Abstract
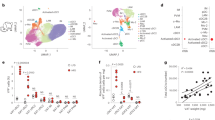
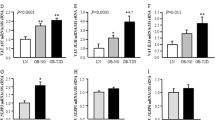
The emergence of chronic inflammation during obesity in the absence of overt infection or well-defined autoimmune processes is a puzzling phenomenon. The Nod-like receptor (NLR) family of innate immune cell sensors, such as the nucleotide-binding domain, leucine-rich–containing family, pyrin domain–containing-3 (Nlrp3, but also known as Nalp3 or cryopyrin) inflammasome are implicated in recognizing certain nonmicrobial originated 'danger signals' leading to caspase-1 activation and subsequent interleukin-1β (IL-1β) and IL-18 secretion. We show that calorie restriction and exercise-mediated weight loss in obese individuals with type 2 diabetes is associated with a reduction in adipose tissue expression of Nlrp3 as well as with decreased inflammation and improved insulin sensitivity. We further found that the Nlrp3 inflammasome senses lipotoxicity-associated increases in intracellular ceramide to induce caspase-1 cleavage in macrophages and adipose tissue. Ablation of Nlrp3 in mice prevents obesity-induced inflammasome activation in fat depots and liver as well as enhances insulin signaling. Furthermore, elimination of Nlrp3 in obese mice reduces IL-18 and adipose tissue interferon-γ (IFN-γ) expression, increases naive T cell numbers and reduces effector T cell numbers in adipose tissue. Collectively, these data establish that the Nlrp3 inflammasome senses obesity-associated danger signals and contributes to obesity-induced inflammation and insulin resistance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Dixit, V.D. Adipose-immune interactions during obesity and caloric restriction: reciprocal mechanisms regulating immunity and health span. J. Leukoc. Biol. 84, 882–892 (2008).
Hotamisligil, G.S. & Erbay, E. Nutrient sensing and inflammation in metabolic diseases. Nat. Rev. Immunol. 8, 923–934 (2008).
Luchsinger, J.A. & Gustafson, D.R. Adiposity, type 2 diabetes and Alzheimer's disease. J. Alzheimers Dis. 16, 693–704 (2009).
Yang, H. et al. Obesity accelerates thymic aging. Blood 114, 3803–3812 (2009).
Weisberg, S.P. et al. Obesity is associated with macrophage accumulation in adipose tissue. J. Clin. Invest. 112, 1796–1808 (2003).
Odegaard, J.I. & Chawla, A. Mechanisms of macrophage activation in obesity-induced insulin resistance. Nat. Clin. Pract. Endocrinol. Metab. 4, 619–626 (2008).
Lumeng, C.N., Bodzin, J.L. & Saltiel, A.R. Obesity induces a phenotypic switch in adipose tissue macrophage polarization. J. Clin. Invest. 117, 175–184 (2007).
Patsouris, D. et al. Ablation of CD11c-positive cells normalizes insulin sensitivity in obese insulin resistant animals. Cell Metab. 8, 301–309 (2008).
Hotamisligil, G.S. Inflammation and metabolic disorders. Nature 444, 860–867 (2006).
Arkan, M.C. et al. IKK-β links inflammation to obesity-induced insulin resistance. Nat. Med. 11, 191–198 (2005).
Jager, J., Grémeaux, T., Cormont, M., Le Marchand-Brustel, Y. & Tanti, J.F. Interleukin-1β–induced insulin resistance in adipocytes through down-regulation of insulin receptor substrate-1 expression. Endocrinology 148, 241–251 (2007).
Larsen, C.M. et al. Interleukin-1–receptor antagonist in type 2 diabetes mellitus. N. Engl. J. Med. 356, 1517–1526 (2007).
Osborn, O. et al. Treatment with an Interleukin 1 β antibody improves glycemic control in diet-induced obesity. Cytokine 44, 141–148 (2008).
Medzhitov, R. Recognition of microorganisms and activation of the immune response. Nature 449, 819–826 (2007).
Barton, G.M. & Kagan, J.C. A cell biological view of Toll-like receptor function: regulation through compartmentalization. Nat. Rev. Immunol. 9, 535–542 (2009).
Agostini, L. et al. NLRP3 forms an IL-1β–processing inflammasome with increased activity in Muckle-Wells autoinflammatory disorder. Immunity 20, 319–325 (2004).
Medzhitov, R. Origin and physiological roles of inflammation. Nature 454, 428–435 (2008).
Lamkanfi, M. & Dixit, V.M. Inflammasomes: guardians of cytosolic sanctity. Immunol. Rev. 227, 95–105 (2009).
Franchi, L., Eigenbrod, T., Muñoz-Planillo, R. & Nuñez, G. The inflammasome: a caspase-1–activation platform that regulates immune responses and disease pathogenesis. Nat. Immunol. 10, 241–247 (2009).
Martinon, F., Mayor, A. & Tschopp, J. The inflammasomes: guardians of the body. Annu. Rev. Immunol. 27, 229–265 (2009).
Dinarello, C.A. Immunological and inflammatory functions of the interleukin-1 family. Annu. Rev. Immunol. 27, 519–550 (2009).
Kostura, M.J. et al. Identification of a monocyte specific pre–interleukin 1 β convertase activity. Proc. Natl. Acad. Sci. USA 86, 5227–5231 (1989).
Kuida, K. et al. Altered cytokine export and apoptosis in mice deficient in interleukin-1 beta converting enzyme. Science 267, 2000–2003 (1995).
Martinon, F., Burns, K. & Tschopp, J. The inflammasome: a molecular platform triggering activation of inflammatory caspases and processing of proIL-β. Mol. Cell 10, 417–426 (2002).
Dostert, C. et al. Innate immune activation through Nlrp3 inflammasome sensing of asbestos and silica. Science 320, 674–677 (2008).
Halle, A. et al. The NLRP3 inflammasome is involved in the innate immune response to amyloid-β. Nat. Immunol. 9, 857–865 (2008).
Mariathasan, S. et al. Cryopyrin activates the inflammasome in response to toxins and ATP. Nature 440, 228–232 (2006).
Martinon, F., Pétrilli, V., Mayor, A., Tardivel, A. & Tschopp, J. Gout-associated uric acid crystals activate the NLRP3 inflammasome. Nature 440, 237–241 (2006).
Mariathasan, S. et al. Differential activation of the inflammasome by caspase-1 adaptors ASC and Ipaf. Nature 430, 213–218 (2004).
Shi, H. et al. TLR4 links innate immunity and fatty acid–induced insulin resistance. J. Clin. Invest. 116, 3015–3025 (2006).
Saberi, M. et al. Hematopoietic cell-specific deletion of Toll-like receptor 4 ameliorates hepatic and adipose tissue insulin resistance in high-fat-fed mice. Cell Metab. 10, 419–429 (2009).
Stienstra, R. et al. The inflammasome-mediated caspase-1 activation controls adipocyte differentiation and insulin sensitivity. Cell. Metab. 12, 593–605 (2009).
Bishop, N.A. & Guarente, L. Genetic links between diet and lifespan: shared mechanisms from yeast to humans. Nat. Rev. Genet. 8, 835–844 (2007).
Galgani, J.E. et al. Metabolic flexibility in response to glucose is not impaired in people with type 2 diabetes after controlling for glucose disposal rate. Diabetes 57, 841–845 (2008).
Kummer, J.A. et al. Inflammasome components NALP 1 and 3 show distinct but separate expression profiles in human tissues suggesting a site–specific role in the inflammatory response. J. Histochem. Cytochem. 55, 443–452 (2007).
Joosten, L.A. Inflammatory arthritis in caspase 1 gene–deficient mice: contribution of proteinase 3 to caspase 1–independent production of bioactive interleukin-1 β. Arthritis Rheum. 60, 3651–3662 (2009).
Virkamäki, A., Ueki, K. & Kahn, C.R. Protein-protein interaction in insulin signaling and the molecular mechanisms of insulin resistance. J. Clin. Invest. 103, 931–943 (1999).
Imaeda, A.B. et al. Acetaminophen-induced hepatotoxicity in mice is dependent on Tlr9 and the Nlrp3 inflammasome. J. Clin. Invest. 119, 305–314 (2009).
Watanabe, A. et al. Inflammasome-mediated regulation of hepatic stellate cells. Am. J. Physiol. Gastrointest. Liver Physiol. 296, G1248–G1257 (2009).
Khan, T. et al. Metabolic dysregulation and adipose tissue fibrosis: role of collagen VI. Mol. Cell. Biol. 29, 1575–1591 (2009).
Håversen, L., Danielsson, K.N., Fogelstrand, L. & Wiklund, O. Induction of proinflammatory cytokines by long-chain saturated fatty acids in human macrophages. Atherosclerosis 202, 382–393 (2009).
Park, T.S. et al. Ceramide is a cardiotoxin in lipotoxic cardiomyopathy. J. Lipid Res. 49, 2101–2112 (2008).
Prieur, X., Roszer, T. & Ricote, M. Lipotoxicity in macrophages: evidence from diseases associated with the metabolic syndrome. Biochim. Biophys. Acta 1801, 327–337 (2010).
Shah, C. et al. Protection from high fat diet-induced increase in ceramide in mice lacking plasminogen activator inhibitor 1. J. Biol. Chem. 283, 13538–13548 (2008).
Feuerer, M. et al. Lean, but not obese, fat is enriched for a unique population of regulatory T cells that affect metabolic parameters. Nat. Med. 15, 930–939 (2009).
Nishimura, S. et al. CD8+ effector T cells contribute to macrophage recruitment and adipose tissue inflammation in obesity. Nat. Med. 15, 914–920 (2009).
Winer, S. et al. Normalization of obesity-associated insulin resistance through immunotherapy. Nat. Med. 15, 921–929 (2009).
Yang, H. et al. Obesity increases the production of proinflammatory mediators from adipose tissue T cells and compromises TCR repertoire diversity: implications for systemic inflammation and insulin resistance. J. Immunol. 185, 1836–1845 (2010).
McGonagle, D. & McDermott, M.F. A proposed classification of the immunological diseases. PLoS Med. 3, e297 (2006).
Duewell, P. et al. NLRP3 inflammasomes are required for atherogenesis and activated by cholesterol crystals. Nature 464, 1357–1361 (2010).
Li, H., Ambade, A. & Re, F. Cutting edge: Necrosis activates the NLRP3 inflammasome. J. Immunol. 183, 1528–1532 (2009).
Halberg, N. et al. Hypoxia-inducible factor 1α induces fibrosis and insulin resistance in white adipose tissue. Mol. Cell. Biol. 29, 4467–4483 (2009).
Strissel, K.J. et al. Adipocyte death, adipose tissue remodeling and obesity complications. Diabetes 56, 2910–2918 (2007).
So, A. & Thorens, B. Uric acid transport and disease. J. Clin. Invest. 120, 1791–1799 (2010).
Feig, D.I., Kang, D.H. & Johnson, R.J. Uric acid and cardiovascular risk. N. Engl. J. Med. 359, 1811–1821 (2008).
Strissel, K.J. et al. T-cell recruitment and TH1 polarization in adipose tissue during diet-induced obesity in C57BL/6 mice. Obesity 18, 1918–1925 (2010).
Duffaut, C. et al. Interplay between human adipocytes and T lymphocytes in obesity: CCL20 as an adipochemokine and T lymphocytes as lipogenic modulators. Arterioscler. Thromb. Vasc. Biol. 29, 1608–1614 (2009).
Zhou, R., Tardivel, A., Thorens, B., Choi, I. & Tschopp, J. Thioredoxin-interacting protein links oxidative stress to inflammasome activation. Nat. Immunol. 11, 136–140 (2010).
Dominguez, H. et al. Metabolic and vascular effects of tumor necrosis factor-alpha blockade with etanercept in obese patients with type 2 diabetes. J. Vasc. Res. 42, 517–525 (2005).
Lamkanfi, M. et al. Glyburide inhibits the Cryopyrin/Nlrp3 inflammasome. J. Cell Biol. 187, 61–70 (2009).
Acknowledgements
We thank Vishva M. Dixit at Genentech for providing caspase-1–specific antibody and Nlrp3−/− mice and J. Suttles from the University of Louisville for L929 media. We also thank S. Bond for expert technical assistance and D.H. Ryan, C. Bouchard and J.M. Salbaum for helpful discussions. This work was supported in part by pilot grants to B.V. and V.D.D. from the Nutrition and Obesity Research Center (US National Institutes of Health (NIH) National Institute of Diabetes and Digestive and Kidney Diseases grant P30 DK072476). The research in the Dixit laboratory is supported in part by the NIH (R01AG31797), the Coypu Foundation and the Pennington Biomedical Research Foundation. K.S. was partially supported by the NIH (DK083615). This work used the facilities of the Genomics and Cell Biology & Bioimaging Core supported by NIH grant 1 P20 RR02/1945.
Author information
Authors and Affiliations
Contributions
B.V. performed real-time PCRs, flow cytometry assays, adipose tissue macrophage selections, some western blots, ITT, GTT and cytokine assays, managed the transgenic animal colony and participated in experimental design, data analysis and manuscript preparation. Y.-H.Y. performed all caspase-1 and IL1β western blots, adipose and liver histologies, and macrophage culture experiments and analyzed the data. A.R. Performed body composition analysis, tissue collections, lipid analysis, animal husbandry, genotyping, ITT, GTT and adipocyte size measurement. J.E.G. and E.R. designed and supervised the human studies, analyzed the glucose and insulin sensitivity data in obese T2DM subjects and discussed the hypotheses. K.S. performed the caspase-1 western blot in the kidneys of HFD fed WT and Nlrp3 null mice. R.L.M. participated in standardizing the ITT and GTT assays and advised on the design of experiments for liver fatty acid synthesis and oxidation gene expression. J.M.S. performed the western blots for some of the insulin-signaling experiments, helped with data interpretation, discussed the hypotheses and participated in manuscript preparation. V.D.D. conceived the project, designed the experiments, performed some of the cytokine assays and flow cytometry, helped with data interpretation, participated in data analysis, directed the project and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 and Supplementary Table 1 (PDF 374 kb)
Rights and permissions
About this article
Cite this article
Vandanmagsar, B., Youm, YH., Ravussin, A. et al. The NLRP3 inflammasome instigates obesity-induced inflammation and insulin resistance. Nat Med 17, 179–188 (2011). https://doi.org/10.1038/nm.2279
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2279