Abstract
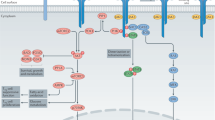
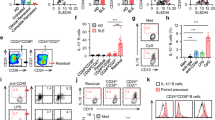
Interleukin-10 (IL-10)-producing regulatory B (Breg) cells suppress autoimmune disease, and increased numbers of Breg cells prevent host defense to infection and promote tumor growth and metastasis by converting resting CD4+ T cells to regulatory T (Treg) cells. The mechanisms mediating the induction and development of Breg cells remain unclear. Here we show that IL-35 induces Breg cells and promotes their conversion to a Breg subset that produces IL-35 as well as IL-10. Treatment of mice with IL-35 conferred protection from experimental autoimmune uveitis (EAU), and mice lacking IL-35 (p35 knockout (KO) mice) or defective in IL-35 signaling (IL-12Rβ2 KO mice) produced less Breg cells endogenously or after treatment with IL-35 and developed severe uveitis. Adoptive transfer of Breg cells induced by recombinant IL-35 suppressed EAU when transferred to mice with established disease, inhibiting pathogenic T helper type 17 (TH17) and TH1 cells while promoting Treg cell expansion. In B cells, IL-35 activates STAT1 and STAT3 through the IL-35 receptor comprising the IL-12Rβ2 and IL-27Rα subunits. As IL-35 also induced the conversion of human B cells into Breg cells, these findings suggest that IL-35 may be used to induce autologous Breg and IL-35+ Breg cells and treat autoimmune and inflammatory disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Change history
30 May 2014
In the version of this article initially published online, there were several errors in the text and figures. Two authors' names were incorrect: Monika Dolinska should have been Monika B. Dolinska and Yuri V. Sergeey should have been Yuri V. Sergeev. Mann-Whitney was misspelled as Mann-Whiney in the legends of Figures 3 and 4 and in the Online Methods (Statistical analyses section). In Figure 4b, Il-10− should have been IL-10−. In Figure 4i, the black key label was PMA + rIL-35 when it should have been PMA + hIL-35, and in the legend for Figure 4h,i, rIL-35 should have been hIL-35. In Figure 5e, IL-0 KO should have been IL-10 KO. In Figure 5f, IL-35 should have been rIL-35. Asterisks indicating statistical significance were missing in Figure 5d and Figure 6b. In Figure 6a, IL27Rα should have been Il27ra. In Figure 6e, on the x axis the second IL-12Rβ2 should have been IL-27Rα. In the Online Methods (Immunoprecipitation and western blot analysis section), IL-12Rb1 and IL-12Rb2 should have been IL-12Rβ1 and IL-12Rβ2. These errors have been corrected for the print, PDF and HTML versions of this article.
References
Edwards, J.C. et al. Efficacy of B cell–targeted therapy with rituximab in patients with rheumatoid arthritis. N. Engl. J. Med. 350, 2572–2581 (2004).
Hu, C.Y. et al. Treatment with CD20-specific antibody prevents and reverses autoimmune diabetes in mice. J. Clin. Invest. 117, 3857–3867 (2007).
Yanaba, K. et al. B-lymphocyte contributions to human autoimmune disease. Immunol. Rev. 223, 284–299 (2008).
Bouaziz, J.D. et al. Therapeutic B cell depletion impairs adaptive and autoreactive CD4+ T cell activation in mice. Proc. Natl. Acad. Sci. USA 104, 20878–20883 (2007).
Hamaguchi, Y. Molecular mechanisms of B lymphocyte depletion by CD20 immunotherapy. Nihon Rinsho Meneki Gakkai Kaishi 32, 29–34 (2009).
Pers, J.O. et al. B cell depletion and repopulation in autoimmune diseases. Clin. Rev. Allergy Immunol. 34, 50–55 (2008).
Bouaziz, J.D., Yanaba, K. & Tedder, T.F. Regulatory B cells as inhibitors of immune responses and inflammation. Immunol. Rev. 224, 201–214 (2008).
Yanaba, K. et al. A regulatory B cell subset with a unique CD1dhiCD5+ phenotype controls T cell–dependent inflammatory responses. Immunity 28, 639–650 (2008).
Mauri, C. & Bosma, A. Immune regulatory function of B cells. Annu. Rev. Immunol. 30, 221–241 (2012).
Tadmor, T., Zhang, Y., Cho, H.M., Podack, E.R. & Rosenblatt, J.D. The absence of B lymphocytes reduces the number and function of T-regulatory cells and enhances the anti-tumor response in a murine tumor model. Cancer Immunol. Immunother. 60, 609–619 (2011).
Schioppa, T. et al. B regulatory cells and the tumor-promoting actions of TNF-a during squamous carcinogenesis. Proc. Natl. Acad. Sci. USA 108, 10662–10667 (2011).
Mauri, C. & Ehrenstein, M.R. The 'short' history of regulatory B cells. Trends Immunol. 29, 34–40 (2008).
Berthelot, J.M. et al. Regulatory B cells play a key role in immune system balance. Joint Bone Spine 80, 18–22 (2013).
Yoshizaki, A. et al. Regulatory B cells control T cell autoimmunity through IL-21–dependent cognate interactions. Nature 491, 264–268 (2012).
Rafei, M. et al. A granulocyte-macrophage colony-stimulating factor and interleukin-15 fusokine induces a regulatory B cell population with immune suppressive properties. Nat. Med. 15, 1038–1045 (2009).
Collison, L.W. et al. The inhibitory cytokine IL-35 contributes to regulatory T cell function. Nature 450, 566–569 (2007).
Collison, L.W. et al. IL-35–mediated induction of a potent regulatory T cell population. Nat. Immunol. 11, 1093–1101 (2010).
Hunter, C.A. New IL-12-family members: IL-23 and IL-27, cytokines with divergent functions. Nat. Rev. Immunol. 5, 521–531 (2005).
Devergne, O., Birkenbach, M. & Kieff, E. Epstein-Barr virus–induced gene 3 and the p35 subunit of interleukin 12 form a novel heterodimeric hematopoietin. Proc. Natl. Acad. Sci. USA 94, 12041–12046 (1997).
Niedbala, W. et al. IL-35 is a novel cytokine with therapeutic effects against collagen-induced arthritis through the expansion of regulatory T cells and suppression of Th17 cells. Eur. J. Immunol. 37, 3021–3029 (2007).
Caspi, R.R. A look at autoimmunity and inflammation in the eye. J. Clin. Invest. 120, 3073–3083 (2010).
Fillatreau, S., Gray, D. & Anderton, S.M. Not always the bad guys: B cells as regulators of autoimmune pathology. Nat. Rev. Immunol. 8, 391–397 (2008).
Carter, N.A. et al. Mice lacking endogenous IL-10–producing regulatory B cells develop exacerbated disease and present with an increased frequency of Th1/Th17 but a decrease in regulatory T cells. J. Immunol. 186, 5569–5579 (2011).
Ding, Q. et al. Regulatory B cells are identified by expression of TIM-1 and can be induced through TIM-1 ligation to promote tolerance in mice. J. Clin. Invest. 121, 3645–3656 (2011).
Nussenblatt, R.B. Proctor Lecture. Experimental autoimmune uveitis: mechanisms of disease and clinical therapeutic indications. Invest. Ophthalmol. Vis. Sci. 32, 3131–3141 (1991).
Caspi, R.R. et al. A new model of autoimmune disease. Experimental autoimmune uveoretinitis induced in mice with two different retinal antigens. J. Immunol. 140, 1490–1495 (1988).
Vasconcellos, R., Carter, N.A., Rosser, E.C. & Mauri, C. IL-12p35 subunit contributes to autoimmunity by limiting IL-27–driven regulatory responses. J. Immunol. 187, 3402–3412 (2011).
Collison, L.W. et al. The composition and signaling of the IL-35 receptor are unconventional. Nat. Immunol. 13, 290–299 (2012).
Tarrant, T.K., Silver, P.B., Chan, C.C., Wiggert, B. & Caspi, R.R. Endogenous IL-12 is required for induction and expression of experimental autoimmune uveitis. J. Immunol. 161, 122–127 (1998).
Egwuagu, C.E., Charukamnoetkanok, P. & Gery, I. Thymic expression of autoantigens correlates with resistance to autoimmune disease. J. Immunol. 159, 3109–3112 (1997).
Shen, P. et al. IL-35–producing B cells are critical regulators of immunity during autoimmune and infectious diseases. Nature 507, 366–370 (2014).
Vignali, D.A. & Kuchroo, V.K. IL-12 family cytokines: immunological playmakers. Nat. Immunol. 13, 722–728 (2012).
Wang, R.X., Yu, C.R., Mahdi, R.M. & Egwuagu, C.E. Novel IL27p28/IL12p40 cytokine suppressed experimental autoimmune uveitis by inhibiting autoreactive Th1/Th17 cells and promoting expansion of regulatory T cells. J. Biol. Chem. 287, 36012–36021 (2012).
Stumhofer, J.S. et al. A role for IL-27p28 as an antagonist of gp130-mediated signaling. Nat. Immunol. 11, 1119–1126 (2010).
Nussenblatt, R.B. Bench to bedside: new approaches to the immunotherapy of uveitic disease. Int. Rev. Immunol. 21, 273–289 (2002).
Olkhanud, P.B. et al. Tumor-evoked regulatory B cells promote breast cancer metastasis by converting resting CD4 T cells to T-regulatory cells. Cancer Res. 71, 3505–3515 (2011).
Liu, X., Lee, Y.S., Yu, C.R. & Egwuagu, C.E. Loss of STAT3 in CD4+ T cells prevents development of experimental autoimmune diseases. J. Immunol. 180, 6070–6076 (2008).
Takase, H. et al. Induction of suppressors of cytokine signaling (SOCS) in the retina during experimental autoimmune uveitis (EAU): potential neuroprotective role of SOCS proteins. J. Neuroimmunol. 168, 118–127 (2005).
Oh, H.M. et al. Autoreactive memory CD4+ T lymphocytes that mediate chronic uveitis reside in the bone marrow through STAT3-dependent mechanisms. J. Immunol. 187, 3338–3346 (2011).
Fillatreau, S., Sweenie, C.H., McGeachy, M.J., Gray, D. & Anderton, S.M. B cells regulate autoimmunity by provision of IL-10. Nat. Immunol. 3, 944–950 (2002).
Paques, M. et al. Panretinal, high-resolution color photography of the mouse fundus. Invest. Ophthalmol. Vis. Sci. 48, 2769–2774 (2007).
Xu, H. et al. A clinical grading system for retinal inflammation in the chronic model of experimental autoimmune uveoretinitis using digital fundus images. Exp. Eye Res. 87, 319–326 (2008).
Chan, C.C. et al. Pathology of experimental autoimmune uveoretinitis in mice. J. Autoimmun. 3, 247–255 (1990).
Amadi-Obi, A. et al. TH17 cells contribute to uveitis and scleritis and are expanded by IL-2 and inhibited by IL-27/STAT1. Nat. Med. 13, 711–718 (2007).
Yu, C.R., Lee, Y.S., Mahdi, R.M., Surendran, N. & Egwuagu, C.E. Therapeutic targeting of STAT3 (signal transducers and activators of transcription 3) pathway inhibits experimental autoimmune uveitis. PLoS ONE 7, e29742 (2012).
Li, W., Nagineni, C.N., Hooks, J.J., Chepelinsky, A.B. & Egwuagu, C.E. Interferon-g signaling in human retinal pigment epithelial cells mediated by STAT1, ICSBP, and IRF-1 transcription factors. Invest. Ophthalmol. Vis. Sci. 40, 976–982 (1999).
Oh, H.M. et al. STAT3 protein promotes T cell survival and inhibits interleukin-2 production through up-regulation of class O Forkhead transcription factors. J. Biol. Chem. 286, 30888–30897 (2011).
Acknowledgements
The Intramural Research Programs of the NEI and NIH provided funding for this research. We thank H. Young (National Cancer Institute, NIH) and R.R. Caspi and C.-C. Chan (NEI, NIH) for critical reading of the manuscript. We also thank C.-C. Chan for expert analyses of Fundus images and histology sections. In addition, we thank A. O'Hara and C.A. Hunter (University of Pennsylvania) for providing IL-27Rα KO B cells and R. Villasmil (NEI/NIH FLOW Cytometry Core facility) for cell sorting and FACS analysis.
Author information
Authors and Affiliations
Contributions
R.-X.W. conducted all experiments, prepared the figures and edited the manuscript. C.-R.Y. assisted with EAU experiments, FACS analysis, preparation of the figures and editing of the manuscript. I.M.D. assisted with EAU experiments, FACS analysis, protein purification, preparation of the figures and editing of the manuscript. R.M.M. performed western blot analyses and assisted with rIL-35 preparation. M.B.D. and Y.V.S. assisted with FPLC analysis. P.T.W. performed equilibrium ultracentrifugation analysis. S.-H.K. assisted with EAU experiments and fundoscopy. C.E.E. conceived, designed and supervised the project and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 (PDF 1276 kb)
Rights and permissions
About this article
Cite this article
Wang, RX., Yu, CR., Dambuza, I. et al. Interleukin-35 induces regulatory B cells that suppress autoimmune disease. Nat Med 20, 633–641 (2014). https://doi.org/10.1038/nm.3554
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3554
This article is cited by
-
The role of regulatory B cells in immune regulation and childhood allergic asthma
Molecular and Cellular Pediatrics (2024)
-
Pathogenese der Uveitis
Spektrum der Augenheilkunde (2024)
-
Elevated plasma levels of specific antiplatelet glycoprotein autoantibodies in patients with primary Sjögren syndrome with thrombocytopenia
Clinical Rheumatology (2024)
-
Interplay of IL-33 and IL-35 Modulates Th2/Th17 Responses in Cigarette Smoke Exposure HDM-Induced Asthma
Inflammation (2024)
-
Interleukin-35 -producing B cells rescues inflammatory bowel disease in a mouse model via STAT3 phosphorylation and intestinal microbiota modification
Cell Death Discovery (2023)