Abstract
There is currently great interest in developing mucosal vaccines against a variety of microbial pathogens. Mucosally induced tolerance also seems to be a promising form of immunomodulation for treating certain autoimmune diseases and allergies. Here we review the properties of the mucosal immune system and discuss advances in the development of mucosal vaccines for protection against infections and for treatment of various inflammatory disorders.
Similar content being viewed by others
Main
The mucous membranes covering the aerodigestive and the urogenital tracts as well as the eye conjunctiva, the inner ear and the ducts of all exocrine glands are endowed with powerful mechanical and chemical cleansing mechanisms that degrade and repel most foreign matter. In addition, a large and highly specialized innate and adaptive mucosal immune system protects these surfaces, and thereby also the body interior, against potential insults from the environment. In a healthy human adult, this local immune system contributes almost 80% of all immunocytes. These cells are accumulated in, or in transit between, various mucosa-associated lymphoid tissues (MALT), which together form the largest mammalian lymphoid organ system1.
The mucosal immune system has three main functions: (i) to protect the mucous membranes against colonization and invasion by potentially dangerous microbes that may be encountered, (ii) to prevent uptake of undegraded antigens including foreign proteins derived from ingested food, airborne matter and commensal microorganisms, and (iii) to prevent the development of potentially harmful immune responses to these antigens if they do reach the body interior. At variance with the systemic immune apparatus, which functions in a normally sterile milieu and often responds vigorously to invaders, the MALT guards organs that are replete with foreign matter. It follows that upon encountering this plethora of antigenic stimuli, the MALT must economically select appropriate effector mechanisms and regulate their intensity to avoid bystander tissue damage and immunological exhaustion.
Mucosal immune responses
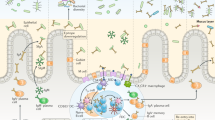
The MALT represents a highly compartmentalized immunological system and functions essentially independent from the systemic immune apparatus. It is comprised of anatomically defined lymphoid microcompartments such as the Peyer patches, the mesenteric lymph nodes, the appendix and solitary follicles in the intestine, and the tonsils and adenoids at the entrance of the aerodigestive tract, which serve as the principal mucosal inductive sites where immune responses are initiated2,3. Small but numerous clusters of immature lymphocytes and dendritic cells have also been described in the subepithelial compartment of the mouse intestine and may represent sites of extrathymic lymphopoiesis4,5; such cryptopatches have not been found in humans, however. The MALT also contains diffuse accumulations of large numbers of lymphoid cells in the parenchyma of mucosal organs and exocrine glands, which form the mucosal effector sites where immune responses are manifested. Consistent with a high degree of compartmentalization, the MALT is populated by phenotypically and functionally distinct B cell, T cell and accessory cell subpopulations as compared with systemic lymphoid tissues, and has also developed strong restrictions upon lymphoid cell recirculation between mucosal sites.
As more extensively reviewed elsewhere6, antigens taken up by absorptive epithelial cells and specialized epithelial cells (membrane, or 'M,' cells) in mucosal inductive sites can be shuttled to, or directly captured by, 'professional' antigen-presenting cells (APCs; including dendritic cells (DCs), B lymphocytes and macrophages), and presented to conventional CD4+ and CD8+ αβ T cells, all located in the inductive sites. Certain antigens may also be processed and presented directly by epithelial cells to neighboring intraepithelial T cells, including T cells with limited repertoire diversity (γδ T cells and NKT cells). Immune responses in mucosal tissues are governed by the nature of the antigen, the type of APCs involved, and the local microenvironment. With most types of nonpathogen antigens (e.g., food proteins), the 'default' pathway for mucosal DCs and other APCs seems to be to generate T helper 2 (TH2) and various regulatory T cell types of responses7; this usually also results in active suppression of systemic immunity—'oral tolerance.' Antigens and adjuvants, including most pathogens, harboring motifs sensed by mucosal APCs as 'danger signals' (e.g., Toll-like receptor (TLR) ligands), on the other hand, and proinflammatory conditions in general, favor the development of stronger and broader immune responses engaging both the humoral-secretory and cellular immunity effector arms and also do not lead to oral tolerance2,6,7. It has been widely assumed that the recognition of pathogens by TLRs on mucosal APCs was distinct from the response to the commensal flora, but recently it was found that microbial commensals are also recognized by TLRs under normal conditions, and that this interaction seems crucial for maintaining epithelial homeostasis in the gut8.
The sensitized mucosal immunocytes, both B and T cells, leave the site of initial encounter with antigen (e.g., a Peyer patch), transit through the lymph, enter the circulation and then seed selected mucosal sites, mainly the mucosa of origin, where they differentiate into memory or effector cells. The anatomic affinity of such cells seems to be largely determined by site-specific integrins ('homing receptors') on their surface and complementary mucosal tissue–specific receptors ('addressins') on vascular endothelial cells9. In addition, chemokines produced in the local microenvironment promote chemotaxis toward mucosal tissues and regulate integrin expression on mucosal immunocytes, thereby controlling cell migration10. Of particular interest are recent studies indicating that mucosal DCs, in addition to presenting antigen to cognate T cells, can also influence their homing properties. Thus, mouse DCs isolated from mesenteric lymph nodes and Peyer patches, but not from spleen and peripheral lymph nodes, increase the expression of the mucosal homing receptor α4β7 (refs. 11,12) and CCR9 (ref. 12), the receptor for the gut-associated chemokine TECK/CCL25 on memory T cells, and license effector/memory CD8+ T cells to home preferentially to the intestinal epithelium. Notably, DC imprinting of gut homing specificity has recently been shown to involve retinoic acid, which is uniquely produced by intestinal DCs, but not by DCs from other lymphoid organs13. Taken together, these observations may explain the notion of a 'common mucosal immune system' whereby immunocytes activated at one site disseminate immunity to remote mucosal tissues rather than to systemic sites. At the same time, because chemokines, integrins and cytokines are differentially expressed among mucosal tissues, this fact may also partly explain why, within the mucosal immune system, there is a significant degree of compartmentalization linking specific mucosal inductive sites with particular effector sites (e.g., the gut with the mammary glands and the nose with the respiratory and genital mucosae).
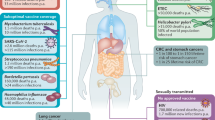
The compartmentalization within the mucosal immune system places constraints on the choice of vaccination route for inducing effective immune responses at the desired sites (Fig. 1). Thus, whereas oral immunization may induce substantial antibody responses in the small intestine (strongest in the proximal segment), ascending colon and mammary and salivary glands, it is relatively inefficient at evoking an IgA antibody response in the distal segments of the large intestines, tonsils or female genital tract mucosa14,15,16. Conversely, rectal immunization evokes strong local antibody responses in the rectum but little, if any, response in the small intestine and in the proximal colon15,16,17. Nasal or tonsilar immunization in humans results in antibody responses in the upper airway mucosa and regional secretions (saliva, nasal secretions) without evoking an immune response in the gut18,19; however, and of special interest for possible vaccination against HIV and other sexually transmitted infections, not only vaginal but also nasal immunization has been found to give rise to substantial IgA and IgG antibody responses in the human cervicovaginal mucosae16,19,20. Another notable finding, if it can be confirmed in humans, is that in mice, transcutaneous immunization may induce a mucosal immune response in the female genital tract21. It should also be borne in mind that the menstrual status of females may influence the intensity of immune responses in genital secretions19,20.
The 'common mucosal immune system' is more restricted than previously thought. In humans, immunization studies with cholera toxin B subunit by different mucosal routes have clearly shown that the strongest response takes place at the directly vaccine-exposed mucosa and the second-best responses at adjacent mucosae or at specifically interconnected inductive-expression mucosal systems such as the gut-mammary gland link in lactating women. A notable exception is the fact that nasal mucosal immunization not only stimulates an immune response in the respiratory tract, but also can give rise to a strong genital-vaginal mucosal immune response. Shading indicates strength of response.
Effector mechanisms
In addition to the barrier function, mechanical cleansing mechanisms and different chemical antimicrobial factors or defensins provided by the lining epithelium of different mucosal tissues, the mucosa contains a number of other cells of the innate immune system, including phagocytic neutrophils and macrophages, DCs, NK cells and mast cells. Through a variety of mechanisms these cells contribute significantly to host defense against pathogens22 and for initiating adaptive mucosal immune responses.
The adaptive humoral immune defense at mucosal surfaces is to a large extent mediated by secretory IgA (SIgA) antibodies, the predominant immunoglobulin class in human external secretions. The resistance of SIgA to proteases makes these antibodies uniquely suited for functioning in mucosal secretions (Box 1). The induction of IgA against mucosal pathogens and soluble protein antigens is dependent on T helper cells23,24, although IgA immunity to commensal flora may be thymus independent25 and of low affinity26. In humans, transforming growth factor (TGF)-β and interleukin (IL)-10 in concert with IL-4 have been shown to promote B-cell switch to IgA and differentiation into IgA-producing cells27,28. In this regard, in addition to mucosal T cells, which produce large amounts of IL-4, IL-10 and TGF-β, human mucoepithelial cells provide a major source of TGF-β and IL-10, suggesting that cooperation between neighboring lymphocytes and epithelial cells in the mucosal microenvironment is pivotal for programming preferential maturation of IgA-committed B cells.
Although SIgA is the predominant humoral defense mechanism at mucosal surfaces, locally produced IgM and IgG, and in the lower respiratory tract and in the genitourinary mucosa, serum-derived IgG can also contribute significantly to immune defense.
Mucosal cytotoxic T lymphocyte (CTL) responses have been described after oral, nasal, rectal or vaginal immunization29,30, and recently also after transcutaneous immunization31. Mucosal CTLs have been shown to be crucial for the immune clearance of pathogens in several animal models of infection with enteric or respiratory viruses and intracellular parasites32,33,34. In most studies, wild-type or attenuated viruses and bacteria have been required to induce CTLs in mucosal tissues. There are, however, exceptions to this rule, inasmuch as use of certain adjuvants such as cholera toxin and related enterotoxins can promote mucosal CTL development when administered orally or nasally with soluble proteins and peptides35,36. Besides CTLs, interferon (IFN)-γ–producing CD4+ T cells, induced either by the live pathogens or by mucosal immunization with inactivated vaccines in combination with cholera toxin or other mucosal adjuvants, have been found to be important for mucosal immune defense to both viral and bacterial infections; their protective mechanism(s), however, remain to be defined37,38,39. Thus, appropriate adjuvants or delivery systems, or both, may critically favor the induction of protective mucosal cellular responses, and this notion is of importance for developing mucosal vaccines against intracellular pathogens.
Regulatory mechanisms
The mucosal immune system has evolved a variety of mechanisms to achieve and maintain tolerance against self-antigens and against the plethora of environmental antigens present in the microflora, in food and among airborne matter. Studies in animal models have identified that mucosal tolerance can be achieved through different mechanisms, including activation-induced cell death, anergy and, most important, the induction of regulatory T cells40. Anergy of antigen-specific T cells has been reported after inhalation or ingestion of large quantities of soluble proteins41, and deletion of specific T cells only after mucosal administration of massive, nonphysiological antigen doses42. Induction of regulatory cells after mucosal delivery of antigens has been reported in animal models for more than 25 years43 and has received a major attention during the last few years given the potential of such regulatory cells as therapeutic agents in immune-mediated diseases.
In mice, four main types of regulatory T cells have been described: (i) antigen-induced CD4+ TH2-like cells that produce IL-4 and IL-10 and antagonize the activity of TH1 effector cells44; (ii) CD4+CD45RBlow Tr1 cells that function through the production of IL-10 (ref. 45); (iii) CD4+ or CD8+ T cells producing TGF-β (TH3 cells)46; and apparently most important, (iv) a population of naturally occurring CD4+CD25+ regulatory T cells (Treg cells) that suppress proliferation through a cell contact–dependent mechanism47,48. Although anergic in vitro, the latter cells can be expanded in an antigen-specific manner in vivo after immunization49,50. Notably, these cells may also confer suppressor activity on other CD4+ T cells by inducing the expression of the transcription factor Foxp3 and/or the major histocompatibility complex (MHC) class II–binding molecule LAG-3 in such cells ('infectious tolerance')51,52. Thereby, they may also provide a direct link between effector T-cell inhibition by Treg, TH3 and Tr1 cells. Thus, natural human CD4+CD25+ Treg expressing the mucosal α4β7 integrin, when co-cultured with conventional CD4+ T cells, induced Tr1-like IL-10–secreting T cells with strong suppressor activity on effector T cells, whereas another, α4β1-positive Treg subset in similar cultures instead induced TH3-like TGF-β–secreting suppressor T cells51. Recent evidence indicates that all of these different regulatory T cell types and mechanisms can be induced or expanded by mucosal administration of antigens leading to peripheral tolerance (oral tolerance; J.-B. Sun et al., unpublished data).
Intraepithelial CD8+ γδ T cells from the respiratory mucosa and from the small intestine have also been suggested to be involved in mucosal tolerance53,54. Despite their strategic location as the first immunocytes to encounter pathogens that have invaded an epithelial surface, the mechanisms of action of intraepithelial regulatory T cells are still largely unknown. The liver also seems to have a significant role in maintaining immunological silence to harmless antigenic material present in food. Regulatory T cells as well as NK and CD1-restricted NKT cells seem to contribute to the overall bias of hepatic immune responses toward tolerance and this bias may account for the survival of liver allografts and the persistence of certain liver pathogens such as hepatitis viruses55,56. Thus, protection of mucosal organs from autoagressive and allergic diseases seems to involve several layers of regulation.
Activation, expansion and survival of these various regulatory cells seem to be controlled by specialized types of APCs, especially tissue-specific DCs such as liver sinusoidal DCs, certain subpopulations of DCs in Peyer patches, mesenteric lymph nodes and small intestinal villi, or in the lungs6,7,56,57. Thus, mucosal DCs, depending on several major factors such as their tissue location, lineage to which they belong and the nature of maturation stimuli, can determine the character of the ensuing immune responses. Targeting regulatory or immunostimulatory mucosal DCs constitutes a major challenge to the development of adjuvants and vaccine formulations.
Mucosal vaccines against infections
The primary reason for using a mucosal route of vaccination is that most infections affect or start from a mucosal surface, and that in these infections, topical application of a vaccine is often required to induce a protective immune response. Examples include gastrointestinal infections caused by Helicobacter pylori, Vibrio cholerae, enterotoxigenic Escherichia coli (ETEC), Shigella spp., Clostridium difficile, rotaviruses and calici viruses; respiratory infections caused by Mycoplasma pneumoniae, influenza virus and respiratory syncytial virus; and sexually transmitted genital infections caused by HIV, Chlamydia trachomatis, Neisseria gonorrhoeae and herpes simplex virus. These infections represent an enormous challenge for development of vaccines targeted to induce immunity that can either prevent the infectious agent from attaching and colonizing at the mucosal epithelium (noninvasive bacteria), or from penetrating and replicating in the mucosa (viruses and invasive bacteria), and/or that can block microbial toxins from binding to and affecting epithelial and other target cells.
A topical-mucosal route of vaccination seems to be crucial for protective efficacy against noninvasive infections at mucosal surfaces that are normally impermeable to serum antibody transudation, or passive passage across an epithelium. Gastrointestinal infections with V. cholerae or ETEC are examples of such infections, in which vaccine-induced protection is mediated mainly, if not exclusively, by locally produced SIgA antibodies and is associated with immunologic memory. In other instances, such as in H. pylori gastrointestinal infection and chlamydial or herpes simplex virus genital infections, the protective immunity is mediated mainly by mucosal CD4+ T helper cells and possibly also CD8+ CTL and NK cells37,38,39. When, on the other hand, the infection occurs at mucosal surfaces such as the respiratory and urogenital mucosae, which are more permeable than the intestine to transudation of serum antibodies, a parenteral route of vaccination may also be effective. The same may hold true for enteric infections in which the pathogen is first translocated across the epithelial barrier by intestinal membrane, or M, cells and then infects the basolateral side of the epithelium as is the case with Shigella spp., or causes disease only after multiplying and inducing inflammation in the submucosal lymphoid tissues (most salmonellae) and/or as for Salmonella typhi after further bacteremic spread in systemic tissues.
But despite the many attractive features of mucosal vaccination, it has often proven difficult in practice to stimulate strong SIgA immune responses and protection by mucosal administration of antigens. In fact, as yet only half a dozen of the vaccines that are currently approved for human use are administered mucosally (Table 1).
Oral polio vaccine. The oral polio vaccine (OPV) is the classical oral-mucosal vaccine. In addition to its enormous impact for reducing polio in the world, this vaccine has also served as a useful tool for elucidating fundamental aspects of mucosal immunity in humans58. Like the injectable inactivated polio vaccine (IPV), OPV produces antibodies in the blood that will protect against myelitis by preventing the spread of poliovirus to the nervous system. But, superior to IPV, OPV also produces a local SIgA immune response in the intestinal mucosa—the primary site for poliovirus entry and multiplication. This intestinal immune response can rapidly stop person-to-person transmission of wild poliovirus, making mass campaigns with OPV a powerful strategy for the global eradication of polio. At the same time, there is a low but real risk of reversion of OPV virus strains toward neurovirulence, which has led to the replacement of OPV by IPV in most industrialized countries. As the global eradication of polio is now approaching, concerns have been raised about the continued use of OPV in most developing countries after such eradication, and how to financially and logistically make it possible in these countries to use the safe but more expensive IPV instead.
Vaccines against enteric infections. Enteric infections resulting in diarrheal disease or typhoid fever remain a leading global health problem, causing an estimated 3 billion disease episodes and 3 million deaths annually in developing countries. Efficacious vaccines are now available against three of the main pathogens—V. cholerae, S. typhi and rotavirus—whereas vaccines are still lacking against the two other most important causes of disease, ETEC and Shigella.
Cholera vaccines. Almost half of all diarrheas are the result of enterotoxin-producing bacteria. Among these, V. cholerae causes the most severe disease and epidemic outbreaks. Previously used injectable cholera vaccines, which did not induce significant gut mucosal immune responses, afforded poor protection and have been abandoned. Recently, however, two improved oral cholera vaccines have become available. The most widely used of these vaccines (Dukoral), consisting of recombinantly produced cholera toxin B subunit (CTB) and inactivated V. cholerae O1 whole cells covering different serotypes and biotypes, has proven to be safe and stable and has in large efficacy (designed to measure the intrinsic effects of vaccination when given under ideal conditions) and effectiveness (designed to measure the vaccination effect under conditions likely to predict how well the vaccine will perform in public health practice) trials in Bangladesh, Peru and Mozambique conferred 85–90% short-term (first 6 months) and 60% long-term (first 3 years) protection against cholera59,60,61 together with evidence of strong herd immunity62. Protection is mediated by local production of antitoxic and antibacterial SIgA antibodies in the gut. Through its CTB component, this vaccine also affords significant cross-protection against ETEC producing cholera toxin–like heat-labile toxin; ETEC is the most common cause of diarrheal disease both in children living in developing countries and among travelers to such countries. A simplified killed whole-cell cholera vaccine, without the CTB component, has also been produced, tested and licensed locally in Vietnam63.
The second internationally licensed oral cholera vaccine is the live, attenuated vaccine CVD 103-HgR, containing a genetically manipulated classical V. cholerae O1 Inaba strain with a deletion in the gene encoding cholera toxin59,64. The vaccine has proven to be safe and has conferred 60–100% protection against experimental challenge with cholera organisms in North American volunteers. Based on this, CVD 103-HgR (Orochol) is licensed in several countries for use in travelers. But when the vaccine was tested in a large field trial in Indonesia, no significant protection was observed65, and hence the usefulness of this vaccine in cholera-endemic areas remains to be established.
Typhoid fever vaccines. The old parenteral, killed whole-cell typhoid vaccine was effective, but it gave rise to severe local reactions and often fever. More recently, two safe and effective vaccines against typhoid fever have been licensed. The most widely used of these vaccines consists of purified capsular polysaccharide Vi antigen. This vaccine, which is given parenterally in a single dose, and is assumed to protect by way of serum antibodies, is well tolerated and, when tested in high-endemic countries, it gave 70% protection against typhoid fever during the first 12–18 months and 55% protection over a 3-year study period66,67. A locally produced Chinese Vi vaccine has afforded comparable (70%) protection68.
The other licensed vaccine against typhoid fever is a live, attenuated oral vaccine, Ty21a, developed by chemical mutagenesis of a pathogenic S. typhi strain. Ty21a is well tolerated, and in schoolchildren in Chile three doses of the commercial enteric-coated capsule formulation were shown to provide 67% protective efficacy during the first 3 years of follow up69. The extent to which protection by this oral vaccine is mediated by local mucosal immunity in the gut or by systemic immunity is not known. Rotavirus vaccines. Rotavirus together with ETEC is the most important cause of diarrheal mortality in infants and children below 2 years of age, causing 500,000–600,000 deaths annually, mostly in developing countries.
Most rotavirus strains belong to one of five antigenic groups. A quadrivalent vaccine based on a Rhesus monkey rotavirus strain equipped with human rotavirus genes was licensed for a short time (RotaShield), but was withdrawn after it was suggested that the vaccine caused intussusception (intestinal invagination)70. Recently, however, a new oral attenuated rotavirus vaccine (RotaRix) has been approved for human use in a few countries71. This single-strain vaccine is based on an attenuated human rotavirus strain and has shown good safety, and when tested in an efficacy trial in Brazil, Mexico and Venezuela, it conferred 61–92% protection against rotavirus hospitalizations. The vaccine is currently being further evaluated in Europe, Latin America, Africa and Asia. Another oral attenuated vaccine (RotaTeq) is also expected to be licensed soon in the United States and other countries70. This is a pentavalent reassortant bovine-human vaccine generated to contain human rotavirus genes for each of the main rotavirus serotypes. In Finnish infants, three doses of vaccine in different concentrations gave 59–77% protection against any rotavirus disease (T. Vesikari, H.F. Clark and P.A. Offit, personal communication).
Respiratory infections. Injectable vaccines against influenza and pneumococcal infections have been in use for a long time. Their main protective mechanism is the induction of serum antibodies, mainly IgG, which prevent systemic spread of the pathogen and which may also, through transudation, exert a local protective effect at the mucosal surfaces of the lower respiratory tract. Recently, however, a live influenza vaccine delivered by intranasal spray (FluMist), comprising cold-adapted, temperature-sensitive attenuated virus reassortant strains that are being adjusted to the antigenic needs for the actual influenza season, was licensed in the United States72. This vaccine induces an immune response that more closely resembles natural immunity than the response elicited by the injectable vaccine73. Both mucosal and systemic immunity contribute to resistance to influenza infection and disease: locally produced SIgA antibodies to virus surface hemagglutinin and neuraminidase are important for protection of the upper respiratory tract and corresponding serum IgG antibodies for protection of the lower respiratory tract and against viremia. Cell-mediated immunity, mainly against virus matrix and nucleoprotein antigens, does not protect against infection, but is important for clearance of virus and recovery from illness. The nasal vaccine induces significantly higher local IgA antibodies in nasal washings and local cell-mediated immunity but less high serum antibody titers than the injectable vaccine. Despite these differences in immune responses, the two types of vaccine have comparable protective efficacy (60–90%), and in elderly people, their combined use may increase the efficacy compared with the use of either vaccine alone.
Mucosal vaccines for immunotherapy
Immune responses are not fail-safe and may underserve or be excessive in protecting the host. As discussed above, several regulatory mechanisms to maintain control of mucosal immune reactivity are in place and are becoming increasingly appreciated as targets for manipulating immunopathologic responses. Mucosally induced immunological tolerance has become an attractive strategy for preventing and possibly treating illnesses resulting from the development of untoward immune reactions against allergens as well as self-antigens.
Vaccines against autoimmune diseases. Because induction of mucosal tolerance is antigen-specific but can be expressed in a nonspecific manner ('bystander suppression') through the production of suppressive cytokines by regulatory T cells in the inflamed microenvironment of the target organ, this approach has been utilized to suppress immune responses against self-antigens; however, it should be noted that bystander suppression remains to be documented in humans. It has been possible to prevent or to delay onset of experimental autoimmune diseases in various animal systems by feeding subjects selected autoantigens or peptide derivatives40 (for example, in rodent models of autoimmune arthritis, type 1 diabetes, experimental autoimmune encephalitis, myasthenia gravis, autoimmune ear chondritis, autoimmune uveitis and autoimmune thyroiditis). In addition to the oral route, virtually all other routes of mucosal administration (nasal, buccal, rectal, genital) are also effective to induce tolerance, although to varying degrees. The dosage, the route and frequency of autoantigen administration have proven to be crucial. Thus, whereas low doses of nasally administered antigens favor expansion of regulatory T cells producing IL-10, low doses of orally administered antigen promote activation of CD8+ and/or CD4+ regulatory T cells producing TGF-β. Large doses of antigens seem to induce anergy of effector CD4+ T cells, whereas massive doses can induce their apoptosis.
Although mucosal tolerance is usually effective in animal models for preventing inducible autoimmune diseases, its efficacy has been more variable and limited when utilized as an intervention strategy in animals in which the disease had already been induced or had spontaneously developed. This may explain, in part, the disappointing results of recent phase 3 clinical trials of oral tolerance in individuals with type 1 diabetes74, multiple sclerosis75 and rheumatoid arthritis76, diseases in which there may be multiple target autoantigens that remain largely unknown.
A significant improvement has been achieved by coadministering immunomodulating agents to enhance the tolerogenic activity of autoantigens as well as allergens given orally or nasally. The most promising such agent is CTB, which when conjugated or coadministered with several autoantigens or allergens can markedly enhance tolerance induction in already sensitized animals and thereby effectively suppress progression of various autoimmune diseases77,78,79,80,81. Recently, a small phase 1-2 trial in individuals with Behcet disease, an autoimmune eye disease often associated with extraocular manifestations and abnormal T cell reactivity to a specific peptide within the human 60-kDa heat-shock protein, has shown the safety and clinical efficacy of treatment with an oral vaccine comprised of this specific peptide linked to CTB82.
Antiallergy vaccines. Less well appreciated, this approach has given almost uniformly promising results in individuals with allergic disorders, to the extent that a World Health Organization expert group in a position paper recommended sublingual allergen-specific immunotherapy as the suitable treatment for allergic rhinitis in adults. The prevalence and severity of allergic diseases, in particular those affecting the respiratory tract, are increasing, and up to 20% of the population in developed countries are affected. Allergic rhinitis is one of the most common types of mucosal allergies, leading to asthma and early symptoms caused by a hypersensitivity response to airborne allergens such as pollens, dust mites, spores and animal dander. Type I allergy seems to result from the selective activation of allergen-specific TH2 CD4+ T cells providing cognate help to IgE-committed B cells and resulting in hyperproduction of IgE. Specific inactivation of these allergen-specific TH2 cells through clonal anergy, induction of TH1-like cells, which are known to antagonize TH2 cells (immune deviation), or induction of regulatory cells, are considered to be promising approaches for intervention in type I allergic diseases. Systemic allergen-specific immunotherapy by the injection of multiple (20 doses or more) small but increasing amounts of allergen can change a pre-existing allergic TH2 immune response to a nonallergic TH1 response. But this usually requires several years of treatment to be effective and the success rate is well below 50%. It is expensive and complicated, and also carries the risk of allergic and sometimes even life-threatening anaphylactic reactions.
The ability of secretory antibodies to interfere with the entry of allergens through the airway and the gut epithelium has been underestimated, despite the fact that SIgA is known to be noninflammatory and its daily output in external secretions exceeds that of IgG and by far that of IgE antibodies, which it could outcompete for binding to the target allergen. Furthermore, and at variance with systemic immunization, mucosal administration of antigens can induce SIgA antibody responses and, concomitantly, local and peripheral suppression of inflammatory responses. Because mucosal, especially oral or sublingual, vaccines are easier to deliver and safer than injectable vaccines, the concept of 'mucosal desensitization' has become increasingly attractive as an alternative to subcutaneous immunotherapy against type I allergies.
To date, more than 20 double-blind, placebo-controlled clinical trials of mucosal desensitization have been performed in individuals with allergic rhinitis and in individuals with bronchial asthma83 (Table 2). Most studies have involved individuals with allergic rhinitis and have been based on oral and/or sublingual administration of allergen extracts from grass pollen, dust mites, cat, birch pollen, ragweed and Parietaria. Beneficial effects have been reported in the majority of these studies. In long-term studies of individuals with allergic asthma to house dust mites, oral-sublingual immunotherapy with allergen extract was efficient in reducing the frequency of asthmatic attacks and the use of antiasthmatic drugs84. Promising results have also been reported in individuals with atopic dermatitis85.
Overall, the doses of allergen used in these trials and the frequency of allergen administrations have been rather high and, in the majority of trials, natural, and thus inherently heterogeneous, allergen extracts have been used. New techniques including allergen modification, allergen gene vaccination or peptide analogs in combination with selected adjuvants should further increase the safety and efficacy of mucosal immunotherapy in allergies and asthma86.
Needs for improved mucosal vaccine formulations
The development of mucosal vaccines, whether for prevention of infectious diseases or for oral-tolerance immunotherapy, requires efficient antigen delivery and adjuvant systems. Ideally, such systems should (i) protect the vaccine from physical elimination and enzymatic digestion, (ii) target mucosal inductive sites including membrane, or M, cells, and (iii) at least for vaccines against infections, appropriately stimulate the innate immune system to generate effective adaptive immunity.
Mucosal delivery systems. A multitude of such vehicles have been developed, including various inert systems as well as live attenuated bacterial or viral vector systems87,88,89.
Best known among the inert systems are various lipid-based structures with entrapped antigens, such as liposomes, immunostimulating complexes (ISCOMs) and so-called cochleates; different types of biodegradable particles based on starch or copolymers of lactic and glycolic acid; and different mucosa-binding proteins, including both classical plant lectins and bacterial proteins such as the binding subunit portions of cholera toxin or E. coli heat-labile enterotoxin, to which antigens have been linked either chemically or as gene fusion proteins.
Among the many live bacterial vectors developed, two main categories can be distinguished: those based on attenuated pathogens such as Salmonella typhi or S. paratyphi, Bacille Calmette-Guérin or Bordetella pertussis; and those that use commensal bacteria, such as lactobacilli or certain streptococci and staphylococci. The initial use of vaccinia as the primary virus vector candidate has progressively been replaced by other poxviruses, such as canary poxvirus, and by adenoviruses. Several of the live vectors of both bacterial and viral origin have also been engineered to provide various cytokines to further stimulate or modulate the immune responses induced. But although many of these systems have shown promise in animal studies, there is still neither an inert nor a live vector approved for human use.
Promising results have recently been reported from the use of so-called pseudoviruses, or virus-like particles (VLPs). These are self-assembling, nonreplicating viral core structures, often from nonenveloped viruses, that are produced recombinantly in vitro. VLPs are cheap and easy to make, as well as highly immunogenic, and are therefore of commercial interest as viral vaccines in their own right. VLPs can, however, also be used as combined carriers and adjuvants both for foreign antigens expressed recombinantly on their surface, and for DNA vaccines carried within VLPs. VLPs are especially interesting from a mucosal vaccine point of view, as they offer the opportunity to use the natural route of transmission of the parent virus for vaccine delivery. Promising use of this principle, resulting in both SIgA and CTL mucosal immune responses and protection against mucosal pathogen challenge, has been reported from studies both in animals and in humans with VLPs from several mucosal viral pathogens including papillomavirus90, calicivirus91 and hepatitis E virus92.
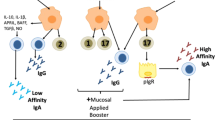
Mucosal adjuvants. When it comes to specific adjuvants, the best-studied and most potent mucosal adjuvants in experimental systems are cholera toxin and E. coli heat-labile enterotoxin93,94, and much effort has been made recently to generate toxicologically acceptable derivatives of these toxins with retained adjuvant activity for human use (Fig. 2).
One such product is the completely nontoxic recombinantly produced CTB, which, depending upon the nature of the coadministered antigen, can be used to promote either mucosal immunity (mainly SIgA) to pathogens or peripheral anti-inflammatory tolerance to self-antigens or allergens93; the latter approach has recently also been tested clinically with promising results in individuals with Behcet disease82.
Mutant heat-labile enterotoxin or cholera toxin proteins have also been made in which the toxic-active A (A1) subunit has been modified in various ways to remove the 'toxic' ADP-ribosylating activity, which leads to toxicity. In general, a loss of toxicity has been matched with a corresponding loss of adjuvanticity, but a few proteins are available with significant adjuvanticity in the absence of detectable toxicity when given intranasally95,96,97.
Yet another approach has been to prepare hybrid molecules in which the fully active cholera toxin A1 subunit has been linked to an engineered specific APC-binding protein derived from Staphylococcus aureus protein A (CTA1-DD)98. This specifically targets the molecule to B cells and has, in experimental systems, proven to be a very efficient and safe adjuvant for coadministered antigens when given intranasally. The incorporation of CTA1-DD and antigen into ISCOM particles may render the adjuvant effective for oral use98.
Bacterial DNA or synthetic oligodeoxynucleotides containing unmethylated 'CpG motifs' (CpG ODN) represent another promising type of mucosal adjuvant. CpG ODN stimulate cells that express Toll-like receptor 9, thereby initiating an immunomodulating cascade. Although as yet mainly considered for systemic use, CpG ODN has been found after nasal, oral or vaginal administration to markedly enhance both innate and adaptive mucosal immunity in animal models99,100, effects which were especially pronounced when CpG ODN was linked to the B subunit protein of cholera toxin100.
Conclusions and perspectives
For many years, mucosal immunity and mucosal vaccines have attracted less than their due share of research and development, considering that most infections and environmental allergies have a mucosal portal of entry.
But in recent years, methodological advances allowing more intense study of mucosal immune responses have led to growing interest in both trying to better understand the specific features of mucosal as compared with systemic immunity, and to develop mucosal vaccines for preventing mucosal infections and for treating allergic or autoimmune diseases. Methods that facilitate the monitoring of mucosal immune responses in humans including infants and young children—the major target groups for vaccination against infectious diseases—have been developed, primarily for measuring secretory antibody responses. But practical assays for assessing mucosal T cell reactivity in clinical and in field settings are still scarce and methods for predicting efficacy of candidate mucosal immunotherapeutics in humans are lacking.
Mucosal immune responses in the humoral-secretory arm of the immune system develop earlier than systemic immune responsiveness, conferring a logistical advantage for mucosal vaccination in infants. On the other hand, it seems that mucosal tolerance develops much later, explaining, in part, the frequency (and often transient nature) of food allergies in young children. There is yet no precise knowledge regarding the ontogeny of the different mucosal regulatory cells for which selective targeting and activation by appropriate delivery systems and immunomodulating agents could be advantageous for preventing allergies and tissue-damaging inflammatory reactions.
Although effective oral-mucosal vaccines for human use are available, it is increasingly appreciated that the development of a broader range of mucosal vaccines, whether for prevention of infectious diseases or for immunotherapy of autoimmune, allergic or infectious inflammatory disorders, will require access to antigen delivery systems that can help present the relevant 'protective antigens' efficiently to the mucosal immune system as well as effective adjuvants to promote and direct the mucosal immune response toward the desired effect. Significant advances have recently been made in the development of improved mucosal vaccine delivery systems. Novel mucosal adjuvants with prospects for human use have also been designed.
Although these developments may promising useful mucosal vaccines, their usefulness in humans has yet to be established. It remains to be seen to what extent the safety and efficacy profiles established in animal models hold true in genetically diverse human subjects who also may differ significantly in their intestinal flora, nutritional status and previous immunological experience, all of which are factors that have been found to affect mucosal vaccine efficacy. Indeed, several mucosal vaccines, including oral live cholera vaccine and rotavirus vaccine candidates as well as OPV, have been found to work less well in developing country settings than in industrialized countries. The pandemic HIV infection problem presents additional challenges with regard to both vaccine safety and efficacy, especially for live attenuated vaccines. Although the main problems to date have dealt with lesser than expected efficacy of mucosal vaccines when tested in specific populations and settings, usually those prevailing in developing countries, it is also notable that two recently developed mucosal vaccines for human use—a live attenuated oral rotavirus vaccine and a nasal influenza subunit vaccine given together with (unmodified) E. coli heat-labile enterotoxin as adjuvant—were withdrawn after a short period because of adverse reactions, underlining the difficult and challenging task for all vaccines to combine vaccine and adjuvant efficacy with safety and public acceptability.
References
Mestecky, J. et al. (eds). Mucosal Immunology 3rd edn. (Academic Press, San Diego, 2005).
Mowat, A.M. Anatomical basis of tolerance and immunity to intestinal antigens. Nat. Rev. Immunol. 3, 331–341 (2003).
Kiyono, H. & Fukuyama, S. NALT- versus Peyer's-patch-mediated mucosal immunity. Nat. Rev. Immunol. 4, 699–710 (2004).
Ishikawa, H., Saito, H., Suzuki, K., Oida, T. & Kanamori, Y. New gut associated lymphoid tissue “cryptopatches” breed murine intestinal intraepithelial T cell precursors. Immunol. Res. 20, 243–250 (1999).
Guy-Grand, D. et al. Extrathymic T cell lymphopoiesis: ontogeny and contribution to gut intraepithelial lymphocytes in athymic and euthymic mice. J. Exp. Med. 197, 333–341 (2003).
Bilsborough, J. & Viney, J.L. Gastrointestinal dendritic cells play a role in immunity, tolerance, and disease. Gastroenterology 127, 300–309 (2004).
Iwasaki, A. & Kelsall, B.L. Freshly isolated Peyer's patch, but not spleen, dendritic cells produce interleukin 10 and induce the differentiation of T helper type 2 cells. J. Exp. Med. 190, 229–239 (1999).
Rakoff-Nahoum, S., Paglino, J., Eslami-Varzaneh, F., Edberg, S. & Medzhitov, R. Recognition of commensal microflora by toll-like receptors is required for intestinal homeostasis. Cell 118, 229–241 (2004).
Kunkel, E.J. & Butcher, E.C. Plasma-cell homing. Nat. Rev. Immunol. 3, 822–829 (2003).
Campbell, D.J., Debes, G.F., Johnston, B., Wilson, E. & Butcher, E.C. Targeting T cell responses by selective chemokine receptor expression. Semin. Immunol. 15, 277–286 (2003).
Stagg, A.J., Kamm, M.A. & Knight, S.C. Intestinal dendritic cells increase T cell expression of α4β7 integrin. Eur. J. Immunol. 32, 1445–1454 (2002).
Mora, J.R. et al. Selective imprinting of gut-homing T cells by Peyer's patch dendritic cells. Nature 424, 88–93 (2003).
Iwata, M. et al. Retinoic acid imprints gut-homing specificity on T cells. Immunity 21, 527–538 (2004).
Quiding, M. et al. Intestinal immune responses in humans. Oral cholera vaccination induces strong intestinal antibody responses, gamma-interferon production, and evokes local immunological memory. J. Clin. Invest. 88, 143–148 (1991).
Eriksson, K. et al. Specific-antibody-secreting cells in the rectums and genital tracts of nonhuman primates following vaccination. Infect. Immun. 66, 5889–5896 (1998).
Kozlowski, P.A., Cu-Uvin, S., Neutra, M.R. & Flanigan, T.P. Comparison of the oral, rectal, and vaginal immunization routes for induction of antibodies in rectal and genital tract secretions of women. Infect. Immun. 65, 1387–1394 (1997).
Jertborn, M., Nordström, I., Kilander, A., Czerkinsky, C. & Holmgren, J. Local and systemic immune responses to rectal administration of recombinant cholera toxin B subunit in humans. Infect. Immun. 69, 4125–4128 (2001).
Johansson, E.L., Bergquist, C., Edebo, A., Johansson, C. & Svennerholm, A.-M. Comparison of different routes of vaccination for eliciting antibody responses in the human stomach. Vaccine 22, 984–990 (2004).
Johansson, E.-L., Wassén, L., Holmgren, J., Jertborn, M. & Rudin, A. Nasal and vaginal vaccinations have differential effects on antibody responses in vaginal and cervical secretions in humans. Infect. Immun. 69, 7481–7486 (2001).
Nardelli-Haefliger, D. et al. Specific antibody levels at the cervix during the menstrual cycle of women vaccinated with human papillomavirus 16 virus-like particles. J. Natl. Cancer Inst. 95, 1128–1137 (2003).
Gockel, C.M., Bao, S. & Beagley, K.W. Transcutaneous immunization induces mucosal and systemic immunity: a potent method for targeting immunity to the female reproductive tract. Mol. Immunol. 37, 537–544 (2000).
Yuan, Q. & Walker, W.A. Innate immunity of the gut: mucosal defense in health and disease. J. Pediatr. Gastroenterol. Nutr. 38, 463–473 (2004).
Lycke, N., Eriksen, L. & Holmgren, J. Protection against cholera toxin after oral immunization is thymus-dependent and associated with intestinal production of neutralizing IgA antitoxin. Scand. J. Immunol. 25, 413–419 (1987).
Hornquist, C.E., Ekman, L., Grdic, K.D., Schon, K. & Lycke, N.Y. Paradoxical IgA immunity in CD4-deficient mice. Lack of cholera toxin-specific protective immunity despite normal gut mucosal IgA differentiation. J. Immunol. 155, 2877–2887 (1995).
Macpherson, A.J. et al. A primitive T cell-independent mechanism of intestinal mucosal IgA responses to commensal bacteria. Science 288, 2222–2226 (2000).
Stoel, M. et al. Restricted IgA repertoire in both B-1 and B-2 cell-derived gut plasmablasts. J. Immunol. 174, 1046–1054 (2005).
Goodrich, M.E. & McGee, D.W. Regulation of mucosal B cell immunoglobulin secretion by intestinal epithelial cell-derived cytokines. Cytokine 10, 948–955 (1998).
Asano, T. et al. Molecular analysis of B cell differentiation in selective or partial IgA defiency. Clin. Exp. Immunol. 136, 284–290 (2004).
Klavinskis, L.S. et al. Mucosal or targeted lymph node immunization of macaques with a particulate SIVp27 protein elicits virus-specific CTL in the genito-rectal mucosa and draining lymph nodes. J. Immunol. 157, 2521–2527 (1996).
Staats, H.F. et al. Cytokine requirements for induction of systemic and mucosal CTL after nasal immunization. J. Immunol. 167, 5386–5394 (2001).
Belyakov, I.M., Hammond, S.A., Ahlers, J.D., Glenn, G.M. & Berzofsky, J.A. Transcutaneous immunization induces mucosal CTLs and protective immunity by migration of primed skin dendritic cells. J. Clin. Invest. 113, 998–1007 (2004).
Franco, M.A. & Greenberg, H.B. Role of B cells and cytotoxic T lymphocytes in clearance of and immunity to rotavirus infection in mice. J. Virol. 69, 7800–7806 (1995).
Bender, B.S., Croghan, T., Zhang, L. & Small, P.A. Jr. Transgenic mice lacking class I major histocompatibility complex-restricted T cells have delayed viral clearance and increased mortality after influenza virus challenge. J. Exp. Med. 175, 1143–1145 (1992).
Buzoni-Gatel, D., Lepage, A.C., Dimier-Poisson, I.H., Bout, D.T. & Kasper, L.H. Adoptive transfer of gut intraepithelial lymphocytes protects against murine infection with Toxoplasma gondii. J. Immunol. 158, 5883–5889 (1997).
Bowen, J.C., Nair, S.K., Reddy, R. & Rouse, B.T. Cholera toxin acts as a potent adjuvant for the induction of cytotoxic T-lymphocyte responses with non-replicating antigens. Immunology 81, 338–342 (1994).
Simmons, C.P. et al. Mucosal delivery of a respiratory syncytial virus CTL peptide with enterotoxin-based adjuvants elicits protective, immunopathogenic, and immunoregulatory antiviral CD8+ T cell responses. J. Immunol. 166, 1106–1113 (2001).
Johansson, M., Schön, K., Ward, M. & Lycke, N. Genital tract infection with Chlamydia trachomatis fails to induce protective immunity in gamma interferon receptor-deficient mice despite a strong local immunoglobulin A response. Infect. Immun. 65, 1032–1044 (1997).
Ermak, T.H. et al. Immunization of mice with urease vaccine affords protection against Helicobacter pylori infection in the absence of antibodies and is mediated by MHC class II-restricted responses. J. Exp. Med. 188, 2277–2288 (1998).
Harandi, A.M., Svennerholm, B., Holmgren, J. & Eriksson, K. Interleukin-12 (IL-12) and IL-18 are important in innate defense against genital herpes simplex virus type 2 infection in mice but are not required for the development of acquired gamma interferon-mediated protective immunity. J. Virol. 75, 6705–6709 (2001).
Wu, H.Y. & Weiner, H.L. Oral tolerance. Immunol. Res. 28, 265–284 (2003).
Whitacre, C.C., Gienapp, I.E., Orosz, C.G. & Bitar, D.M. Oral tolerance in experimental autoimmune encephalomyelitis. III. Evidence for clonal anergy. J. Immunol. 147, 2155–2163 (1991).
Chen, Y. et al. Peripheral deletion of antigen-reactive T cells in oral tolerance. Nature 376, 177–180 (1995).
Ngan, J. & Kind, L.S. Suppressor T cells for IgE and IgG in Peyer's patches of mice made tolerant by the oral administration of ovalbumin. J. Immunol. 120, 861–865 (1978).
Polanski, M., Melican, N.S., Zhang, J. & Weiner, H.L. Oral administration of the immunodominant B-chain of insulin reduces diabetes in a co-transfer model of diabetes in the NOD mouse and is associated with a switch from Th1 to Th2 cytokines. J. Autoimmun. 10, 339–346 (1997).
Groux, H. et al. A CD4+ T-cell subset inhibits antigen-specific T-cell responses and prevents colitis. Nature 389, 737–742 (1997).
Chen, Y., Kuchroo, V.K., Inobe, J., Hafler, D.A. & Weiner, H.L. Regulatory T cell clones induced by oral tolerance: suppression of autoimmune encephalomyelitis. Science 265, 1237–1240 (1994).
Thornton, A.M. & Shevach, E.M. CD4+CD25+ immunoregulatory T cells suppress polyclonal T cell activation in vitro by inhibiting interleukin 2 production. J. Exp. Med. 188, 287–296 (1998).
Sakaguchi, S., Sakaguchi, N., Asano, M., Itoh, M. & Toda, M. Immunologic self-tolerance maintained by activated T cells expressing IL-2 receptor alpha-chains (CD25). Breakdown of a single mechanism of self-tolerance causes various autoimmune diseases. J. Immunol. 155, 1151–1164 (1995).
Klein, L., Khazaie, K. & von Boehmer, H. In vivo dynamics of antigen-specific regulatory T cells not predicted from behavior in vitro. Proc. Natl Acad. Sci. USA 100, 8886–8891 (2003).
Walker, L.S., Chodos, A., Eggena, M., Dooms, H. & Abbas, A.K. Antigen-dependent proliferation of CD4+ CD25+ regulatory T cells in vivo. J. Exp. Med. 198, 249–258 (2003).
Stassen, M. et al. Human CD25+ regulatory T cells: two subsets defined by the integrins α4β7 or α4β1 confer distinct suppressive properties upon CD4+ T helper cells. Eur. J. Immunol. 34, 1303–1311 (2004).
Huang, C.T. et al. Role of LAG-3 in regulatory T cells. Immunity 21, 503–513 (2004).
McMenamin, C., Pimm, C., McKersey, M. & Holt, P.G. Regulation of IgE responses to inhaled antigen in mice by antigen-specific γδ T cells. Science 265, 1869–1871 (1994).
Hanninen, A. & Harrison, L.C. γδ T cells as mediators of mucosal tolerance: the autoimmune diabetes model. Immunol. Rev. 173, 109–119 (2000).
Crispe, I.N. Hepatic T cells and liver tolerance. Nat. Rev. Immunol. 3, 51–62 (2003).
Roelofs-Haarhuis, K., Wu, X. & Gleichmann, E. Oral tolerance to nickel requires CD4+ invariant NKT cells for the infectious spread of tolerance and the induction of specific regulatory T cells. J. Immunol. 173, 1043–1050 (2004).
Akbari, O., DeKruyff, R.H. & Umetsu, D.T. Pulmonary dendritic cells producing IL-10 mediate tolerance induced by respiratory exposure to antigen. Nat. Immunol. 2, 725–731 (2001).
Zhaori, G., Sun, M. & Ogra, P.L. Characteristics of the immune response to poliovirus virion polypeptides after immunization with live or inactivated polio vaccines. J. Infect. Dis. 158, 160–165 (1988).
World Health Organization. Cholera vaccines. WHO position paper. Wkly. Epidemiol. Rec. 76, 117–124 (2001).
Holmgren, J. & Bergquist, C. Oral B subunit killed whole-cell cholera vaccines. in New Generation Vaccines 3rd edn. (ed. Levine, M.M. et al.) 499–510 (Marcel Dekker, New York, 2004).
Lucas, M. et al. High-level effectiveness of a mass oral cholera vaccination in Beira, Mozambique. N. Engl. J. Med. 352, 757–767 (2005).
Ali, M. et al. Herd immunity conferred by killed oral cholera vaccines in Bangladesh. Lancet (in the press).
Trach, D.D. et al. Field trial of a locally produced, killed, oral cholera vaccine in Vietnam. Lancet 349, 231–235 (1997).
Levine, M.M. & Kaper, J.B. Live oral cholera vaccine: from principle to product. Bull. Inst. Pasteur 93, 243–253 (1995).
Richie, E. et al. Efficacy trial of single-dose live oral cholera vaccine CVD 103-HgR in North Jakarta, Indonesia, a cholera-endemic area. Vaccine 18, 2399–2410 (2000).
Acharya, I.L. et al. Prevention of typhoid fever in Nepal with the Vi capsular polysaccharide of Salmonella typhi. A preliminary report. N. Engl. J. Med. 317, 1101–1104 (1987).
Klugman, K. et al. Immunogenicity, efficacy and serological correlate of protection of Salmonella typhi Vi capsular polysaccharide vaccine three years after immunization. Vaccine 14, 435–438 (1996).
Yang, H.H. et al. Efficacy trial of Vi polysaccharide vaccine against typhoid fever in Southwestern China. Bull. World Health Organ. 79, 625–631 (2001).
Levine, M.M. et al. Duration of efficacy of Ty21a attenuated Salmonella typhi live oral vaccine. Vaccine 17, S22–S27 (1999).
Svennerholm, A.M. & Steele, D. Microbial-gut interactions in health and disease. Progress in enteric vaccine development. Best Pract. Res. Clin. Gastroenterol. 18, 421–445 (2004).
De Vos, B. et al. A rotavirus vaccine for prophylaxis of infants against rotavirus gastroenteritis. Pediatr. Infect. Dis. J. 23, S179–S182 (2004).
Belshe, R., Lee, M.-S., Walker, R.E., Stoddard, J. & Mendelman, P.M. Safety, immunogenicity and efficacy of intranasal, live attenuated influenza vaccine. Expert Rev. Vaccines 3, 643–654 (2004).
Cox, R.J., Brokstad, K.A. & Ogra, P. Influenza virus: immunity and vaccination strategies. Comparison of the immune response to inactivated and live, attenuated influenza vaccines. Scand. J. Immunol. 59, 1–15 (2004).
Chaillous, L. et al. Oral insulin administration and residual beta-cell function in recent-onset type 1 diabetes: a multicentre randomised controlled trial. Lancet 356, 545–549 (2000).
Wiendl, H. & Hohlfeld, R. Therapeutic approaches in multiple sclerosis: lessons from failed and interrupted treatment trials. BioDrugs 16, 183–200 (2002).
Postlethwaite, A.E. Can we induce tolerance in rheumatoid arthritis? Curr. Rheumatol. Rep. 3, 64–69 (2001).
Sun, J.B., Rask, C., Olsson, T., Holmgren, J. & Czerkinsky, C. Treatment of experimental autoimmune encephalomyelitis by feeding myelin basic protein conjugated to cholera toxin B subunit. Proc. Natl Acad. Sci. USA 93, 7196–7201 (1996).
Bergerot, I. et al. A cholera toxoid-insulin conjugate as an oral vaccine against spontaneous autoimmune diabetes. Proc. Natl Acad. Sci. USA 94, 4610–4614 (1997).
Tarkowski, A., Sun, J.-B., Holmdahl, R., Holmgren, J. & Czerkinsky, C. Treatment of experimental autoimmune arthritis by nasal administration of a type II collagen-cholera toxoid conjugate vaccine. Arthritis Rheum. 42, 1628–1634 (1999).
Phipps, P.A. et al. Prevention of mucosally induced uveitis with a HSP60-derived peptide linked to cholera toxin B subunit. Eur. J. Immunol. 33, 224–232 (2003).
Tamura, S., Hatori, E., Tsuruhara, T., Aizawa, C. & Kurata, T. Suppression of delayed-type hypersensitivity and IgE antibody responses to ovalbumin by intranasal administration of Escherichia coli heat-labile enterotoxin B subunit-conjugated ovalbumin. Vaccine 15, 225–229 (1997).
Stanford, M. et al. Oral tolerization with peptide 336–351 linked to cholera toxin B subunit preventing relapses of uveitis in Behcet's disease. Clin. Exp. Immunol. 137, 201–208 (2004).
Wilson, D.R., Torres Lima, M. & Dirham, S.R. Sublingual immunotherapy for allergic rhinitis: systematic review and meta-analysis. Allergy 60, 4–12 (2005).
Di Rienzo, V. et al. Long-lasting effect of sublingual immunotherapy in children with asthma due to house dust mite: a 10-year prospective study. Clin. Exp. Allergy 33, 206–210 (2003).
Mastrandrea, F. The potential role of allergen-specific sublingual immunotherapy in atopic dermatitis. Am. J. Clin. Dermatol. 5, 281–294 (2004).
Vrtala, S., Focke-Tejkl, M., Swoboda, I., Kraft, D. & Valenta, R. Strategies for converting allergens into hypoallergenic vaccine candidates. Methods 32, 313–320 (2004).
Holmgren, J., Czerkinsky, C., Eriksson, K. & Harandi, A. Mucosal immunisation and adjuvants: a brief overview of recent advances and challenges. Vaccine 21, S89–S95 (2003).
Schoen, C., Stritzker, J., Goebel, W. & Pilgrim, S. Bacteria as DNA vaccine carriers for genetic immunization. Int. J. Med. Microbiol. 294, 319–335 (2004).
Vajdy, M. et al. Mucosal adjuvants and delivery systems for protein-, DNA- and RNA-based vaccines. Immunol. Cell Biol. 82, 617–627 (2004).
Shi, W., Liu, J., Huang, Y. & Qiao, L. Papillomavirus pseudovirus: a novel vaccine to induce mucosal and systemic cytotoxic T lymphocyte responses. J. Virol. 75, 10139–10148 (2001).
Guerrero, R.A. et al. Recombinant Norwalk virus-like particles administered intranasally to mice induce systemic and mucosal (fecal and vaginal) immune responses. J. Virol. 75, 9713–9722 (2001).
Niikura, M. et al. Chimeric recombinant hepatitis E virus-like particles as an oral vaccine vehicle presenting foreign epitopes. Virology 293, 273–280 (2002).
Holmgren, J., Harandi, A.M. & Czerkinsky, C. Mucosal adjuvants and anti-infection and anti-immunopathology vaccines based on cholera toxin, cholera toxin B subunit and CpG DNA. Expert Rev. Vaccines 2, 205–217 (2003).
Plant, A. & Williams, N.A. Modulation of the immune response by the cholera-like enterotoxins. Curr. Top. Med. Chem. 4, 509–519 (2004).
Pizza, M. et al. Mucosal vaccines: non-toxic derivatives of LT and CT as mucosal adjuvants. Vaccine 19, 2534–2541 (2001).
Lu, X., Clements, J.D. & Katz, J.M. Mutant Escherichia coli heat-labile enterotoxin [LT(R192G)] enhances protective humoral and cellular immune responses to orally administered inactivated influenza vaccine. Vaccine 20, 1019–1029 (2002).
Sanchez, J., Wallerström, G., Fredriksson, M., Angstrom, J. & Holmgren, J. Detoxification of cholera toxin without removal of its immunoadjuvanticity by the addition of (STa-related) peptides to the catalytic subunit. A potential new strategy to generate immunostimulants for vaccination. J. Biol. Chem. 277, 33369–33377 (2002).
Lycke, N. From toxin to adjuvant: the rational design of a vaccine adjuvant vector, CTA1-DD/ISCOM. Cell. Microbiol. 6, 23–32 (2004).
McCluskie, M.J., Weeratna, R.D., Payette, P.J. & Davis, H.L. The potential of CpG oligodeoxynucleotides as mucosal adjuvants. Crit. Rev. Immunol. 21, 103–120 (2001).
Harandi, A.M. & Holmgren, J. CpG DNA as a potent inducer of mucosal immunity: implications for immunoprophylaxis and immunotherapy of mucosal infections. Curr. Opin. Investig. Drugs 5, 141–145 (2004).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Holmgren, J., Czerkinsky, C. Mucosal immunity and vaccines. Nat Med 11 (Suppl 4), S45–S53 (2005). https://doi.org/10.1038/nm1213
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1213
This article is cited by
-
Intranasal immunization of mice with chimera of Salmonella Typhi protein elicits protective intestinal immunity
npj Vaccines (2024)
-
Assessment of the influence of ABO blood groups on oral cholera vaccine immunogenicity in a cholera endemic area in Zambia
BMC Public Health (2023)
-
Multifunctional effects of Lactobacillus sakei HEM 224 on the gastrointestinal tract and airway inflammation
Scientific Reports (2023)
-
Development of a spore-based mucosal vaccine against the bovine respiratory pathogen Mannheimia haemolytica
Scientific Reports (2023)
-
Early mucosal events promote distinct mucosal and systemic antibody responses to live attenuated influenza vaccine
Nature Communications (2023)