiner, H.L. Regulatory T cell clones induced by oral tolerance: suppression of autoimmune encephalomyelitis. Science 265, 1237–1240 (1994).
Zhang, X., Izikson, L., Liu, L. & Weiner, H.L. Activation of CD25(+)CD4(+) regulatory T cells by oral antigen administration. J. Immunol. 167, 4245–4253 (2001).
Lider, O., Santos, L.M., Lee, C.S., Higgins, P.J. & Weiner, H.L. Suppression of experimental autoimmune encephalomyelitis by oral administration of myelin basic protein. II. Suppression of disease and in vitro immune responses is mediated by antigen-specific CD8+ T lymphocytes. J. Immunol. 142, 748–752 (1989).
Oida, T. et al. CD4+CD25− T cells that express latency-associated peptide on the surface suppress CD4+CD45RBhigh-induced colitis by a TGF-β-dependent mechanism. J. Immunol. 170, 2516–2522 (2003).
Nakamura, K., Kitani, A. & Strober, W. Cell contact-dependent immunosuppression by CD4(+)CD25(+) regulatory T cells is mediated by cell surface-bound transforming growth factor β. J. Exp. Med. 194, 629–644 (2001).
Nakamura, K. et al. TGF-β 1 plays an important role in the mechanism of CD4+CD25+ regulatory T cell activity in both humans and mice. J. Immunol. 172, 834–842 (2004).
Inobe, J. et al. IL-4 is a differentiation factor for transforming growth factor-β secreting Th3 cells and oral administration of IL-4 enhances oral tolerance in experimental allergic encephalomyelitis. Eur. J. Immunol. 28, 2780–2790 (1998).
Slavin, A.J., Maron, R. & Weiner, H.L. Mucosal administration of IL-10 enhances oral tolerance in autoimmune encephalomyelitis and diabetes. Int. Immunol. 13, 825–833 (2001).
Sayegh, M.H., Khoury, S.J., Hancock, W.W., Weiner, H.L. & Carpenter, C.B. Induction of immunity and oral tolerance with polymorphic class II major histocompatibility complex allopeptides in the rat. Proc. Natl. Acad. Sci. USA 89, 7762–7766 (1992).
Miller, A., al-Sabbagh, A., Santos, L.M., Das, M.P. & Weiner, H.L. Epitopes of myelin basic protein that trigger TGF-β release after oral tolerization are distinct from encephalitogenic epitopes and mediate epitope-driven bystander suppression. J. Immunol. 151, 7307–7315 (1993).
Maron, R., Melican, N.S. & Weiner, H.L. Regulatory Th2-type T cell lines against insulin and GAD peptides derived from orally- and nasally-treated NOD mice suppress diabetes. J. Autoimmun. 12, 251–258 (1999).
Wesselborg, S., Janssen, O. & Kabelitz, D. Induction of activation-driven death (apoptosis) in activated but not resting peripheral blood T cells. J. Immunol. 150, 4338–4345 (1993).
Friedman, A. & Weiner, H.L. Induction of anergy or active suppression following oral tolerance is determined by antigen dosage. Proc. Natl. Acad. Sci. USA 91, 6688–6692 (1994).
Chen, Y., Inobe, J., Marks, R., Gonnella, P., Kuchroo, V.J. & Weiner, H.L. Peripheral deletion of antigen-reactive T-cells in oral tolerance. Nature 376, 177–180 (1995).
Jorritsma, P.J., Brogdon, J.L. & Bottomly, K. Role of TCR-induced extracellular signal-regulated kinase activation in the regulation of early IL-4 expression in naive CD4+ T cells. J. Immunol. 170, 2427–2434 (2003).
Leitenberg, D. & Bottomly, K. Regulation of naive T cell differentiation by varying the potency of TCR signal transduction. Semin. Immunol. 11, 283–292 (1999).
Khoury, S.J., Hancock, W.W. & Weiner, H.L. Oral tolerance to myelin basic protein and natural recovery from experimental autoimmune encephalomyelitis are associated with downregulation of inflammatory cytokines and differential upregulation of transforming growth factor beta, interleukin 4, and prostaglandin E expression in the brain. J. Exp. Med. 176, 1355–1364 (1992).
Zhang, X. et al. Recovery from experimental allergic encephalomyelitis is TGF-{β} dependent and associated with increases in CD4+LAP+ and CD4+CD25+ T cells. Int. Immunol. 18, 495–503 (2006).
Weinshenker, B.G., Bass, B., Karlik, S., Ebers, G.C. & Rice, G.P. An open trial of OKT3 in patients with multiple sclerosis. Neurology 41, 1047–1052 (1991).
Hafler, D.A., Ritz, J., Schlossman, S.F. & Weiner, H.L. Anti-CD4 and anti-CD2 monoclonal antibody infusions in subjects with multiple sclerosis. Immunosuppressive effects and human anti-mouse responses. J. Immunol. 141, 131–138 (1988).
Acknowledgements
We would like to thank V. Kuchroo, S. Khoury and B. Waksman for discussions, and S. Ori for administrative support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Proliferative response to different antigens. (PDF 76 kb)
Supplementary Fig. 2
Proliferation after intravenous treatment with anti-CD3 Ab. (PDF 54 kb)
Supplementary Fig. 3
Cytokine profile after intravenous treatment with anti-CD3 Ab. (PDF 43 kb)
Supplementary Table 1
Percentage of CD3+, CD25+ and LAP+ cells in lymphoid organs after feeding. (PDF 30 kb)
Supplementary Table 2
Percentage of CD69+ cells among CD4+CD25−LAP+ T cells in mesenteric lymph node after anti-CD3 treatment. (PDF 24 kb)
Supplementary Table 3
Percentage of LAP+ cells in popliteal lymph nodes after feeding and immunization. (PDF 24 kb)
Rights and permissions
About this article
Cite this article
Ochi, H., Abraham, M., Ishikawa, H. et al. Oral CD3-specific antibody suppresses autoimmune encephalomyelitis by inducing CD4+CD25−LAP+ T cells. Nat Med 12, 627–635 (2006). https://doi.org/10.1038/nm1408
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1408
This article is cited by
-
How could nanobiotechnology improve treatment outcomes of anti-TNF-α therapy in inflammatory bowel disease? Current knowledge, future directions
Journal of Nanobiotechnology (2021)
-
Mechanisms of Oral Tolerance
Clinical Reviews in Allergy & Immunology (2018)
-
Oral immune therapy: targeting the systemic immune system via the gut immune system for the treatment of inflammatory bowel disease
Clinical & Translational Immunology (2016)
-
Regulatory T cells in atherosclerosis: critical immune regulatory function and therapeutic potential
Cellular and Molecular Life Sciences (2016)
-
High sugar and butter (HSB) diet induces obesity and metabolic syndrome with decrease in regulatory T cells in adipose tissue of mice
Inflammation Research (2016)