Abstract
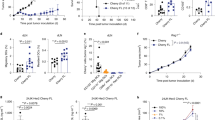
T cell immunity can potentially eradicate malignant cells and lead to clinical remission in a minority of patients with cancer. In the majority of these individuals, however, there is a failure of the specific T cell receptor (TCR)–mediated immune recognition and activation process. Here we describe the engineering and characterization of new reagents termed immune-mobilizing monoclonal TCRs against cancer (ImmTACs). Four such ImmTACs, each comprising a distinct tumor-associated epitope-specific monoclonal TCR with picomolar affinity fused to a humanized cluster of differentiation 3 (CD3)-specific single-chain antibody fragment (scFv), effectively redirected T cells to kill cancer cells expressing extremely low surface epitope densities. Furthermore, these reagents potently suppressed tumor growth in vivo. Thus, ImmTACs overcome immune tolerance to cancer and represent a new approach to tumor immunotherapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Köhler, G. & Milstein, C. Continuous cultures of fused cells secreting antibody of predefined specificity. Nature 256, 495–497 (1975).
Isaacs, J.D. et al. Humanised monoclonal antibody therapy for rheumatoid arthritis. Lancet 340, 748–752 (1992).
Maynard, J. & Georgiou, G. Antibody engineering. Annu. Rev. Biomed. Eng. 2, 339–376 (2000).
Kufer, P., Lutterbuse, R. & Baeuerle, P.A. A revival of bispecific antibodies. Trends Biotechnol. 22, 238–244 (2004).
Clynes, R.A., Towers, T.L., Presta, L.G. & Ravetch, J.V. Inhibitory Fc receptors modulate in vivo cytoxicity against tumor targets. Nat. Med. 6, 443–446 (2000).
Cartron, G. et al. Therapeutic activity of humanized anti-CD20 monoclonal antibody and polymorphism in IgG Fc receptor FcγRIIIa gene. Blood 99, 754–758 (2002).
von Mehren, M., Adams, G.P. & Weiner, L.M. Monoclonal antibody therapy for cancer. Annu. Rev. Med. 54, 343–369 (2003).
Withoff, S., Helfrich, W., de Leij, L.F. & Molema, G. Bi-specific antibody therapy for the treatment of cancer. Curr. Opin. Mol. Ther. 3, 53–62 (2001).
Bargou, R. et al. Tumor regression in cancer patients by very low doses of a T cell–engaging antibody. Science 321, 974–977 (2008).
Baeuerle, P.A., Kufer, P. & Bargou, R. BiTE: teaching antibodies to engage T-cells for cancer therapy. Curr. Opin. Mol. Ther. 11, 22–30 (2009).
Kiewe, P. et al. Phase I trial of the trifunctional anti-HER2 x anti-CD3 antibody ertumaxomab in metastatic breast cancer. Clin. Cancer Res. 12, 3085–3091 (2006).
Sebastian, M. et al. Treatment of malignant pleural effusion with the trifunctional antibody catumaxomab (Removab) (anti-EpCAM x anti-CD3): results of a phase 1/2 study. J. Immunother. 32, 195–202 (2009).
Cole, D.K. et al. Human TCR-binding affinity is governed by MHC class restriction. J. Immunol. 178, 5727–5734 (2007).
Boulter, J.M. et al. Stable, soluble T-cell receptor molecules for crystallization and therapeutics. Protein Eng. 16, 707–711 (2003).
Li, Y. et al. Directed evolution of human T-cell receptors with picomolar affinities by phage display. Nat. Biotechnol. 23, 349–354 (2005).
Holler, P.D. et al. In vitro evolution of a T cell receptor with high affinity for peptide/MHC. Proc. Natl. Acad. Sci. USA 97, 5387–5392 (2000).
Chlewicki, L.K., Holler, P.D., Monti, B.C., Clutter, M.R. & Kranz, D.M. High-affinity, peptide-specific T cell receptors can be generated by mutations in CDR1, CDR2 or CDR3. J. Mol. Biol. 346, 223–239 (2005).
Adema, G.J. et al. Melanocyte lineage-specific antigens recognized by monoclonal antibodies NKI-beteb, HMB-50, and HMB-45 are encoded by a single cDNA. Am. J. Pathol. 143, 1579–1585 (1993).
Gaugler, B. et al. Human gene MAGE-3 codes for an antigen recognized on a melanoma by autologous cytolytic T lymphocytes. J. Exp. Med. 179, 921–930 (1994).
Coulie, P.G. et al. A new gene coding for a differentiation antigen recognized by autologous cytolytic T lymphocytes on HLA-A2 melanomas. J. Exp. Med. 180, 35–42 (1994).
Kawakami, Y. et al. Cloning of the gene coding for a shared human melanoma antigen recognized by autologous T cells infiltrating into tumor. Proc. Natl. Acad. Sci. USA 91, 3515–3519 (1994).
Chen, Y.T. et al. A testicular antigen aberrantly expressed in human cancers detected by autologous antibody screening. Proc. Natl. Acad. Sci. USA 94, 1914–1918 (1997).
Cox, A.L. et al. Identification of a peptide recognized by five melanoma-specific human cytotoxic T cell lines. Science 264, 716–719 (1994).
Celis, E. et al. Induction of anti-tumor cytotoxic T lymphocytes in normal humans using primary cultures and synthetic peptide epitopes. Proc. Natl. Acad. Sci. USA 91, 2105–2109 (1994).
Kawakami, Y. et al. Identification of the immunodominant peptides of the MART-1 human melanoma antigen recognized by the majority of HLA-A2–restricted tumor infiltrating lymphocytes. J. Exp. Med. 180, 347–352 (1994).
Jäger, E. et al. Simultaneous humoral and cellular immune response against cancer-testis antigen NY-ESO-1: definition of human histocompatibility leukocyte antigen (HLA)-A2–binding peptide epitopes. J. Exp. Med. 187, 265–270 (1998).
Purbhoo, M.A. et al. Quantifying and imaging NY-ESO-1/LAGE-1–derived epitopes on tumor cells using high affinity T cell receptors. J. Immunol. 176, 7308–7316 (2006).
Chattopadhyay, P.K. et al. The cytolytic enzymes granyzme A, granzyme B, and perforin: expression patterns, cell distribution, and their relationship to cell maturity and bright CD57 expression. J. Leukoc. Biol. 85, 88–97 (2009).
Schietinger, A., Philip, M., Liu, R.B., Schreiber, K. & Schreiber, H. Bystander killing of cancer requires the cooperation of CD4+ and CD8+ T cells during the effector phase. J. Exp. Med. 207, 2469–2477 (2010).
Arstila, T.P. et al. A direct estimate of the human αβ T cell receptor diversity. Science 286, 958–961 (1999).
Purbhoo, M.A., Irvine, D.J., Huppa, J.B. & Davis, M.M. T cell killing does not require the formation of a stable mature immunological synapse. Nat. Immunol. 5, 524–530 (2004).
Price, D.A., Klenerman, P., Booth, B.L., Phillips, R.E. & Sewell, A.K. Cytotoxic T lymphocytes, chemokines and antiviral immunity. Immunol. Today 20, 212–216 (1999).
Quezada, S.A. et al. Tumor-reactive CD4+ T cells develop cytotoxic activity and eradicate large established melanoma after transfer into lymphopenic hosts. J. Exp. Med. 207, 637–650 (2010).
Rosenberg, S.A. & Dudley, M.E. Adoptive cell therapy for the treatment of patients with metastatic melanoma. Curr. Opin. Immunol. 21, 233–240 (2009).
Benowitz, S. Rethinking cancer vaccine trials: would new measures of success make a difference? J. Natl. Cancer Inst. 100, 237–238 (2008).
Johnson, L.A. et al. Gene therapy with human and mouse T-cell receptors mediates cancer regression and targets normal tissues expressing cognate antigen. Blood 114, 535–546 (2009).
Moysey, R., Vuidepot, A.L. & Boulter, J.M. Amplification and one-step expression cloning of human T cell receptor genes. Anal. Biochem. 326, 284–286 (2004).
Dunn, S.M. et al. Directed evolution of human T cell receptor CDR2 residues by phage display dramatically enhances affinity for cognate peptide-MHC without increasing apparent cross-reactivity. Protein Sci. 15, 710–721 (2006).
Price, D.A. et al. Avidity for antigen shapes clonal dominance in CD8+ T cell populations specific for persistent DNA viruses. J. Exp. Med. 202, 1349–1361 (2005).
Bertozzi, C.C. et al. Multiple initial culture conditions enhance the establishment of cell lines from primary ovarian cancer specimens. In Vitro Cell. Dev. Biol. Anim. 42, 58–62 (2006).
Acknowledgements
We would like to thank Sanofi Pasteur for funding affinity maturation of the gp100 and MAGE-A3 mTCRs; C. Yee (Fred Hutchinson Cancer Research Centre, Seattle, Washington, USA), P. Coulie (University of Louvain, Brussels, Belgium) and V. Cerundolo (Weatherall Institute of Molecular Medicine, University of Oxford, UK) for providing T cell clones; Southern Research and Cellvax for conducting mouse xenograft experiments; Southern Research for immunohistochemistry staining; R. Liu for assistance with mouse imaging and tumor measurements; K. Haines (Translational and Correlative Studies Laboratory, University of Pennsylvania School of Medicine, Philadelphia, Pennsylvania, USA) for technical support; and A. Secreto, C. Keefer and G. Danet-Desnoyers (Stem Cell and Xenograft Core, University of Pennsylvania School of Medicine, Philadelphia, Pennsylvania, USA) for assistance with the established tumor xenograft studies.
Author information
Authors and Affiliations
Contributions
N.L., N.E.H., P.E.M. and E.G. isolated wild-type mTCRs and carried out the phage display process under the supervision of Y.L.; A.V. and Y.L. were involved in ImmTAC construct optimization; T.M.M., J.G., A.V., E.E.B., N.J.P., N.M.L. and B.J.C. were involved in protein production and biochemical testing; G.B., K.J.A., A.L., N.J.H., K.L., S.J.P., J.V.H. and R.E.D. performed in vitro experiments under the supervision of D.D.W., R.A., D.H.S., A.K.S. and D.A.P. Large-scale production, stability testing, quality control and biochemical testing of ImmTACs was conducted by F.C.B., M.S., A.J., E.E.B., P.T.T. and S.M.D. under the supervision of Y.M. Mouse xenograft experiments were designed, coordinated and conducted by G.P., C.H.J., M.K. and D.D.W. Data analysis and interpretation were performed by D.H.S. and D.A.P.; D.H.S. and D.A.P. wrote the paper. B.K.J. conceived the idea and directed the project. All authors contributed to discussions.
Corresponding author
Ethics declarations
Competing interests
N.L., G.B., K.J.A., T.M.M., N.J.H., J.G., F.C.B., N.J.P., N.M.L., N.E.H., P.E.M., Y.L., B.J.C., M.S., E.E.B., P.T.T., S.J.P., R.E.D., J.V.H., S.M.D., R.A., A.J., Y.M., A.V., D.D.W., D.H.S. and B.K.J. are employed by ImmunoCore Ltd., and the reagents studied in this manuscript were developed by ImmunoCore Ltd.
Supplementary information
Supplementary Text and Figures
Supplementary Methods and Supplementary Table 1 (PDF 305 kb)
Supplementary Video 1a
Supplementary Video 1a (MOV 382 kb)
Supplementary Video 1b
Supplementary Video 1b (MOV 3096 kb)
Rights and permissions
About this article
Cite this article
Liddy, N., Bossi, G., Adams, K. et al. Monoclonal TCR-redirected tumor cell killing. Nat Med 18, 980–987 (2012). https://doi.org/10.1038/nm.2764
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2764
This article is cited by
-
CAR-T cell therapy targeting surface expression of TYRP1 to treat cutaneous and rare melanoma subtypes
Nature Communications (2024)
-
T cell receptor therapeutics: immunological targeting of the intracellular cancer proteome
Nature Reviews Drug Discovery (2023)
-
Immune mobilising T cell receptors redirect polyclonal CD8+ T cells in chronic HIV infection to form immunological synapses
Scientific Reports (2022)
-
Therapeutic high affinity T cell receptor targeting a KRASG12D cancer neoantigen
Nature Communications (2022)
-
Clinical and molecular response to tebentafusp in previously treated patients with metastatic uveal melanoma: a phase 2 trial
Nature Medicine (2022)