Abstract
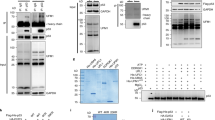
The TP53 gene (encoding the p53 tumor suppressor) is rarely mutated, although frequently inactivated, in medulloblastoma and ependymoma. Recent work in mouse models showed that the loss of p53 accelerated the development of medulloblastoma. The mechanism underlying p53 inactivation in human brain tumors is not completely understood. We show that ubiquitination factor E4B (UBE4B), an E3 and E4 ubiquitin ligase, physically interacts with p53 and Hdm2 (also known as Mdm2 in mice). UBE4B promotes p53 polyubiquitination and degradation and inhibits p53-dependent transactivation and apoptosis. Notably, silencing UBE4B expression impairs xenotransplanted tumor growth in a p53-dependent manner and overexpression of UBE4B correlates with decreased expression of p53 in these tumors. We also show that UBE4B overexpression is often associated with amplification of its gene in human brain tumors. Our data indicate that amplification and overexpression of UBE4B represent previously undescribed molecular mechanisms of inactivation of p53 in brain tumors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Farrell, P.J., Allan, G.J., Shanahan, F., Vousden, K.H. & Crook, T. p53 is frequently mutated in Burkitt's lymphoma cell lines. EMBO J. 10, 2879–2887 (1991).
Crook, T. & Vousden, K.H. Properties of p53 mutations detected in primary and secondary cervical cancers suggest mechanisms of metastasis and involvement of environmental carcinogens. EMBO J. 11, 3935–3940 (1992).
Greenblatt, M.S., Bennet, W.P., Hollstein, M. & Harris, C.C. Mutations in the p53 tumor suppressor gene: Clues to cancer etiology and molecular pathogenesis. Cancer Res. 54, 4855–4878 (1994).
Levine, A.J. p53, the cellular gatekeeper for growth and division. Cell 88, 323–331 (1997).
Vogelstein, B., Lane, D. & Levine, A.J. Surfing the p53 network. Nature 408, 307–310 (2000).
Donehower, L.A. et al. Mice deficient for p53 are developmentally normal but susceptible to spontaneous tumours. Nature 356, 215–221 (1992).
Haupt, Y., Maya, R., Kazaz, A. & Oren, M. Mdm2 promotes the rapid degradation of p53. Nature 387, 296–299 (1997).
Kubbutat, M.H., Jones, S.N. & Vousden, K.H. Regulation of p53 stability by Mdm2. Nature 387, 299–303 (1997).
Honda, R., Tanaka, H. & Yasuda, Y. Oncoprotein MDM2 is a ubiquitin ligase E3 for tumor suppressor p53. FEBS Lett. 420, 25–27 (1997).
Montes de Oca Luna, R., Wagner, D.S. & Lozano, G. Rescue of early embryonic lethality in mdm2-deficient mice by deletion of p53. Nature 378, 203–206 (1995).
Jones, S.N., Roe, A.E., Donehower, L.A. & Bradley, A. Rescue of embryonic lethality in Mdm2-deficient mice by absence of p53. Nature 378, 206–208 (1995).
Piotrowski, J. et al. Inhibition of the 26 S proteasome by polyubiquitin chains synthesized to have defined lengths. J. Biol. Chem. 272, 23712–23721 (1997).
Thrower, J.S., Hoffman, L., Rechsteiner, M. & Pickart, C.M. Recognition of the polyubiquitin proteolytic signal. EMBO J. 19, 94–102 (2000).
Pickart, C.M. Ubiquitin in chains. Trends Biochem. Sci. 25, 544–548 (2000).
Rodriguez, M.S., Desterro, J.M., Lain, S., Lane, D.P. & Hay, R.T. Multiple C-terminal lysine residues target p53 for ubiquitin-proteasome-mediated degradation. Mol. Cell. Biol. 20, 8458–8467 (2000).
Lai, Z. et al. Human mdm2 mediates multiple mono-ubiquitination of p53 by a mechanism requiring enzyme isomerization. J. Biol. Chem. 276, 31357–31367 (2001).
Grossman, S.R. et al. Polyubiquitination of p53 by a ubiquitin ligase activity of p300. Science 300, 342–344 (2003).
Li, M. et al. Mono- versus polyubiquitination: differential control of p53 fate by Mdm2. Science 302, 1972–1975 (2003).
Johnson, E.S., Ma, P.C.M., Ota, I. & Varshavsky, A. A proteolytic pathway that recognizes ubiquitin as a degradation signal. J. Biol. Chem. 270, 17442–17456 (1995).
Koegl, M. et al. A novel ubiquitination factor, E4, is involved in multiubiquitin chain assembly. Cell 96, 635–644 (1999).
Hatakeyama, S., Yada, M., Matsumoto, M., Ishida, N. & Nakayama, K.I. U box proteins as a new family of ubiquitin-protein ligases. J. Biol. Chem. 276, 33111–33120 (2001).
Tu, D., Li, W., Ye, Y. & Brunger, A.T. Structure and function of the yeast U-box-containing ubiquitin ligase Ufd2p. Proc. Natl. Acad. Sci. USA 104, 15599–15606 (2007).
Koepp, D.M., Harper, J.W. & Elledge, S.J. How the cyclin became a cyclin: regulated proteolysis in the cell cycle. Cell 97, 431–434 (1999).
Matsumoto, M. et al. Molecular clearance of ataxin-3 is regulated by a mammalian E4. EMBO J. 23, 659–669 (2004).
Okumura, F., Hatakeyama, S., Matsumoto, M., Kamura, T. & Nakayama, K.I. Functional regulation of FEZ1 by the U-box–type ubiquitin ligase E4B contributes to neuritogenesis. J. Biol. Chem. 279, 53533–53543 (2004).
Hosoda, M. et al. UFD2a mediates the proteasomal turnover of p73 without promoting p73 ubiquitination. Oncogene 24, 7156–7169 (2005).
Kaneko-Oshikawa, C. et al. Mammalian E4 is required for cardiac development and maintenance of the nervous system. Mol. Cell. Biol. 25, 10953–10964 (2005).
Leng, R.P. et al. Pirh2, a p53 induced ubiquitin-protein ligase, promotes p53 degradation. Cell 112, 779–791 (2003).
Sheng, Y. et al. Molecular basis of Pirh2-mediated p53 ubiquitylation. Nat. Struct. Mol. Biol. 15, 1334–1342 (2008).
Dornan, D. et al. The ubiquitin ligase COP1 is a critical negative regulator of p53. Nature 429, 86–92 (2004).
Shi, D. et al. CBP and p300 are cytoplasmic E4 polyubiquitin ligases for p53. Proc. Natl. Acad. Sci. USA 106, 16275–16280 (2009).
Sui, G. et al. Yin Yang 1 is a negative regulator of p53. Cell 117, 859–872 (2004).
Finlay, C.A. The mdm-2 oncogene can overcome wild-type p53 suppression of transformed cell growth. Mol. Cell. Biol. 13, 301–306 (1993).
McCurrach, M.E., Connor, T.M., Knudson, C.M., Korsmeyer, S.J. & Lowe, S.W. bax-deficiency promotes drug resistance and oncogenic transformation by attenuating p53-dependent apoptosis. Proc. Natl. Acad. Sci. USA 94, 2345–2349 (1997).
Serrano, M., Lin, A.W., McCurrach, M.E., Beach, D. & Lowe, S.W. Oncogenic ras provokes premature cell senescence associated with accumulation of p53 and p16INK4a. Cell 88, 593–602 (1997).
Waldman, T. et al. Cell-cycle arrest versus cell death in cancer therapy. Nat. Med. 3, 1034–1036 (1997).
Bunz, F. et al. Requirement for p53 and p21 to sustain G2 arrest after DNA damage. Science 282, 1497–1501 (1998).
Saylors, R.L. et al. Infrequent p53 gene mutations in medulloblastomas. Cancer Res. 51, 4721–4723 (1991).
Gaspar, N. et al. p53 Pathway dysfunction in primary childhood ependymomas. Pediatr. Blood Cancer 46, 604–613 (2006).
Adesina, A.M., Nalbantoglu, J. & Cavenee, W.K. p53 gene mutation and mdm2 gene amplification are uncommon in medulloblastoma. Cancer Res. 54, 5649–5651 (1994).
Crawford, J.R., MacDonald, T.J. & Packer, R.J. Medulloblastoma in childhood: new biological advances. Lancet Neurol. 6, 1073–1085 (2007).
Uziel, T. et al. The tumor suppressors Ink4c and p53 collaborate independently with Patched to suppress medulloblastoma formation. Genes Dev. 19, 2656–2667 (2005).
Acknowledgements
We gratefully acknowledge W. Gu (Columbia University) for the His-ubiquitin (wild-type) and His-Ub-ko plasmids, A.G. Jochemsen (Erasmus University Medical Center) for the Myc-Mdm2 plasmid, C. Blattner (Universität Heidelberg) for pSuper.neo.gpf-Mdm2 siRNA plasmid, J.A. Mahoney (Johns Hopkins University) for pEF-DEST51-Flag-UBE4B plasmid, B. Vogelstein (Johns Hopkins University) for HCT116 TP53−/− cells, S. Benchimol (York University) for BJT and BJT/DD cell lines and G. Lozano (University of Texas, M.D. Anderson Cancer Center) for Mdm2−/− Trp53−/− MEFs as described in the text. We thank T. Turner for technical help in making the figures. This work was supported by grants from the Alberta Heritage Foundation for Medical Research and Canadian Institutes of Health Research (to R.P.L.) and from the US National Institutes of Health (to S.L.P.). R.P.L. is an Alberta Heritage Foundation for Medical Research scholar.
Author information
Authors and Affiliations
Contributions
H.W. and R.P.L. contributed to study design, performed most of the experiments, analyzed and interpreted the data and wrote the manuscript. S.L.P. provided logistical support and all tumor samples and interpreted and discussed the data. M.F. provided technical support and experimental assistance. N.T. conducted the western blotting for the pediatric astrocytoma tissues, isolated genomic DNAs from various tumor samples, carried out mutation detection for p53 in various tumor tissues and medulloblastoma cell lines and did long-term colony assays. J.M. collected tissue samples from various types of human brain tumors, cared for Ptch+/− mice, conducted all the mouse genotyping and isolated the cerebellum and cortex from the Ptch+/− mice. K.I.N. and S.H. provided the study material and technical support. V.A.T. provided technical support. L.F.S. conducted the FPLC protein purification experiments. L.S. provided technical support for the gel filtration. R.P.L. supervised and directed the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8, Supplementary Table 1 and Supplementary Methods (PDF 1891 kb)
Rights and permissions
About this article
Cite this article
Wu, H., Pomeroy, S., Ferreira, M. et al. UBE4B promotes Hdm2-mediated degradation of the tumor suppressor p53. Nat Med 17, 347–355 (2011). https://doi.org/10.1038/nm.2283
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2283
This article is cited by
-
UBE4B interacts with the ITCH E3 ubiquitin ligase to induce Ku70 and c-FLIPL polyubiquitination and enhanced neuroblastoma apoptosis
Cell Death & Disease (2023)
-
p63, a key regulator of Ago2, links to the microRNA-144 cluster
Cell Death & Disease (2022)
-
DNA damage response revisited: the p53 family and its regulators provide endless cancer therapy opportunities
Experimental & Molecular Medicine (2022)
-
The biology of ependymomas and emerging novel therapies
Nature Reviews Cancer (2022)
-
UBE4B, a microRNA-9 target gene, promotes autophagy-mediated Tau degradation
Nature Communications (2021)