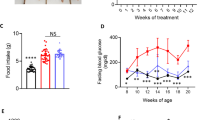
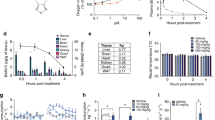
Abstract
Type 2 diabetes (T2D) has reached an epidemic level globally. Most current treatments ameliorate the hyperglycemic symptom of the disease but are not effective in correcting its underlying cause. One important causal factor of T2D is ectopic accumulation of lipids in metabolically sensitive organs such as liver and muscle. Mitochondrial uncoupling, which reduces cellular energy efficiency and increases lipid oxidation, is an appealing therapeutic strategy. The challenge, however, is to discover safe mitochondrial uncouplers for practical use. Niclosamide is an anthelmintic drug approved by the US Food and Drug Administration that uncouples the mitochondria of parasitic worms. Here we show that niclosamide ethanolamine salt (NEN) uncouples mammalian mitochondria at upper nanomolar concentrations. Oral NEN increases energy expenditure and lipid metabolism in mice. It is also efficacious in preventing and treating hepatic steatosis and insulin resistance induced by a high-fat diet. Moreover, it improves glycemic control and delays disease progression in db/db mice. Given the well-documented safety profile of NEN, our study provides a potentially new and practical pharmacological approach for treating T2D.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Boyle, J.P., Thompson, T.J., Gregg, E.W., Barker, L.E. & Williamson, D.F. Projection of the year 2050 burden of diabetes in the US adult population: dynamic modeling of incidence, mortality, and prediabetes prevalence. Popul. Health Metr. 8, 29 (2010).
US Center for Disease Control and Prevention. National diabetes statistics report, 2014. Diabetes Public Health Resource http://www.cdc.gov/diabetes/pubs/statsreport14.htm (2014).
Qaseem, A., Humphrey, L.L., Sweet, D.E., Starkey, M. & Shekelle, P. Oral pharmacologic treatment of type 2 diabetes mellitus: a clinical practice guideline from the American College of Physicians. Ann. Intern. Med. 156, 218–231 (2012).
Nathan, D.M., et al. Medical management of hyperglycemia in type 2 diabetes: a consensus algorithm for the initiation and adjustment of therapy: a consensus statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care 32, 193–203 (2009).
Kahn, S.E., Hull, R.L. & Utzschneider, K.M. Mechanisms linking obesity to insulin resistance and type 2 diabetes. Nature 444, 840–846 (2006).
Muoio, D.M. & Newgard, C.B. Obesity-related derangements in metabolic regulation. Annu. Rev. Biochem. 75, 367–401 (2006).
Randle, P.J. Regulatory interactions between lipids and carbohydrates: the glucose fatty acid cycle after 35 years. Diabetes Metab. Rev. 14, 263–283 (1998).
Samuel, V.T., Petersen, K.F. & Shulman, G.I. Lipid-induced insulin resistance: unravelling the mechanism. Lancet 375, 2267–2277 (2010).
An, J. et al. Hepatic expression of malonyl-CoA decarboxylase reverses muscle, liver and whole-animal insulin resistance. Nat. Med. 10, 268–274 (2004).
Chavez, J.A. & Summers, S.A. A ceramide-centric view of insulin resistance. Cell Metab. 15, 585–594 (2012).
Griffin, M.E. et al. Free fatty acid–induced insulin resistance is associated with activation of protein kinase Cθ and alterations in the insulin signaling cascade. Diabetes 48, 1270–1274 (1999).
Holland, W.L. et al. Inhibition of ceramide synthesis ameliorates glucocorticoid-, saturated-fat–, and obesity-induced insulin resistance. Cell Metab. 5, 167–179 (2007).
Samuel, V.T. & Shulman, G.I. Mechanisms for insulin resistance: common threads and missing links. Cell 148, 852–871 (2012).
Zhang, Y. et al. Positional cloning of the mouse obese gene and its human homologue. Nature 372, 425–432 (1994).
Pelleymounter, M.A. et al. Effects of the obese gene product on body weight regulation in ob/ob mice. Science 269, 540–543 (1995).
Uysal, K.T., Wiesbrock, S.M., Marino, M.W. & Hotamisligil, G.S. Protection from obesity-induced insulin resistance in mice lacking TNF-α function. Nature 389, 610–614 (1997).
Yamauchi, T. et al. The fat-derived hormone adiponectin reverses insulin resistance associated with both lipoatrophy and obesity. Nat. Med. 7, 941–946 (2001).
Steppan, C.M. et al. The hormone resistin links obesity to diabetes. Nature 409, 307–312 (2001).
Yuan, M. et al. Reversal of obesity- and diet-induced insulin resistance with salicylates or targeted disruption of Ikkβ. Science 293, 1673–1677 (2001).
Yang, Q. et al. Serum retinol binding protein 4 contributes to insulin resistance in obesity and type 2 diabetes. Nature 436, 356–362 (2005).
Henry, R.R., Wallace, P. & Olefsky, J.M. Effects of weight loss on mechanisms of hyperglycemia in obese non–insulin-dependent diabetes mellitus. Diabetes 35, 990–998 (1986).
Kantartzis, K. et al. High cardiorespiratory fitness is an independent predictor of the reduction in liver fat during a lifestyle intervention in non-alcoholic fatty liver disease. Gut 58, 1281–1288 (2009).
Perseghin, G. et al. Increased glucose transport-phosphorylation and muscle glycogen synthesis after exercise training in insulin-resistant subjects. N. Engl. J. Med. 335, 1357–1362 (1996).
Petersen, K.F. et al. Reversal of nonalcoholic hepatic steatosis, hepatic insulin resistance, and hyperglycemia by moderate weight reduction in patients with type 2 diabetes. Diabetes 54, 603–608 (2005).
Terada, H. Uncouplers of oxidative phosphorylation. Environ. Health Perspect. 87, 213–218 (1990).
Nedergaard, J., Ricquier, D. & Kozak, L.P. Uncoupling proteins: current status and therapeutic prospects. EMBO Rep. 6, 917–921 (2005).
Si, Y., Shi, H. & Lee, K. Metabolic flux analysis of mitochondrial uncoupling in 3T3–L1 adipocytes. PLoS ONE 4, e7000 (2009).
Tseng, Y.H., Cypess, A.M. & Kahn, C.R. Cellular bioenergetics as a target for obesity therapy. Nat. Rev. Drug Discov. 9, 465–482 (2010).
Harper, J.A., Dickinson, K. & Brand, M.D. Mitochondrial uncoupling as a target for drug development for the treatment of obesity. Obes. Rev. 2, 255–265 (2001).
Harper, M.E., Green, K. & Brand, M.D. The efficiency of cellular energy transduction and its implications for obesity. Annu. Rev. Nutr. 28, 13–33 (2008).
Clapham, J.C. et al. Mice overexpressing human uncoupling protein-3 in skeletal muscle are hyperphagic and lean. Nature 406, 415–418 (2000).
Kopecky, J., Clarke, G., Enerback, S., Spiegelman, B. & Kozak, L.P. Expression of the mitochondrial uncoupling protein gene from the aP2 gene promoter prevents genetic obesity. J. Clin. Invest. 96, 2914–2923 (1995).
Li, B. et al. Skeletal muscle respiratory uncoupling prevents diet-induced obesity and insulin resistance in mice. Nat. Med. 6, 1115–1120 (2000).
Neschen, S. et al. Uncoupling protein 1 expression in murine skeletal muscle increases AMPK activation, glucose turnover, and insulin sensitivity in vivo. Physiol. Genomics 33, 333–340 (2008).
Ishigaki, Y. et al. Dissipating excess energy stored in the liver is a potential treatment strategy for diabetes associated with obesity. Diabetes 54, 322–332 (2005).
Choi, C.S. et al. Overexpression of uncoupling protein 3 in skeletal muscle protects against fat-induced insulin resistance. J. Clin. Invest. 117, 1995–2003 (2007).
Amara, C.E. et al. Mild mitochondrial uncoupling impacts cellular aging in human muscles in vivo. Proc. Natl. Acad. Sci. USA 104, 1057–1062 (2007).
Maragos, W.F. & Korde, A.S. Mitochondrial uncoupling as a potential therapeutic target in acute central nervous system injury. J. Neurochem. 91, 257–262 (2004).
Mattiasson, G. et al. Uncoupling protein-2 prevents neuronal death and diminishes brain dysfunction after stroke and brain trauma. Nat. Med. 9, 1062–1068 (2003).
Parascandola, J. Dinitrophenol and bioenergetics: an historical perspective. Mol. Cell. Biochem. 5, 69–77 (1974).
Perry, R.J. et al. Reversal of hypertriglyceridemia, fatty liver disease, and insulin resistance by a liver-targeted mitochondrial uncoupler. Cell Metab. 18, 740–748 (2013).
Frayha, G.J., Smyth, J.D., Gobert, J.G. & Savel, J. The mechanisms of action of antiprotozoal and anthelmintic drugs in man. Gen. Pharmacol. 28, 273–299 (1997).
Sheth, U.K. Mechanisms of anthelmintic action. Prog. Drug Res. 19, 147–157 (1975).
Weinbach, E.C. & Garbus, J. Mechanism of action of reagents that uncouple oxidative phosphorylation. Nature 221, 1016–1018 (1969).
Andrews, P., Thyssen, J. & Lorke, D. The biology and toxicology of molluscicides, Bayluscide. Pharmc. Ther. 19, 245–295 (1982).
Hecht, G. & Gloxhuber, C. Tolerance to 2′,5-dichloro-4-nitrosalicylanilide ethanolamine salt. Z. Tropenmed. Parasitol. 13, 1–8 (1962).
Owen, M.R., Doran, E. & Halestrap, A.P. Evidence that metformin exerts its anti-diabetic effects through inhibition of complex 1 of the mitochondrial respiratory chain. Biochem. J. 348, 607–614 (2000).
Misbin, R.I. et al. Lactic acidosis in patients with diabetes treated with metformin. N. Engl. J. Med. 338, 265–266 (1998).
Kobayashi, K. et al. The db/db mouse, a model for diabetic dyslipidemia: molecular characterization and effects of Western diet feeding. Metabolism 49, 22–31 (2000).
Samuel, V.T. et al. Mechanism of hepatic insulin resistance in non-alcoholic fatty liver disease. J. Biol. Chem. 279, 32345–32353 (2004).
Fryer, L.G., Parbu-Patel, A. & Carling, D. The anti-diabetic drugs rosiglitazone and metformin stimulate AMP-activated protein kinase through distinct signaling pathways. J. Biol. Chem. 277, 25226–25232 (2002).
Inoki, K., Zhu, T. & Guan, K.L. TSC2 mediates cellular energy response to control cell growth and survival. Cell 115, 577–590 (2003).
Zhou, G. et al. Role of AMP-activated protein kinase in mechanism of metformin action. J. Clin. Invest. 108, 1167–1174 (2001).
Chen, M. et al. The anti-helminthic niclosamide inhibits Wnt/Frizzled1 signaling. Biochemistry 48, 10267–10274 (2009).
Osada, T. et al. Antihelminth compound niclosamide downregulates Wnt signaling and elicits antitumor responses in tumors with activating APC mutations. Cancer Res. 71, 4172–4182 (2011).
Ren, X. et al. Identification of niclosamide as a new small-molecule inhibitor of the STAT3 signaling pathway. ACS Med. Chem. Lett. 1, 454–459 (2010).
Fullerton, M.D. et al. Single phosphorylation sites in Acc1 and Acc2 regulate lipid homeostasis and the insulin-sensitizing effects of metformin. Nat. Med. 19, 1649–1654 (2013).
Madiraju, A.K. et al. Metformin suppresses gluconeogenesis by inhibiting mitochondrial glycerophosphate dehydrogenase. Nature 510, 542–546 (2014).
Hollingworth, R.M. in Handbook of Pesticide Toxicology Vol. 2, 2nd edn. (ed. Krieger, R.) 1225–1247 (Academic Press, San Diego, California, 2001).
Swan, G.E. The pharmacology of halogenated salicylanilides and their anthelmintic use in animals. J. S. Afr. Vet. Assoc. 70, 61–70 (1999).
Frezza, C., Cipolat, S. & Scorrano, L. Organelle isolation: functional mitochondria from mouse liver, muscle and cultured fibroblasts. Nat. Protoc. 2, 287–295 (2007).
Trounce, I.A., Kim, Y.L., Jun, A.S. & Wallace, D.C. Assessment of mitochondrial oxidative phosphorylation in patient muscle biopsies, lymphoblasts, and transmitochondrial cell lines. Methods Enzymol. 264, 484–509 (1996).
Muoio, D.M., Seefeld, K., Witters, L.A. & Coleman, R.A. AMP-activated kinase reciprocally regulates triacylglycerol synthesis and fatty acid oxidation in liver and muscle: evidence that sn-glycerol-3-phosphate acyltransferase is a novel target. Biochem. J. 338, 783–791 (1999).
Zhang, Y. et al. Adipose-specific deletion of autophagy-related gene 7 (atg7) in mice reveals a role in adipogenesis. Proc. Natl. Acad. Sci. USA 106, 19860–19865 (2009).
Prince, A., Zhang, Y., Croniger, C. & Puchowicz, M. Oxidative metabolism: glucose versus ketones. Adv. Exp. Med. Biol. 789, 323–328 (2013).
Chang, Y.-W. et al. Pharmacokinetics of anti–SARS-CoV agent niclosamide and its analogs in rats. J. Food Drug Anal. 14, 329–333 (2006).
Kim, H.J. et al. Differential effects of interleukin-6 and -10 on skeletal muscle and liver insulin action in vivo. Diabetes 53, 1060–1067 (2004).
Yu, C. et al. Mechanism by which fatty acids inhibit insulin activation of insulin receptor substrate-1 (IRS-1)-associated phosphatidylinositol 3-kinase activity in muscle. J. Biol. Chem. 277, 50230–50236 (2002).
Acknowledgements
We thank M. Kahn at the Yale University School of Medicine for the analysis of hepatocellular DAG and ceramide contents. We thank the US National Mouse Metabolic Phenotyping Centers (MMPC) at various sites for the various metabolic studies: C. Croniger and L. Wang at Case Western Reserve University (U24 DK076174) for the metabolic cage experiments; J. Kim and D.Y. Jung at the University of Massachusetts (U24 DK093000) for the hyperinsulinemic-euglycemic clamp studies and glycogen synthesis rate determination; and S.S. Wirth and J. Graham at University of California, Davis (U24 DK092993) for determination of glycated hemoglobin (HbA1c) in db/db mice. We also thank W. Hu and Y. Zhao at Rutgers University for assistance with the quantitative real-time PCR analyses and R. Patel at the Rutgers Core Facility for technical assistance with electron microscopy studies. H.T., Y.Z., X.Z. and S.J. are supported by the US National Institutes of Health (R01AG030081 and R01CA116088) and Mito Biopharm, LLC. G.I.S. is supported by the US National Institutes of Health (R24 DK085638, P30 DK45735 and U24 DK059635) and the Novo Nordisk Foundation for Basic Metabolic Research.
Author information
Authors and Affiliations
Contributions
H.T. and S.J. designed the experiments, analyzed the data and wrote the manuscript. H.T. conducted most of the experiments. Y.Z. conducted the mitochondrial oxygen consumption assay and contributed to the quantitative PCR analyses and other in vivo studies. X.Z. contributed to the discussion and design of some experiments. G.I.S. designed and supervised the analyses of hepatic lipid metabolites and contributed to revision of the manuscript. S.J. conceived and directed the project.
Corresponding author
Ethics declarations
Competing interests
S.J. is a founder of Mito BioPharm, which has licensed the patents surrounding the development of chemical mitochondrial uncouplers, including niclosamide ethanolamine (described here), for treating metabolic diseases.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 and Supplementary Table 1. (PDF 4372 kb)
Rights and permissions
About this article
Cite this article
Tao, H., Zhang, Y., Zeng, X. et al. Niclosamide ethanolamine–induced mild mitochondrial uncoupling improves diabetic symptoms in mice. Nat Med 20, 1263–1269 (2014). https://doi.org/10.1038/nm.3699
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3699
This article is cited by
-
Carbonyl cyanide-4-(trifluoromethoxy)phenylhydrazone-induced toxicities in rats: comparative study with other mitochondrial uncouplers (2,4-dinitrophenol, OPC-163493 and tolcapone)
Toxicological Research (2023)
-
Activation of UCP2 by anethole trithione suppresses neuroinflammation after intracerebral hemorrhage
Acta Pharmacologica Sinica (2022)
-
Approaches to Mitigate Mitochondrial Dysfunction in Sensorineural Hearing Loss
Annals of Biomedical Engineering (2022)
-
Niclosamide downregulates LOX-1 expression in mouse vascular smooth muscle cells and changes the composition of atherosclerotic plaques in ApoE−/− mice
Heart and Vessels (2022)
-
RAS activation induces synthetic lethality of MEK inhibition with mitochondrial oxidative metabolism in acute myeloid leukemia
Leukemia (2022)