Abstract
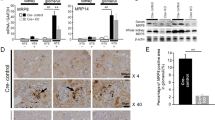
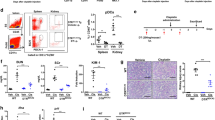
Excess levels of protein in urine (proteinuria) is a hallmark of kidney disease that typically occurs in conjunction with diabetes, hypertension, gene mutations, toxins or infections but may also be of unknown cause (idiopathic)1. Systemic soluble urokinase plasminogen activator receptor (suPAR) is a circulating factor implicated in the onset and progression of chronic kidney disease (CKD)2, such as focal segmental glomerulosclerosis (FSGS)3,4. The cellular source(s) of elevated suPAR associated with future and progressing kidney disease is unclear, but is likely extra-renal, as the pathological uPAR is circulating and FSGS can recur even after a damaged kidney is replaced with a healthy donor organ. Here we report that bone marrow (BM) Gr-1lo immature myeloid cells are responsible for the elevated, pathological levels of suPAR, as evidenced by BM chimera and BM ablation and cell transfer studies. A marked increase of Gr-1lo myeloid cells was commonly found in the BM of proteinuric animals having high suPAR, and these cells efficiently transmit proteinuria when transferred to healthy mice. In accordance with the results seen in suPAR-associated proteinuric animal models, in which kidney damage is caused not by local podocyte-selective injury but more likely by systemic insults, a humanized xenograft model of FSGS resulted in an expansion of Gr-1lo cells in the BM, leading to high plasma suPAR and proteinuric kidney disease. Together, these results identify suPAR as a functional connection between the BM and the kidney, and they implicate BM immature myeloid cells as a key contributor to glomerular dysfunction.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
References
Fogo, A.B. Mechanisms of progression of chronic kidney disease. Pediatr. Nephrol. 22, 2011–2022 (2007).
Hayek, S.S. et al. Soluble urokinase receptor and chronic kidney disease. N. Engl. J. Med. 373, 1916–1925 (2015).
Wei, C. et al. Modification of kidney barrier function by the urokinase receptor. Nat. Med. 14, 55–63 (2008).
Wei, C. et al. Circulating urokinase receptor as a cause of focal segmental glomerulosclerosis. Nat. Med. 17, 952–960 (2011).
Cravedi, P., Kopp, J.B. & Remuzzi, G. Recent progress in the pathophysiology and treatment of FSGS recurrence. Am. J. Transplant. 13, 266–274 (2013).
Winn, M.P. et al. A mutation in the TRPC6 cation channel causes familial focal segmental glomerulosclerosis. Science 308, 1801–1804 (2005).
D'Agati, V.D., Kaskel, F.J. & Falk, R.J. Focal segmental glomerulosclerosis. N. Engl. J. Med. 365, 2398–2411 (2011).
Kitiyakara, C., Eggers, P. & Kopp, J.B. Twenty-one-year trend in ESRD due to focal segmental glomerulosclerosis in the United States. Am. J. Kidney Dis. 44, 815–825 (2004).
Fogo, A.B. Causes and pathogenesis of focal segmental glomerulosclerosis. Nat. Rev. Nephrol. 11, 76–87 (2015).
Gallon, L., Leventhal, J., Skaro, A., Kanwar, Y. & Alvarado, A. Resolution of recurrent focal segmental glomerulosclerosis after retransplantation. N. Engl. J. Med. 366, 1648–1649 (2012).
McCarthy, E.T., Sharma, M. & Savin, V.J. Circulating permeability factors in idiopathic nephrotic syndrome and focal segmental glomerulosclerosis. Clin. J. Am. Soc. Nephrol. 5, 2115–2121 (2010).
Savin, V.J. et al. Circulating factor associated with increased glomerular permeability to albumin in recurrent focal segmental glomerulosclerosis. N. Engl. J. Med. 334, 878–883 (1996).
Thunø, M., Macho, B. & Eugen-Olsen, J. suPAR: the molecular crystal ball. Dis. Markers 27, 157–172 (2009).
Blasi, F. & Carmeliet, P. uPAR: a versatile signalling orchestrator. Nat. Rev. Mol. Cell Biol. 3, 932–943 (2002).
Smith, H.W. & Marshall, C.J. Regulation of cell signalling by uPAR. Nat. Rev. Mol. Cell Biol. 11, 23–36 (2010).
Faul, C. et al. The actin cytoskeleton of kidney podocytes is a direct target of the antiproteinuric effect of cyclosporine A. Nat. Med. 14, 931–938 (2008).
Reiser, J. et al. Induction of B7-1 in podocytes is associated with nephrotic syndrome. J. Clin. Invest. 113, 1390–1397 (2004).
Bertelli, R. et al. LPS nephropathy in mice is ameliorated by IL-2 independently of regulatory T cells activity. PLoS One 9, e111285 (2014).
Shultz, L.D. et al. Human lymphoid and myeloid cell development in NOD/LtSz-scid IL2R gamma null mice engrafted with mobilized human hemopoietic stem cells. J. Immunol. 174, 6477–6489 (2005).
Basu, S., Hodgson, G., Katz, M. & Dunn, A.R. Evaluation of role of G-CSF in the production, survival, and release of neutrophils from bone marrow into circulation. Blood 100, 854–861 (2002).
Liu, F., Wu, H.Y., Wesselschmidt, R., Kornaga, T. & Link, D.C. Impaired production and increased apoptosis of neutrophils in granulocyte colony-stimulating factor receptor-deficient mice. Immunity 5, 491–501 (1996).
Kopp, J.B. et al. Transgenic mice with increased plasma levels of TGF-beta 1 develop progressive renal disease. Lab. Invest. 74, 991–1003 (1996).
Schiffer, M. et al. Apoptosis in podocytes induced by TGF-beta and Smad7. J. Clin. Invest. 108, 807–816 (2001).
Kistler, A.D. et al. Transient receptor potential channel 6 (TRPC6) protects podocytes during complement-mediated glomerular disease. J. Biol. Chem. 288, 36598–36609 (2013).
Hudkins, K.L. et al. BTBR Ob/Ob mutant mice model progressive diabetic nephropathy. J. Am. Soc. Nephrol. 21, 1533–1542 (2010).
Yu, H. et al. Rac1 activation in podocytes induces rapid foot process effacement and proteinuria. Mol. Cell. Biol. 33, 4755–4764 (2013).
Papeta, N. et al. Prkdc participates in mitochondrial genome maintenance and prevents Adriamycin-induced nephropathy in mice. J. Clin. Invest. 120, 4055–4064 (2010).
Alfano, M. et al. Full-length soluble urokinase plasminogen activator receptor down-modulates nephrin expression in podocytes. Sci. Rep. 5, 13647 (2015).
Cathelin, D. et al. Administration of recombinant soluble urokinase receptor per se is not sufficient to induce podocyte alterations and proteinuria in mice. J. Am. Soc. Nephrol. 25, 1662–1668 (2014).
Spinale, J.M. et al. A reassessment of soluble urokinase-type plasminogen activator receptor in glomerular disease. Kidney Int. 87, 564–574 (2015).
Delville, M. et al. A circulating antibody panel for pretransplant prediction of FSGS recurrence after kidney transplantation. Sci. Transl. Med. 6, 256ra136 (2014).
Huang, J. et al. Urinary soluble urokinase receptor levels are elevated and pathogenic in patients with primary focal segmental glomerulosclerosis. BMC Med. 12, 81 (2014).
Holmes, C. & Stanford, W.L. Concise review: stem cell antigen-1: expression, function, and enigma. Stem Cells 25, 1339–1347 (2007).
Shi, X. et al. Toll-like receptor 4/stem cell antigen 1 signaling promotes hematopoietic precursor cell commitment to granulocyte development during the granulopoietic response to Escherichia coli bacteremia. Infect. Immun. 81, 2197–2205 (2013).
Shultz, L.D., Brehm, M.A., Garcia-Martinez, J.V. & Greiner, D.L. Humanized mice for immune system investigation: progress, promise and challenges. Nat. Rev. Immunol. 12, 786–798 (2012).
Brehm, M.A., Shultz, L.D. & Greiner, D.L. Humanized mouse models to study human diseases. Curr. Opin. Endocrinol. Diabetes Obes. 17, 120–125 (2010).
Ito, R., Takahashi, T., Katano, I. & Ito, M. Current advances in humanized mouse models. Cell. Mol. Immunol. 9, 208–214 (2012).
Sellier-Leclerc, A.L. et al. A humanized mouse model of idiopathic nephrotic syndrome suggests a pathogenic role for immature cells. J. Am. Soc. Nephrol. 18, 2732–2739 (2007).
Niu, H. et al. The function of hematopoietic stem cells is altered by both genetic and inflammatory factors in lupus mice. Blood 121, 1986–1994 (2013).
Zhao, J.L. et al. Conversion of danger signals into cytokine signals by hematopoietic stem and progenitor cells for regulation of stress-induced hematopoiesis. Cell Stem Cell 14, 445–459 (2014).
Acknowledgements
We thank A.S. Shaw (Washington University School of Medicine) for the Rac1 transgenic mice; S. Shankland (University of Washington) for providing the nephrotoxic serum (NTS); A. Finnegan (Rush University Medical Center) for reagents and advice; B. Samelko (née Tryniszewska) (Rush University Medical Center) for technical assistance; and S. Mangos (Rush University Medical Center) for help with manuscript revisions. This research was supported by National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) R01DK101350 (J.R.), R01DK106051 (J.R.), R01DK107984 (V.G.) and R01DK084195 (V.G.).
Author information
Authors and Affiliations
Contributions
E.H. designed and performed experiments, and wrote the paper; I.F., J.L., and Y.C. performed animal experiments; N.J.T. generated electron micrographs; A.Z. and J.L. contributed to xenotransplantation experiment; C.W., M.T., S.W., V.P., S.S.H., C.O., M.B., V.G., S.S., D.B.S., and D.T.S. contributed to experiments; J.R. designed and supervised the study, and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
E.H., C.W., S.S., and J.R. are inventors on pending and issued patents related to anti-proteinuric therapies. They stand to gain royalties from present and future commercialization. J.R. and S.S. are also co-founders and advisors to TRISAQ, a biotechnology company. The remaining authors report no conflicts.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–12 (PDF 18323 kb)
Rights and permissions
About this article
Cite this article
Hahm, E., Wei, C., Fernandez, I. et al. Bone marrow-derived immature myeloid cells are a main source of circulating suPAR contributing to proteinuric kidney disease. Nat Med 23, 100–106 (2017). https://doi.org/10.1038/nm.4242
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.4242
This article is cited by
-
Predictive value of suPAR in AKI: a systematic review and meta-analysis
Clinical and Experimental Nephrology (2023)
-
Gut Microbiota and Its Role in the Brain-Gut-Kidney Axis in Hypertension
Current Hypertension Reports (2023)
-
Role of Soluble Urokinase-Type Plasminogen Activator Receptor in Cardiovascular Disease
Current Cardiology Reports (2023)
-
SuPAR mediates viral response proteinuria by rapidly changing podocyte function
Nature Communications (2023)
-
Soluble urokinase plasminogen activator receptor is associated with short-term mortality and enhanced reactive oxygen species production in acute-on-chronic liver failure
BMC Gastroenterology (2021)