Abstract
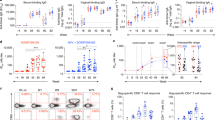
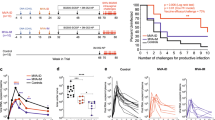
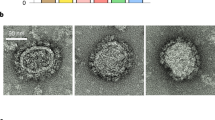
Neutralizing antibodies are thought to be crucial for HIV vaccine protection, but studies in animal models suggest that high antibody concentrations are required1. This is a major potential hurdle for vaccine design. However, these studies typically apply a large virus inoculum to ensure infection in control animals in single-challenge experiments. In contrast, most human infection via sexual encounter probably involves repeated exposures to much lower doses of virus2,3,4. Therefore, animal studies may have provided an overestimate of the levels of antibodies required for protection in humans. We investigated whether plasma concentrations of antibody corresponding to relatively modest neutralization titers in vitro could protect macaques from repeated intravaginal exposure to low doses of a simian immunodeficiency virus–HIV chimera (SHIV) that uses the CC chemokine receptor 5 (CCR5) co-receptor. An effector function–deficient variant of the neutralizing antibody was also included. The results show that a substantially larger number of challenges is required to infect macaques treated with neutralizing antibody than control antibody–treated macaques, and support the notion that effector function may contribute to antibody protection. Overall, the results imply that lower amounts of antibody than previously considered protective may provide benefit in the context of typical human exposure to HIV-1.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Mascola, J.R. Defining the protective antibody response for HIV-1. Curr. Mol. Med. 3, 209–216 (2003).
Wawer, M.J. et al. Rates of HIV-1 transmission per coital act, by stage of HIV-1 infection, in Rakai, Uganda. J. Infect. Dis. 191, 1403–1409 (2005).
Hollingsworth, T.D., Anderson, R.M. & Fraser, C. HIV-1 transmission, by stage of infection. J. Infect. Dis. 198, 687–693 (2008).
Royce, R.A., Sena, A., Cates, W. Jr. & Cohen, M.S. Sexual transmission of HIV. N. Engl. J. Med. 336, 1072–1078 (1997).
Gauduin, M.C. et al. Passive immunization with a human monoclonal antibody protects hu-PBL-SCID mice against challenge by primary isolates of HIV-1. Nat. Med. 3, 1389–1393 (1997).
Shibata, R. et al. Neutralizing antibody directed against the HIV-1 envelope glycoprotein can completely block HIV-1/SIV chimeric virus infections of macaque monkeys. Nat. Med. 5, 204–210 (1999).
Mascola, J.R. et al. Protection of macaques against pathogenic simian/human immunodeficiency virus 89.6PD by passive transfer of neutralizing antibodies. J. Virol. 73, 4009–4018 (1999).
Mascola, J.R. et al. Protection of macaques against vaginal transmission of a pathogenic HIV-1/SIV chimeric virus by passive infusion of neutralizing antibodies. Nat. Med. 6, 207–210 (2000).
Baba, T.W. et al. Human neutralizing monoclonal antibodies of the IgG1 subtype protect against mucosal simian-human immunodeficiency virus infection. Nat. Med. 6, 200–206 (2000).
Parren, P.W. et al. Antibody protects macaques against vaginal challenge with a pathogenic R5 simian/human immunodeficiency virus at serum levels giving complete neutralization in vitro. J. Virol. 75, 8340–8347 (2001).
Nishimura, Y. et al. Determination of a statistically valid neutralization titer in plasma that confers protection against simian-human immunodeficiency virus challenge following passive transfer of high-titered neutralizing antibodies. J. Virol. 76, 2123–2130 (2002).
Chakraborty, H. et al. Viral burden in genital secretions determines male-to-female sexual transmission of HIV-1: a probabilistic empiric model. AIDS 15, 621–627 (2001).
Pilcher, C.D. et al. Brief but efficient: acute HIV infection and the sexual transmission of HIV. J. Infect. Dis. 189, 1785–1792 (2004).
Otten, R.A. et al. Multiple vaginal exposures to low doses of R5 simian-human immunodeficiency virus: strategy to study HIV preclinical interventions in nonhuman primates. J. Infect. Dis. 191, 164–173 (2005).
Klasse, P.J., Shattock, R.J. & Moore, J.P. Which topical microbicides for blocking HIV-1 transmission will work in the real world? PLoS Med. 3, e351 (2006).
Kim, C.N. et al. Repetitive exposures with simian/human immunodeficiency viruses: strategy to study HIV pre-clinical interventions in non-human primates. J. Med. Primatol. 35, 210–216 (2006).
Burton, D.R. et al. Efficient neutralization of primary isolates of HIV-1 by a recombinant human monoclonal antibody. Science 266, 1024–1027 (1994).
Binley, J.M. et al. Comprehensive cross-clade neutralization analysis of a panel of anti-human immunodeficiency virus type 1 monoclonal antibodies. J. Virol. 78, 13232–13252 (2004).
Zhou, T. et al. Structural definition of a conserved neutralization epitope on HIV-1 gp120. Nature 445, 732–737 (2007).
Hessell, A.J. et al. Fc receptor but not complement binding is important in antibody protection against HIV. Nature 449, 101–104 (2007).
Subbarao, S. et al. Direct stringency comparison of two macaque models (single-high vs. repeat-low) for mucosal HIV transmission using an identical anti-HIV chemoprophylaxis intervention. J. Med. Primatol. 36, 238–243 (2007).
Regoes, R.R., Longini, I.M., Feinberg, M.B. & Staprans, S.I. Preclinical assessment of HIV vaccines and microbicides by repeated low-dose virus challenges. PLoS Med. 2, e249 (2005).
Dhillon, A.K. et al. Dissecting the neutralizing antibody specificities of broadly neutralizing sera from HIV-1 infected donors. J. Virol. 81, 6548–6562 (2007).
Li, Y. et al. Broad HIV-1 neutralization mediated by CD4-binding site antibodies. Nat. Med. 13, 1032–1034 (2007).
Sather, D.N. et al. Factors associated with the development of cross-reactive neutralizing antibodies during human immunodeficiency virus type 1 infection. J. Virol. 83, 757–769 (2009).
Doria-Rose, N.A. et al. Frequency and phenotype of human immunodeficiency virus envelope-specific B cells from patients with broadly cross-neutralizing antibodies. J. Virol. 83, 188–199 (2009).
Li, Y. et al. Analysis of neutralization specificities in polyclonal sera derived from human immunodeficiency virus type 1-infected individuals. J. Virol. 83, 1045–1059 (2009).
Harouse, J.M. et al. Mucosal transmission and induction of simian AIDS by CCR5-specific simian/human immunodeficiency virus SHIV(SF162P3). J. Virol. 75, 1990–1995 (2001).
Tan, R.C., Harouse, J.M., Gettie, A. & Cheng-Mayer, C. In vivo adaptation of SHIV(SF162): chimeric virus expressing a NSI, CCR5-specific envelope protein. J. Med. Primatol. 28, 164–168 (1999).
Hezareh, M., Hessell, A.J., Jensen, R.C., van de Winkel, J.G. & Parren, P.W. Effector function activities of a panel of mutants of a broadly neutralizing antibody against human immunodeficiency virus type 1. J. Virol. 75, 12161–12168 (2001).
Friedrich, T.C. et al. Subdominant CD8+ T-cell responses are involved in durable control of AIDS virus replication. J. Virol. 81, 3465–3476 (2007).
Cline, A.N., Bess, J.W., Piatak, M. Jr. & Lifson, J.D. Highly sensitive SIV plasma viral load assay: practical considerations, realistic performance expectations and application to reverse engineering of vaccines for AIDS. J. Med. Primatol. 34, 303–312 (2005).
Richman, D.D., Wrin, T., Little, S.J. & Petropoulos, C.J. Rapid evolution of the neutralizing antibody response to HIV type 1 infection. Proc. Natl. Acad. Sci. USA 100, 4144–4149 (2003).
Yant, L.J. et al. The high-frequency major histocompatibility complex class I allele Mamu-B*17 is associated with control of simian immunodeficiency virus SIVmac239 replication. J. Virol. 80, 5074–5077 (2006).
Kaizu, M. et al. Molecular typing of major histocompatibility complex class I alleles in the Indian rhesus macaque which restrict SIV CD8+ T cell epitopes. Immunogenetics 59, 693–703 (2007).
Acknowledgements
ELISA for recombinant monomeric HIV-1 gp120JR-FL was provided by Progenics. We thank K. Saye-Francisco for antibody production and quality control assistance at The Scripps Research Institute and S. Hoffenberg at the IAVI AIDS Vaccine Design Laboratory. We thank A. Haahr Andreasen for statistical advice and analysis. We are grateful for the assistance provided by E. Rakasz, G. Borchardt, and C. McNair with genotyping and viral load assessments at the Wisconsin National Primate Research Center. We also thank M. Huber and R. Astronomo for reviewing the manuscript. Support for this work was provided by US National Institutes of Health (NIH) grant AI55332 to D.R.B., by the Neutralizing Antibody Consortium of the International AIDS Vaccine Initiative and by the Swiss National Foundation, Fellowship PA00A-109033.
Author information
Authors and Affiliations
Contributions
Project planning was performed by A.J.H., L.H., P.A.M. and D.R.B.; experimental work was done by A.J.H., L.H., M.H. and D.M.T.; data analysis was done by A.J.H., L.H., P.P., W.K.B., P.W.H.I.P. and D.R.B.; and A.J.H., P.P., P.W.H.I.P. and D.R.B. composed the manuscript.
Ethics declarations
Competing interests
D.R.B. is listed as an inventor on patents describing the human neutralizing antibody b12. These patents include Australia Patent No. 681360, Patent Co-operation Treaty (PCT) Patent No. WO 94/07922, US Patent No. 5,652,138, Australia Patent No. 706601, US Patent No. 5,804,440 and PCT Patent No. WO 96/02273. Patents with the patent offices of Canada, the EU, Finland, Japan and Norway are pending. D.R.B. is also listed on patents describing the synthetic human neutralizing antibody, including the continuation-in-part PCT Patent No. 95/11317 and patents pending in the patent offices of Canada, the EU and Japan.
Supplementary information
Supplementary Text and Figures
Supplementary Figs. 1–4 and Supplementary Tables 1–3 (PDF 868 kb)
Rights and permissions
About this article
Cite this article
Hessell, A., Poignard, P., Hunter, M. et al. Effective, low-titer antibody protection against low-dose repeated mucosal SHIV challenge in macaques. Nat Med 15, 951–954 (2009). https://doi.org/10.1038/nm.1974
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.1974
This article is cited by
-
Antiviral neutralizing antibodies: from in vitro to in vivo activity
Nature Reviews Immunology (2023)
-
Mucosal application of the broadly neutralizing antibody 10-1074 protects macaques from cell-associated SHIV vaginal exposure
Nature Communications (2023)
-
Enhanced HIV SOSIP Envelope yields in plants through transient co-expression of peptidyl-prolyl isomerase B and calreticulin chaperones and ER targeting
Scientific Reports (2022)
-
Impact of adjuvants on the biophysical and functional characteristics of HIV vaccine-elicited antibodies in humans
npj Vaccines (2022)
-
Engaging innate immunity in HIV-1 cure strategies
Nature Reviews Immunology (2022)