Abstract
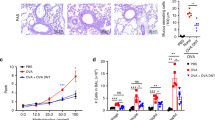
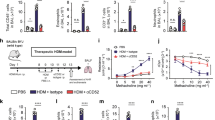
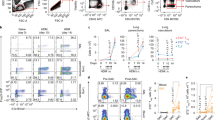
Allergic asthma is a chronic inflammatory disease and despite the introduction of potent and effective drugs, the prevalence has increased substantially over the past few decades1. The explanation that has attracted the most attention is the 'hygiene hypothesis', which suggests that the increase in allergic diseases is caused by a cleaner environment and fewer childhood infections2,3,4. Indeed, certain mycobacterial strains can cause a shift from T-helper cell 2 (Th2) to Th1 immune responses, which may subsequently prevent the development of allergy in mice5,6,7. Although the reconstitution of the balance between Th1 and Th2 is an attractive theory, it is unlikely to explain the whole story, as autoimmune diseases characterized by Th1 responses can also benefit from treatment with mycobacteria and their prevalence has also increased in parallel to allergies8. Here we show that treatment of mice with SRP299, a killed Mycobacterium vaccae-suspension, gives rise to allergen-specific CD4+CD45RBLo regulatory T cells, which confer protection against airway inflammation. This specific inhibition was mediated through interleukin-10 (IL-10) and transforming growth factor-β (TGF-β), as antibodies against IL-10 and TGF-β completely reversed the inhibitory effect of CD4+CD45RBLo T cells. Thus, regulatory T cells generated by mycobacteria treatment may have an essential role in restoring the balance of the immune system to prevent and treat allergic diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Bousquet, J., Jeffery, P.K., Busse, W.W., Johnson, M. & Vignola, A.M. Asthma. From bronchoconstriction to airways inflammation and remodeling. Am. J. Respir. Crit. Care Med. 161, 1720–1745 (2000).
Martinez, F.D. & Holt, P.G. Role of microbial burden in aetiology of allergy and asthma. Lancet 354 (suppl. 2), 12–15 (1999).
Shirakawa, T., Enomoto, T., Shimazu, S. & Hopkin, J.M. The inverse association between tuberculin responses and atopic disorder. Science, 275, 77–79 (1997).
Farooqi, I.S. & Hopkin, J.M. Early childhood infection and atopic disorder. Thorax, 53, 927–932 (1998).
Erb, K.J., Holloway, J.W., Sobeck, A., Moll, H. & Le Gros, G. Infection of mice with Mycobacterium bovis-Bacillus Calmette-Guerin (BCG) suppresses allergen-induced airway eosinophilia. J. Exp. Med. 187, 561–569 (1998).
Herz, U. et al. BCG infection suppresses allergic sensitization and development of increased airway reactivity in an animal model. J. Allergy Clin. Immunol. 102: 867–874 (1998).
Gajewski, T.F. & Fitch, F.W. Anti-proliferative effect of IFN-γ in immune regulation: IFN-γ inhibits the proliferation of Th2 but not Th1 murine helper T-lymphocyte clones. J. Immunol. 140, 4245–4252 (1988).
Stene, L.C. & Nafstad, P. Relation between occurrence of type 1 diabetes and asthma. Lancet 357, 607 (2001).
Wang, C.C. & Rook, G.A.W. Inhibition of an established allergic response to ovalbumin in BALB/c mice by killedMycobacterium vaccae. Immunology 93, 307–313 (1998).
Roncarolo, M.G. & Levings, M.K. The role of different subsets of T regulatory cells in controlling autoimmunity. Curr. Opin. Immunol. 12, 676–683 (2000).
Groux, H. et al. A CD4+ T-cell subset inhibits antigen-specific T-cell responses and prevents colitis. Nature 389, 737–742 (1997).
Zuany-Amorim, C. et al. Modulation by IL-10 of antigen-induced IL-5 generation and CD4+ T lymphocyte and eosinophil infiltration into the mouse peritoneal cavity. J. Immunol. 157, 377–384 (1996).
Maloy, K.J. & Powrie, F. Regulatory T cells in the control of immune pathology. Nature Immunol. 2, 816–822 (2001)
Thornton, A.M. & Shevack, E.M. CD4+CD25+ immunoregulatory T cells suppress polyclonal T-cell activation in vitro by inhibiting interleukin 2 production. J. Exp. Med. 188, 287–296 (1998).
Levings, M.K., Sangregorio, R. & Roncarolo, M.G. Human CD25+CD4+ T regulatory cells suppress naive and memory T-cell proliferation and can be expanded in vitro without loss of function. J. Exp. Med. 193, 1295–1301 (2001).
Roncarolo, M.G., Levings, M.K. & Traversari, K. Differentiation of T regulatory cells by immature dendritic cells. J. Exp. Med. 193, F5–F10 (2001).
Cottrez, F., Hurst, S.D., Coffman, R.L. & Groux, H. T regulatory cells 1 inhibit a Th2–specific response in vivo. J. Immunol. 165, 4848–4853 (2000).
Asseman, C., Mauze, S., Leach, M.W., Coffman, R.L. & Powrie, F. An essential role for interleukin 10 in the function of regulatory T cells that inhibit intestinal inflammation. J. Exp. Med. 190, 995–1004 (1999).
Davies, J.D. et al. CD4+CD45RB low-density cells from untreated mice prevent acute allograft rejection. J. Immunol. 163, 5353–5357 (1999).
Hopkins, J.M. Mycobacterial immunization: agents to limit asthma. in New Drugs for Asthma, Allergy and COPD (eds. Hansel, T.T. & Barnes, P.J.) 226–228 (Karger, Basel, Switzerland, 2001).
Arkwright, P.D. & David, T.J. Intradermal administration of a killed Mycobacterium vaccae suspension (SRL172) is associated with improvement in atopic dermatitis in children with moderate-to-severe disease. J. Allergy Clin. Immunol. 107, 531–534 (2001).
Camporota, L. et al. Effects of intradermal injection of SRL172 (killed Mycobacterium vaccae suspension) on allergen-induced airway response and IL-5 generation by PBMC in asthma. Am. J. Resp. Crit. Care Med. 161, A477 (2000).
Shirtcliffe, P.M. et al. The effect of delipidated deglycolipidated (DDMV) and heat-killed Mycobacterium vaccae in asthma. Am. J. Respir. Crit. Care Med. 163, 1410–1414 (2001).
Zuany-Amorim, C. et al. Requirement for γδ T cells in allergic airway inflammation. Science. 280, 1265–1267 (1998).
Acknowledgements
We thank J. Lee, A. Nicholls and G. Dubois for technical support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
L.R.B., G.B. and G.R. are employed by SR Pharma, the company that produces the heat-killed Mycobacterium vaccae.
Rights and permissions
About this article
Cite this article
Zuany-Amorim, C., Sawicka, E., Manlius, C. et al. Suppression of airway eosinophilia by killed Mycobacterium vaccae-induced allergen-specific regulatory T-cells. Nat Med 8, 625–629 (2002). https://doi.org/10.1038/nm0602-625
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm0602-625
This article is cited by
-
Mycobacterium vaccae Lysate Induces Anti-Allergic Immune Response In Vitro
Bulletin of Experimental Biology and Medicine (2020)
-
Old Friends, immunoregulation, and stress resilience
Pflügers Archiv - European Journal of Physiology (2019)
-
Identification and characterization of a novel anti-inflammatory lipid isolated from Mycobacterium vaccae, a soil-derived bacterium with immunoregulatory and stress resilience properties
Psychopharmacology (2019)
-
Therapeutic Implications of Brain–Immune Interactions: Treatment in Translation
Neuropsychopharmacology (2017)
-
The Microbiota, Immunoregulation, and Mental Health: Implications for Public Health
Current Environmental Health Reports (2016)