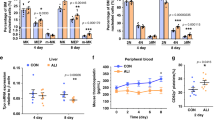
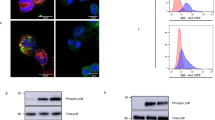
Abstract
The hepatic Ashwell-Morell receptor (AMR) can bind and remove desialylated platelets. Here we demonstrate that platelets become desialylated as they circulate and age in blood. Binding of desialylated platelets to the AMR induces hepatic expression of thrombopoietin (TPO) mRNA and protein, thereby regulating platelet production. Endocytic AMR controls TPO expression through Janus kinase 2 (JAK2) and the acute phase response signal transducer and activator of transcription 3 (STAT3) in vivo and in vitro. Recognition of this newly identified physiological feedback mechanism illuminates the pathophysiology of platelet diseases, such as essential thrombocythemia and immune thrombocytopenia, and contributes to an understanding of the mechanisms of thrombocytopenia observed with JAK1/2 inhibition.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Kaushansky, K. The molecular mechanisms that control thrombopoiesis. J. Clin. Invest. 115, 3339–3347 (2005).
Kaushansky, K. Determinants of platelet number and regulation of thrombopoiesis. Hematology (Am. Soc. Hematol. Educ. Program) 2009, 147–152 (2009).
Kuter, D.J. Thrombopoietin and thrombopoietin mimetics in the treatment of thrombocytopenia. Annu. Rev. Med. 60, 193–206 (2009).
Kuter, D.J. & Rosenberg, R.D. The reciprocal relationship of thrombopoietin (Mpl ligand) to changes in the platelet mass during busulfan-induced thrombocytopenia in the rabbit. Blood 85, 2720–2730 (1995).
Cohen-Solal, K. et al. Constitutive expression of Mpl ligand transcripts during thrombocytopenia or thrombocytosis. Blood 88, 2578–2584 (1996).
Fielder, P.J. et al. Regulation of thrombopoietin levels by c-mpl-mediated binding to platelets. Blood 87, 2154–2161 (1996).
Shinjo, K. et al. Serum thrombopoietin levels in patients correlate inversely with platelet counts during chemotherapy-induced thrombocytopenia. Leukemia 12, 295–300 (1998).
Engel, C., Loeffler, M., Franke, H. & Schmitz, S. Endogenous thrombopoietin serum levels during multicycle chemotherapy. Br. J. Haematol. 105, 832–838 (1999).
McCarty, J.M., Sprugel, K., Fox, N., Sabath, D. & Kaushansky, K. Murine thrombopoietin mRNA levels are modulated by platelet count. Blood 86, 3668–3675 (1995).
Sungaran, R., Markovic, B. & Chong, B. Localization and regulation of thrombopoietin mRNa expression in human kidney, liver, bone marrow, and spleen using in situ hybridization. Blood 89, 101–107 (1997).
Qian, S., Fu, F., Li, W., Chen, Q. & de Sauvage, F. Primary role of the liver in thrombopoietin production shown by tissue-specific knockout. Blood 92, 2189–2191 (1998).
Wolber, E.M., Fandrey, J., Frackowski, U. & Jelkmann, W. Hepatic thrombopoietin mRNA is increased in acute inflammation. Thromb. Haemost. 86, 1421–1424 (2001).
McIntosh, B. & Kaushansky, K. Transcriptional regulation of bone marrow thrombopoietin by platelet proteins. Exp. Hematol. 36, 799–806 (2008).
Wolber, E.M. & Jelkmann, W. Interleukin-6 increases thrombopoietin production in human hepatoma cells HepG2 and Hep3B. J. Interferon Cytokine Res. 20, 499–506 (2000).
Kaser, A. et al. Interleukin-6 stimulates thrombopoiesis through thrombopoietin: role in inflammatory thrombocytosis. Blood 98, 2720–2725 (2001).
Stone, R.L. et al. Paraneoplastic thrombocytosis in ovarian cancer. N. Engl. J. Med. 366, 610–618 (2012).
Rumjantseva, V. & Hoffmeister, K.M. Novel and unexpected clearance mechanisms for cold platelets. Transfus. Apher. Sci. 42, 63–70 (2010).
Hoffmeister, K.M. The role of lectins and glycans in platelet clearance. J. Thromb. Haemost. 9 (suppl. 1), 35–43 (2011).
Rumjantseva, V. et al. Dual roles for hepatic lectin receptors in the clearance of chilled platelets. Nat. Med. 15, 1273–1280 (2009).
Sørensen, A.L. et al. Role of sialic acid for platelet life span: exposure of β-galactose results in the rapid clearance of platelets from the circulation by asialoglycoprotein receptor–expressing liver macrophages and hepatocytes. Blood 114, 1645–1654 (2009).
Grewal, P.K. et al. Inducing host protection in pneumococcal sepsis by preactivation of the Ashwell-Morell receptor. Proc. Natl. Acad. Sci. USA 110, 20218–20223 (2013).
Grewal, P.K. et al. The Ashwell receptor mitigates the lethal coagulopathy of sepsis. Nat. Med. 14, 648–655 (2008).
Grewal, P.K. The Ashwell-Morell receptor. Methods Enzymol. 479, 223–241 (2010).
Levine, R.L. & Gilliland, D.G. Myeloproliferative disorders. Blood 112, 2190–2198 (2008).
LaFave, L.M. & Levine, R.L. JAK2 the future: therapeutic strategies for JAK-dependent malignancies. Trends Pharmacol. Sci. 33, 574–582 (2012).
Horikawa, Y. et al. Markedly reduced expression of platelet c-mpl receptor in essential thrombocythemia. Blood 90, 4031–4038 (1997).
Moliterno, A.R., Hankins, W.D. & Spivak, J.L. Impaired expression of the thrombopoietin receptor by platelets from patients with polycythemia vera. N. Engl. J. Med. 338, 572–580 (1998).
Cohen-Solal, K., Vitrat, N., Titeux, M., Vainchenker, W. & Wendling, F. High-level expression of Mpl in platelets and megakaryocytes is independent of thrombopoietin. Blood 93, 2859–2866 (1999).
Karpatkin, S. & Shulman, S. Asialo platelets enhance thrombopoiesis. Trans. Assoc. Am. Physicians 93, 244–250 (1980).
Mason, K.D. et al. Programmed anuclear cell death delimits platelet life span. Cell 128, 1173–1186 (2007).
Josefsson, E.C. et al. Megakaryocytes possess a functional intrinsic apoptosis pathway that must be restrained to survive and produce platelets. J. Exp. Med. 208, 2017–2031 (2011).
Debrincat, M.A. et al. Mcl-1 and Bcl-xL co-ordinately regulate megakaryocyte survival. Blood 119, 5850–5858 (2012).
Zhang, H. et al. Bcl-2 family proteins are essential for platelet survival. Cell Death Differ. 14, 943–951 (2007).
Rowley, J.W. et al. Genome-wide RNA-seq analysis of human and mouse platelet transcriptomes. Blood 118, e101–e111 (2011).
de Graaf, C.A. & Metcalf, D. Thrombopoietin and hematopoietic stem cells. Cell Cycle 10, 1582–1589 (2011).
Ng, A.P. et al. Mpl expression on megakaryocytes and platelets is dispensable for thrombopoiesis but essential to prevent myeloproliferation. Proc. Natl. Acad. Sci. USA 111, 5884–5889 (2014).
Mouthon, M.A., Vandamme, M., Gourmelon, P., Vainchenker, W. & Wendling, F. Preferential liver irradiation enhances hematopoiesis through a thrombopoietin-independent mechanism. Radiat. Res. 152, 390–397 (1999).
Kosugi, S. et al. Circulating thrombopoietin level in chronic immune thrombocytopenic purpura. Br. J. Haematol. 93, 704–706 (1996).
Ichikawa, N. et al. Regulation of serum thrombopoietin levels by platelets and megakaryocytes in patients with aplastic anaemia and idiopathic thrombocytopenic purpura. Thromb. Haemost. 76, 156–160 (1996).
Hiyoyama, K. et al. Increased serum levels of thrombopoietin in patients with thrombotic thrombocytopenic purpura, idiopathic thrombocytopenic purpura, or disseminated intravascular coagulation. Blood Coagul. Fibrinolysis 8, 345–349 (1997).
Wang, J.C. et al. Blood thrombopoietin levels in clonal thrombocytosis and reactive thrombocytosis. Am. J. Med. 104, 451–455 (1998).
Griesshammer, M. et al. High levels of thrombopoietin in sera of patients with essential thrombocythemia: cause or consequence of abnormal platelet production? Ann. Hematol. 77, 211–215 (1998).
Shivdasani, R.A. et al. Transcription factor NF-E2 is required for platelet formation independent of the actions of thrombopoietin/MGDF in megakaryocyte development. Cell 81, 695–704 (1995).
Eulenfeld, R. et al. Interleukin-6 signalling: more than Jaks and STATs. Eur. J. Cell Biol. 91, 486–495 (2012).
Fallon, R.J., Danaher, M. & Saxena, A. The asialoglycoprotein receptor is associated with a tyrosine kinase in HepG2 cells. J. Biol. Chem. 269, 26626–26629 (1994).
Park, S.O. et al. Conditional deletion of Jak2 reveals an essential role in hematopoiesis throughout mouse ontogeny: implications for Jak2 inhibition in humans. PLoS ONE 8, e59675 (2013).
Grisouard, J., Hao-Shen, H., Dirnhofer, S., Wagner, K.U. & Skoda, R.C. Selective deletion of Jak2 in adult mouse hematopoietic cells leads to lethal anemia and thrombocytopenia. Haematologica 99, e52–e54 (2014).
Jansen, A.J. et al. Desialylation accelerates platelet clearance following refrigeration and initiates GPIbα metalloproteinase-mediated cleavage in mice. Blood 119, 1263–1273 (2012).
Hoffmeister, K.M. et al. The clearance mechanism of chilled blood platelets. Cell 112, 87–97 (2003).
Grozovsky, R. et al. Type 2 deiodinase expression is induced by peroxisomal proliferator-activated receptor-γ agonists in skeletal myocytes. Endocrinology 150, 1976–1983 (2009).
Bergmeier, W. et al. Metalloproteinase inhibitors improve the recovery and hemostatic function of in vitro–aged or –injured mouse platelets. Blood 102, 4229–4235 (2003).
Jurak Begonja, A., Hoffmeister, K., Hartwig, J. & Falet, H. FlnA-null megakaryocytes prematurely release large and fragile platelets that circulate poorly. Blood 118, 2285–2295 (2011).
Acknowledgements
We thank M. Adelman, S. Yang and R. Abdu for technical assistance. We are grateful to M. Sola-Visner and T. Stossel (Harvard Medical School) for helpful discussions and to W. Tong (University of Pennsylvania), for the anti-Mpl antibody. This work was supported by US National Institutes of Health grants R01 HL089224, P01 HL059561 and the Program of Excellence in Glycosciences (P01 HL107146). We thank F. Maigen for statistical guidance and expertise. We dedicate this work to the memory of Gilbert Ashwell (1916–2014) and Anatol Morell (1913–2009).
Author information
Authors and Affiliations
Contributions
R.G. designed and performed research, collected, analyzed and interpreted results, and assisted with writing the manuscript. A.J.B. analyzed and interpreted results. K.L. performed splenectomy. G.V. interpreted results. J.H.H. analyzed and interpreted results and assisted with writing the manuscript. H.F. and K.M.H. designed research, analyzed and interpreted results, and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 and Supplementary Table 1 (PDF 7334 kb)
Rights and permissions
About this article
Cite this article
Grozovsky, R., Begonja, A., Liu, K. et al. The Ashwell-Morell receptor regulates hepatic thrombopoietin production via JAK2-STAT3 signaling. Nat Med 21, 47–54 (2015). https://doi.org/10.1038/nm.3770
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3770
This article is cited by
-
Reference guide for the diagnosis of adult primary immune thrombocytopenia, 2023 edition
International Journal of Hematology (2024)
-
Recent progress in ITP treatment
International Journal of Hematology (2023)
-
The GPIb-IX complex on platelets: insight into its novel physiological functions affecting immune surveillance, hepatic thrombopoietin generation, platelet clearance and its relevance for cancer development and metastasis
Experimental Hematology & Oncology (2022)
-
Thrombopoietin Receptor Agonist in Pregnancy: a Case For Romiplostim in Refractory Immune Thrombocytopenic Purpura
SN Comprehensive Clinical Medicine (2022)
-
Platelet desialylation and TFH cells–the novel pathway of immune thrombocytopenia
Experimental Hematology & Oncology (2021)