Abstract
The past decade witnessed unprecedented efforts to control malaria, including renewed political and financial commitment and increased availability of both old and new strategies and tools. However, malaria still represents a major health burden, particularly in Africa. Important challenges such as the fragility of many health systems, the rise of insecticide and drug resistance, and particularly the expected decline both in funding and in the coverage of key interventions if they are not replaced as needed, urgently need to be addressed. Further research and development is also becoming increasingly crucial. Among other needs, common methodologies for estimating and tracking the malaria burden, new strategies to measure transmission, better understanding of immunity, and increased knowledge of the mechanisms and effects of resistance to drugs and insecticides stand out. The ongoing efforts in research and development for new antimalarial drugs, more sensitive point-of-care rapid diagnostic tests and new insecticides need further innovation and substantial strengthening. Clearly, efforts should focus not only on Plasmodium falciparum but also and increasingly on Plasmodium vivax, the neglected human malaria parasite. Addressing these challenges in a comprehensive and timely way will allow us to sustain the gains made so far and make further progress in control and progressive elimination.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
World Health Organization. World malaria report 2012. World Health Organization http://www.who.int/malaria/publications/world_malaria_report_2012/wmr2012_full_report.pdf (2012).
Roll Back Malaria Partnership. The global malaria action plan. World Health Organization http://www.rbm.who.int/gmap/gmap.pdf (2008).
Roll Back Malaria Partnership. Refined/updated GMAP objectives, targets, milestones and priorities beyond 2011. Roll Back Malaria http://www.rbm.who.int/gmap/gmap2011update.pdf (2011).
Moran, M. et al. Neglected disease research and development: is innovation under threat? Policy Cures http://www.hst.org.za/sites/default/files/g-finder_2011.pdf (2011).
Alonso, P.L. et al. A research agenda to underpin malaria eradication. PLoS Med. 8, e1000406 (2011).
Murray, C.J.L. et al. Global malaria mortality between 1980 and 2010: a systematic analysis. Lancet 379, 413–431 (2012).
Guerra, C.A. et al. The limits and intensity of Plasmodium falciparum transmission: implications for malaria control and elimination worldwide. PLoS Med. 5, e38 (2008).
Okiro, E.A. et al. The decline in pediatric malaria admissions on the coast of Kenya. Malar. J. 6, 151 (2007).
Schellenberg, D. et al. The changing epidemiology of malaria in Ifakara Town, Southern Tanzania. Trop. Med. Int. Health 9, 68–76 (2004).
Bhattarai, A. et al. Impact of artemesinin-based combination therapy and insecticide-treated nets on malaria burden in Zanzibar. PLoS Med. 4, e309 (2007).
Sharp, B.L. et al. Seven years of regional malaria control collaboration- Mozambique, South Africa and Swaziland. Am. J. Trop. Med. Hyg. 76, 42–47 (2007).
Rodrigues, A. et al. Changing pattern of malaria in Bissau, Guinea Bissau. Trop. Med. Int. Health 13, 410–417 (2008).
Nyarango, P.M. et al. A steep decline of malaria morbidity and mortality trends in Eritrea between 2000 and 2004: the effect of combination of control methods. Malar. J. 5, 33 (2006).
World Health Organization. Global plan for artemisinin resistance containment. World Health Organization http://www.who.int/malaria/publications/atoz/artemisinin_resistance_containment_2011.pdf (2011).
Charman, S.A. et al. Synthetic ozonide drug candidate OZ439 offers new hope for a single-dose cure of uncomplicated malaria. Proc. Natl. Acad. Sci. USA 108, 4400–4405 (2011).
Rottmann, M. et al. Spiroindolones, a potent compound class for the treatment of malaria. Science 329, 1175–1180 (2010).
The malERA Consultative Group on Drugs. A research agenda for malaria eradication: Drugs. PLoS Med. 8, e1000402 (2011).
Davies, T.E., O'Reilly, A.O., Field, L.M., Wallace, B. & Williamson, M.S. Knockdown resistance to DDT and pyrethroids: from target-site mutations to molecular modelling. Pest Manag. Sci. 64, 1126–1130 (2008).
Matambo, T.S. et al. Insecticide resistance in the malaria mosquito Anopheles arabiensis and association with the kdr mutation. Med. Vet. Entomol. 21, 97–102 (2007).
Enayati, A. & Hemingway, J. Malaria management: Past, present, and future. Annu. Rev. Entomol. 55, 569–591 (2010).
World Health Organization. Global plan for insecticide resistance management in malaria vectors. World Health Organization http://whqlibdoc.who.int/publications/2012/9789241564472_eng.pdf (2012).
The malERA Consultative Group on Vector Control. A research agenda for malaria eradication: Vector control. PLoS Med. 8, e1000401 (2011).
World Health Organization. Guidelines for the treatment of malaria, second edition. World Health Organization http://whqlibdoc.who.int/publications/2010/9789241547925_eng.pdf (2010).
D'Acremont, V. et al. Reduction of anti-malarial consumption after rapid diagnostic tests implementation in Dar es Salaam: a before-after and cluster randomized controlled study. Malar. J. 10, 107 (2011).
The malERA Consultative Group on Diagnoses and Diagnostics. A research agenda for malaria eradication: diagnoses and diagnostics. PLoS Med. 8, e1000396 (2011).
Aponte, J.J. et al. Efficacy and safety of intermittent preventive treatment with sulfadoxine-pyrimethamine for malaria in African infants: a pooled analysis of six randomised, placebo-controlled trials. Lancet 374, 1533–1542 (2009).
Greenwood, B.M. Comparison of two strategies for control of malaria within a primary health care programme in The Gambia. Lancet 1, 1121–1127 (1988).
WHO/GMP Technical Expert Group on Preventive Chemotherapy. Report of the technical consultation on seasonal malaria chemoprevention (SMC). World Health Organization http://www.who.int/malaria/publications/atoz/smc_report_teg_meetingmay2011.pdf (2011).
WHO Global Malaria Programme. WHO policy recommendation: seasonal malaria chemoprevention (SMC) for Plasmodium falciparum malaria control in highly seasonal transmission areas of the Sahel sub-region in Africa. World Health Organization http://www.who.int/malaria/publications/atoz/smc_policy_recommendation_en_032012.pdf (2012).
Alonso, P.L. et al. Efficacy of the RTS,S/AS02A vaccine against Plasmodium falciparum infection and disease in young African children: randomised controlled trial. Lancet 364, 1411–1420 (2004).
Aponte, J.J. et al. Safety of the RTS,S/AS02D candidate malaria vaccine in infants living in a highly endemic area of Mozambique: a double blind randomised controlled phase I/IIb trial. Lancet 370, 1543–1551 (2007).
Abdulla, S. et al. Safety and immunogenicity of RTS,S/AS02D malaria vaccine in infants. N. Engl. J. Med. 359, 2533–2544 (2008).
RTS,S Clinical Trials Partnership. et al. First results of phase 3 trial of RTS,S/AS01 malaria vaccine in African children. N. Engl. J. Med. 365, 1863–1875 (2011).
RTS,S Clinical Trials Partnership. et al. A phase 3 trial of RTS,S/AS01 malaria vaccine in African infants. N. Engl. J. Med. 367, 2284–2295 (2012).
Aide, P. et al. Safety, immunogenicity and duration of protection of the RTS,S/AS02D malaria vaccine: one year follow-up of a randomized controlled phase I/IIb trial. PLoS ONE 5, e13838 (2010).
Guinovart, C. et al. Insights into long-lasting protection induced by RTS,S/AS02A malaria vaccine: further results from a phase IIb trial in Mozambican children. PLoS ONE 4, e5165 (2009).
Hoffman, S.L. et al. Development of a metabolically active, non-replicating sporozoite vaccine to prevent Plasmodium falciparum malaria. Hum. Vaccin. 6, 97–106 (2010).
Cech, P.G. et al. Virosome-formulated Plasmodium falciparum AMA-1 & CSP derived peptides as malaria vaccine: randomized phase 1b trial in semi-immune adults & children. PLoS ONE 6, e22273 (2011).
Reyes-Sandoval, A. et al. Prime-boost immunization with adenoviral and modified vaccinia virus ankara vectors enhances the durability and polyfunctionality of protective malaria CD8+ T-cell responses. Infect. Immun. 78, 142–153 (2010).
Mendis, K., Sina, B.J., Marchesini, P. & Carter, R. The neglected burden of Plasmodium vivax malaria. Am. J. Trop. Med. Hyg. 64, 97–106 (2001).
Acknowledgements
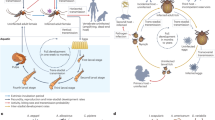
We thank M. Winkler for assistance in compiling Figure 1 and M. González-Silva for editorial assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Alonso, P., Tanner, M. Public health challenges and prospects for malaria control and elimination. Nat Med 19, 150–155 (2013). https://doi.org/10.1038/nm.3077
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3077
This article is cited by
-
Malaria therapeutics: are we close enough?
Parasites & Vectors (2023)
-
Insecticide-treated bed net use and associated factors among households having under-five children in East Africa: a multilevel binary logistic regression analysis
Malaria Journal (2023)
-
Risk factors associated with malaria infection along China–Myanmar border: a case–control study
Malaria Journal (2022)
-
Pharmacokinetic profile of amodiaquine and its active metabolite desethylamodiaquine in Ghanaian patients with uncomplicated falciparum malaria
Malaria Journal (2021)
-
A natural symbiotic bacterium drives mosquito refractoriness to Plasmodium infection via secretion of an antimalarial lipase
Nature Microbiology (2021)