Abstract
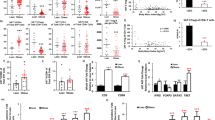
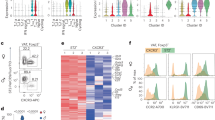
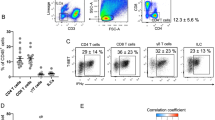
Obesity is accompanied by chronic, low-grade inflammation of adipose tissue, which promotes insulin resistance and type-2 diabetes. These findings raise the question of how fat inflammation can escape the powerful armamentarium of cells and molecules normally responsible for guarding against a runaway immune response. CD4+ Foxp3+ T regulatory (Treg) cells with a unique phenotype were highly enriched in the abdominal fat of normal mice, but their numbers were strikingly and specifically reduced at this site in insulin-resistant models of obesity. Loss-of-function and gain-of-function experiments revealed that these Treg cells influenced the inflammatory state of adipose tissue and, thus, insulin resistance. Cytokines differentially synthesized by fat-resident regulatory and conventional T cells directly affected the synthesis of inflammatory mediators and glucose uptake by cultured adipocytes. These observations suggest that harnessing the anti-inflammatory properties of Treg cells to inhibit elements of the metabolic syndrome may have therapeutic potential.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Accession codes
References
Shoelson, S.E., Lee, J. & Goldfine, A.B. Inflammation and insulin resistance. J. Clin. Invest. 116, 1793–1801 (2006).
Hotamisligil, G.S., Shargill, N.S. & Spiegelman, B.M. Adipose expression of tumor necrosis factor-α: direct role in obesity-linked insulin resistance. Science 259, 87–91 (1993).
Cai, D. et al. Local and systemic insulin resistance resulting from hepatic activation of IKKβand NF-κB. Nat. Med. 11, 183–190 (2005).
Bosello, O. & Zamboni, M. Visceral obesity and metabolic syndrome. Obes. Rev. 1, 47–56 (2000).
Fantuzzi, G. Adipose tissue, adipokines and inflammation. J. Allergy Clin. Immunol. 115, 911–919 (2005).
Weisberg, S.P. et al. Obesity is associated with macrophage accumulation in adipose tissue. J. Clin. Invest. 112, 1796–1808 (2003).
Xu, H. et al. Chronic inflammation in fat plays a crucial role in the development of obesity-related insulin resistance. J. Clin. Invest. 112, 1821–1830 (2003).
Lumeng, C.N., Bodzin, J.L. & Saltiel, A.R. Obesity induces a phenotypic switch in adipose tissue macrophage polarization. J. Clin. Invest. 117, 175–184 (2007).
Lumeng, C.N., Deyoung, S.M., Bodzin, J.L. & Saltiel, A.R. Increased inflammatory properties of adipose tissue macrophages recruited during diet-induced obesity. Diabetes 56, 16–23 (2007).
Odegaard, J.I. et al. Macrophage-specific PPARgamma controls alternative activation and improves insulin resistance. Nature 447, 1116–1120 (2007).
Suganami, T., Nishida, J. & Ogawa, Y. A paracrine loop between adipocytes and macrophages aggravates inflammatory changes: role of free fatty acids and tumor necrosis factor α. Arterioscler. Thromb. Vasc. Biol. 25, 2062–2068 (2005).
Caspar-Bauguil, S. et al. Adipose tissues as an ancestral immune organ: site-specific change in obesity. FEBS Lett. 579, 3487–3492 (2005).
Zheng, Y. & Rudensky, A.Y. Foxp3 in control of the regulatory T cell lineage. Nat. Immunol. 8, 457–462 (2007).
Sakaguchi, S., Yamaguchi, T., Nomura, T. & Ono, M. Regulatory T cells and immune tolerance. Cell 133, 775–787 (2008).
Maloy, K.J. et al. CD4+CD25+ TR cells suppress innate immune pathology through cytokine-dependent mechanisms. J. Exp. Med. 197, 111–119 (2003).
Murphy, T.J., Choileain, N.N., Zang, Y., Mannick, J.A. & Lederer, J.A. CD4+CD25+ regulatory T cells control innate immune reactivity after injury. J. Immunol. 174, 2957–2963 (2005).
Nguyen, L.T., Jacobs, J., Mathis, D. & Benoist, C. Where FoxP3-dependent regulatory T cells impinge on the development of inflammatory arthritis. Arthritis Rheum. 56, 509–520 (2007).
Wu, H. et al. T-cell accumulation and regulated on activation, normal T cell expressed and secreted upregulation in adipose tissue in obesity. Circulation 115, 1029–1038 (2007).
Tran, T.T., Yamamoto, Y., Gesta, S. & Kahn, C.R. Beneficial effects of subcutaneous fat transplantation on metabolism. Cell Metab. 7, 410–420 (2008).
Cinti, S. et al. Adipocyte death defines macrophage localization and function in adipose tissue of obese mice and humans. J. Lipid Res. 46, 2347–2355 (2005).
Fontenot, J.D. et al. Regulatory T cell lineage specification by the forkhead transcription factor Foxp3. Immunity 22, 329–341 (2005).
Huehn, J. et al. Developmental stage, phenotype, and migration distinguish naive- and effector/memory-like CD4+ regulatory T cells. J. Exp. Med. 199, 303–313 (2004).
Herman, A.E., Freeman, G.J., Mathis, D. & Benoist, C. CD4+CD25+ T regulatory cells dependent on ICOS promote regulation of effector cells in the prediabetic lesion. J. Exp. Med. 199, 1479–1489 (2004).
Hill, J. et al. Foxp3-transcription-factor–dependent and –independent regulation of the regulatory T cell transcriptional signature. Immunity 27, 786–800 (2007).
Nolan, K.F. et al. IL-10–conditioned dendritic cells, decommissioned for recruitment of adaptive immunity, elicit innate inflammatory gene products in response to danger signals. J. Immunol. 172, 2201–2209 (2004).
Wong, J. et al. Adaptation of TCR repertoires to self-peptides in regulatory and nonregulatory CD4+ T cells. J. Immunol. 178, 7032–7041 (2007).
Hsieh, C.S., Zheng, Y., Liang, Y., Fontenot, J.D. & Rudensky, A.Y. An intersection between the self-reactive regulatory and nonregulatory T cell receptor repertoires. Nat. Immunol. 7, 401–410 (2006).
Pacholczyk, R., Ignatowicz, H., Kraj, P. & Ignatowicz, L. Origin and T cell receptor diversity of Foxp3+CD4+CD25+ T cells. Immunity 25, 249–259 (2006).
Kretschmer, K. et al. Inducing and expanding regulatory T cell populations by foreign antigen. Nat. Immunol. 6, 1219–1227 (2005).
Correia-Neves, M., Waltzinger, C., Mathis, D. & Benoist, C. The shaping of the T cell repertoire. Immunity 14, 21–32 (2001).
Samad, F., Yamamoto, K., Pandey, M. & Loskutoff, D.J. Elevated expression of transforming growth factor-β in adipose tissue from obese mice. Mol. Med. 3, 37–48 (1997).
Chen, W. et al. Conversion of peripheral CD4+CD25− naive T cells to CD4+CD25+ regulatory T cells by TGF-β induction of transcription factor Foxp3. J. Exp. Med. 198, 1875–1886 (2003).
Peng, Y., Laouar, Y., Li, M.O., Green, E.A. & Flavell, R.A. TGF-β regulates in vivo expansion of Foxp3-expressing CD4+CD25+ regulatory T cells responsible for protection against diabetes. Proc. Natl. Acad. Sci. USA 101, 4572–4577 (2004).
Marie, J.C., Letterio, J.J., Gavin, M. & Rudensky, A.Y. TGF-β1 maintains suppressor function and Foxp3 expression in CD4+CD25+ regulatory T cells. J. Exp. Med. 201, 1061–1067 (2005).
Pelleymounter, M.A. et al. Effects of the obese gene product on body weight regulation in ob/ob mice. Science 269, 540–543 (1995).
Klebig, M.L., Wilkinson, J.E., Geisler, J.G. & Woychik, R.P. Ectopic expression of the agouti gene in transgenic mice causes obesity, features of type II diabetes and yellow fur. Proc. Natl. Acad. Sci. USA 92, 4728–4732 (1995).
De Rosa, V. et al. A key role of leptin in the control of regulatory T cell proliferation. Immunity 26, 241–255 (2007).
Kim, J.M., Rasmussen, J.P. & Rudensky, A.Y. Regulatory T cells prevent catastrophic autoimmunity throughout the lifespan of mice. Nat. Immunol. 8, 191–197 (2007).
Bennett, C.L. & Clausen, B.E. DC ablation in mice: promises, pitfalls and challenges. Trends Immunol. 28, 525–531 (2007).
Thorburn, J., Frankel, A.E. & Thorburn, A. Apoptosis by leukemia cell–targeted diphtheria toxin occurs via receptor-independent activation of Fas-associated death domain protein. Clin. Cancer Res. 9, 861–865 (2003).
Miyake, Y. et al. Protective role of macrophages in noninflammatory lung injury caused by selective ablation of alveolar epithelial type II cells. J. Immunol. 178, 5001–5009 (2007).
Bennett, C.L. et al. Inducible ablation of mouse Langerhans cells diminishes but fails to abrogate contact hypersensitivity. J. Cell Biol. 169, 569–576 (2005).
Duffield, J.S. et al. Conditional ablation of macrophages halts progression of crescentic glomerulonephritis. Am. J. Pathol. 167, 1207–1219 (2005).
Duffield, J.S. et al. Selective depletion of macrophages reveals distinct, opposing roles during liver injury and repair. J. Clin. Invest. 115, 56–65 (2005).
Walzer, T. et al. Identification, activation and selective in vivo ablation of mouse NK cells via NKp46. Proc. Natl. Acad. Sci. USA 104, 3384–3389 (2007).
Yuan, M. et al. Reversal of obesity- and diet-induced insulin resistance with salicylates or targeted disruption of Ikkbeta. Science 293, 1673–1677 (2001).
Boyman, O., Kovar, M., Rubinstein, M.P., Surh, C.D. & Sprent, J. Selective stimulation of T cell subsets with antibody-cytokine immune complexes. Science 311, 1924–1927 (2006).
Tang, Q. et al. Central role of defective interleukin-2 production in the triggering of islet autoimmune destruction. Immunity 28, 687–697 (2008).
Wolf, A.M., Wolf, D., Rumpold, H., Enrich, B. & Tilg, H. Adiponectin induces the anti-inflammatory cytokines IL-10 and IL-1RA in human leukocytes. Biochem. Biophys. Res. Commun. 323, 630–635 (2004).
Kumada, M. et al. Adiponectin specifically increased tissue inhibitor of metalloproteinase-1 through interleukin-10 expression in human macrophages. Circulation 109, 2046–2049 (2004).
Kim, H.J. et al. Differential effects of interleukin-6 and -10 on skeletal muscle and liver insulin action in vivo. Diabetes 53, 1060–1067 (2004).
Blüher, M. et al. Association of interleukin-6, C-reactive protein, interleukin-10 and adiponectin plasma concentrations with measures of obesity, insulin sensitivity and glucose metabolism. Exp. Clin. Endocrinol. Diabetes 113, 534–537 (2005).
Scarpelli, D. et al. Variants of the interleukin-10 promoter gene are associated with obesity and insulin resistance but not type 2 diabetes in Caucasian Italian subjects. Diabetes 55, 1529–1533 (2006).
Doganci, A. et al. The IL-6R α chain controls lung CD4+CD25+ Treg development and function during allergic airway inflammation in vivo. J. Clin. Invest. 115, 313–325 (2005).
Wan, S., Xia, C. & Morel, L. IL-6 produced by dendritic cells from lupus-prone mice inhibits CD4+CD25+ T cell regulatory functions. J. Immunol. 178, 271–279 (2007).
Hosogai, N. et al. Adipose tissue hypoxia in obesity and its impact on adipocytokine dysregulation. Diabetes 56, 901–911 (2007).
Furukawa, S. et al. Increased oxidative stress in obesity and its impact on metabolic syndrome. J. Clin. Invest. 114, 1752–1761 (2004).
Reich, M. et al. GenePattern 2.0. Nat. Genet. 38, 500–501 (2006).
Acknowledgements
We thank D. Littman (New York University) for the DTR construct, L. Roser and K. Hattori for assistance with mice, S. Rudensky (Memorial Sloan Kettering Cancer Center) for providing us with Foxp3DTR mice, J. LaVecchio and G. Buruzala for flow cytometry and J. Hill, J. Perez and R. Melamed for help with the microarray analysis. This work was supported by Young Chair funds to D.M. and C.B., by the US National Institutes of Health (DK51729 and DK73547) and Adler Chair funds to S.S. and by Joslin's National Institutes of Diabetes and Digestive and Kidney Diseases–funded Diabetes and Endocrinology Research Center core facilities. Postdoctoral fellowship support for M.F. was from the German Research Foundation (Emmy-Noether Fellowship, FE 801/1-1) and the Charles A. King Trust Postdoctoral Fellowship, and for L.H. from the Ministry of Science of Spain. J.W. and D.C. were supported by predoctoral fellowships from the US National Institutes of Health (T32 DK7260) and the European School of Molecular Medicine, respectively.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figs. 1–8, Supplementary Tables 1–4 and Supplementary Methods (PDF 6767 kb)
Supplementary Table 5
Characteristics of adipose tissue donors (XLS 95 kb)
Rights and permissions
About this article
Cite this article
Feuerer, M., Herrero, L., Cipolletta, D. et al. Lean, but not obese, fat is enriched for a unique population of regulatory T cells that affect metabolic parameters. Nat Med 15, 930–939 (2009). https://doi.org/10.1038/nm.2002
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2002
This article is cited by
-
Macrophage and T cell networks in adipose tissue
Nature Reviews Endocrinology (2024)
-
Association between systemic immune-inflammation index and insulin resistance and mortality
Scientific Reports (2024)
-
Wnt10b knockdown regulates the relative balance of adipose tissue-resident T cells and inhibits white fat deposition
Molecular Biology Reports (2024)
-
E-GWAS: an ensemble-like GWAS strategy that provides effective control over false positive rates without decreasing true positives
Genetics Selection Evolution (2023)
-
Obesity and atrial fibrillation: a narrative review from arrhythmogenic mechanisms to clinical significance
Cardiovascular Diabetology (2023)