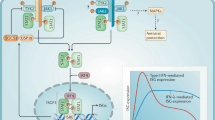
Abstract
Interferon-λ (IFN-λ) is a central regulator of mucosal immunity; however, its signaling specificity relative to that of type I interferons is poorly defined. IFN-λ can induce antiviral interferon-stimulated genes (ISGs) in epithelia, while the effect of IFN-λ in non-epithelial cells remains unclear. Here we report that neutrophils responded to IFN-λ. We found that in addition to inducing ISG transcription, IFN-λ (but not IFN-β) specifically activated a translation-independent signaling pathway that diminished the production of reactive oxygen species and degranulation in neutrophils. In mice, IFN-λ was elicited by enteric viruses and acted on neutrophils to decrease oxidative stress and intestinal damage. Thus, IFN-λ acted as a unique immunomodulatory agent by modifying transcriptional and non-translational neutrophil responses, which might permit a controlled development of the inflammatory process.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Schneider, W.M., Chevillotte, M.D. & Rice, C.M. Interferon-stimulated genes: a complex web of host defenses. Annu. Rev. Immunol. 32, 513–545 (2014).
Baldridge, M.T. et al. Commensal microbes and interferon-λ determine persistence of enteric murine norovirus infection. Science 347, 266–269 (2015).
Pott, J. et al. IFN-lambda determines the intestinal epithelial antiviral host defense. Proc. Natl. Acad. Sci. USA 108, 7944–7949 (2011).
Mahlakõiv, T., Hernandez, P., Gronke, K., Diefenbach, A. & Staeheli, P. Leukocyte-derived IFN-α/β and epithelial IFN-λ constitute a compartmentalized mucosal defense system that restricts enteric virus infections. PLoS Pathog. 11, e1004782 (2015).
Lin, J.D. et al. distinct roles of type I and type III interferons in intestinal immunity to homologous and heterologous rotavirus infections. PLoS Pathog. 12, e1005600 (2016).
Hernández, P.P. et al. Interferon-λ and interleukin 22 act synergistically for the induction of interferon-stimulated genes and control of rotavirus infection. Nat. Immunol. 16, 698–707 (2015).
Zhou, Z. et al. Type III interferon (IFN) induces a type I IFN-like response in a restricted subset of cells through signaling pathways involving both the Jak-STAT pathway and the mitogen-activated protein kinases. J. Virol. 81, 7749–7758 (2007).
Doyle, S.E. et al. Interleukin-29 uses a type 1 interferon-like program to promote antiviral responses in human hepatocytes. Hepatology 44, 896–906 (2006).
Marcello, T. et al. Interferons alpha and lambda inhibit hepatitis C virus replication with distinct signal transduction and gene regulation kinetics. Gastroenterology 131, 1887–1898 (2006).
Souza-Fonseca-Guimaraes, F. et al. NK cells require IL-28R for optimal in vivo activity. Proc. Natl. Acad. Sci. USA 112, E2376–E2384 (2015).
Yin, Z. et al. Type III IFNs are produced by and stimulate human plasmacytoid dendritic cells. J. Immunol. 189, 2735–2745 (2012).
Finotti, G., Tamassia, N., Calzetti, F., Fattovich, G. & Cassatella, M.A. Endogenously produced TNF-α contributes to the expression of CXCL10/IP-10 in IFN-λ3-activated plasmacytoid dendritic cells. J. Leukoc. Biol. 99, 107–119 (2016).
Dai, J., Megjugorac, N.J., Gallagher, G.E., Yu, R.Y. & Gallagher, G. IFN-lambda1 (IL-29) inhibits GATA3 expression and suppresses Th2 responses in human naive and memory T cells. Blood 113, 5829–5838 (2009).
Megjugorac, N.J., Gallagher, G.E. & Gallagher, G. Modulation of human plasmacytoid DC function by IFN-λ1 (IL-29). J. Leukoc. Biol. 86, 1359–1363 (2009).
Blazek, K. et al. IFN-λ resolves inflammation via suppression of neutrophil infiltration and IL-1β production. J. Exp. Med. 212, 845–853 (2015).
de Groen, R.A. et al. IFN-λ-mediated IL-12 production in macrophages induces IFN-γ production in human NK cells. Eur. J. Immunol. 45, 250–259 (2015).
de Groen, R.A., Groothuismink, Z.M., Liu, B.S. & Boonstra, A. IFN-λ is able to augment TLR-mediated activation and subsequent function of primary human B cells. J. Leukoc. Biol. 98, 623–630 (2015).
Wolk, K. et al. IL-29 is produced by TH17 cells and mediates the cutaneous antiviral competence in psoriasis. Sci. Transl. Med. 5, 204ra129 (2013).
Sommereyns, C., Paul, S., Staeheli, P. & Michiels, T. IFN-lambda (IFN-λ) is expressed in a tissue-dependent fashion and primarily acts on epithelial cells in vivo. PLoS Pathog. 4, e1000017 (2008).
Mordstein, M. et al. Lambda interferon renders epithelial cells of the respiratory and gastrointestinal tracts resistant to viral infections. J. Virol. 84, 5670–5677 (2010).
Davidson, S. et al. IFNλ is a potent anti-influenza therapeutic without the inflammatory side effects of IFNα treatment. EMBO Mol. Med. 8, 1099–1112 (2016).
Kotenko, S.V. et al. IFN-lambdas mediate antiviral protection through a distinct class II cytokine receptor complex. Nat. Immunol. 4, 69–77 (2003).
Sheppard, P. et al. IL-28, IL-29 and their class II cytokine receptor IL-28R. Nat. Immunol. 4, 63–68 (2003).
Meager, A., Visvalingam, K., Dilger, P., Bryan, D. & Wadhwa, M. Biological activity of interleukins-28 and -29: comparison with type I interferons. Cytokine 31, 109–118 (2005).
Mócsai, A. Diverse novel functions of neutrophils in immunity, inflammation, and beyond. J. Exp. Med. 210, 1283–1299 (2013).
Ivashkiv, L.B. & Donlin, L.T. Regulation of type I interferon responses. Nat. Rev. Immunol. 14, 36–49 (2014).
Wegrzyn, J. et al. Function of mitochondrial Stat3 in cellular respiration. Science 323, 793–797 (2009).
Gough, D.J. et al. Mitochondrial STAT3 supports Ras-dependent oncogenic transformation. Science 324, 1713–1716 (2009).
Meier, J.A. et al. Stress-induced dynamic regulation of mitochondrial STAT3 and its association with cyclophilin D reduce mitochondrial ROS production. Sci. Signal. 10, eaag2588 (2017).
Odendall, C. et al. Diverse intracellular pathogens activate type III interferon expression from peroxisomes. Nat. Immunol. 15, 717–726 (2014).
El-Benna, J., Dang, P.M., Gougerot-Pocidalo, M.A., Marie, J.C. & Braut-Boucher, F. p47phox, the phagocyte NADPH oxidase/NOX2 organizer: structure, phosphorylation and implication in diseases. Exp. Mol. Med. 41, 217–225 (2009).
Reeves, E.P. et al. Direct interaction between p47phox and protein kinase C: evidence for targeting of protein kinase C by p47phox in neutrophils. Biochem. J. 344, 859–866 (1999).
Hoyal, C.R. et al. Modulation of p47PHOX activity by site-specific phosphorylation: Akt-dependent activation of the NADPH oxidase. Proc. Natl. Acad. Sci. USA 100, 5130–5135 (2003).
Chen, Q. et al. Akt phosphorylates p47phox and mediates respiratory burst activity in human neutrophils. J. Immunol. 170, 5302–5308 (2003).
El Benna, J. et al. Activation of p38 in stimulated human neutrophils: phosphorylation of the oxidase component p47phox by p38 and ERK but not by JNK. Arch. Biochem. Biophys. 334, 395–400 (1996).
Baldridge, M.T. et al. Expression of IFNLR1 on intestinal epithelial cells is critical to the antiviral effects of interferon λ against norovirus and reovirus. J. Virol. 91, e02079–16 (2017).
Yang, J.Y. et al. Enteric viruses ameliorate gut inflammation via Toll-like receptor 3 and Toll-like receptor 7-mediated interferon-β production. Immunity 44, 889–900 (2016).
Nice, T.J. et al. Interferon-λ cures persistent murine norovirus infection in the absence of adaptive immunity. Science 347, 269–273 (2015).
Galani, I.E. et al. Interferon-λ mediates non-redundant front-line antiviral protection against influenza virus infection without compromising host fitness. Immunity 46, 875–890 (2017).
Glocker, E.O. et al. Inflammatory bowel disease and mutations affecting the interleukin-10 receptor. N. Engl. J. Med. 361, 2033–2045 (2009).
Kotlarz, D. et al. Loss of interleukin-10 signaling and infantile inflammatory bowel disease: implications for diagnosis and therapy. Gastroenterology 143, 347–355 (2012).
Shouval, D.S. et al. Interleukin-10 receptor signaling in innate immune cells regulates mucosal immune tolerance and anti-inflammatory macrophage function. Immunity 40, 706–719 (2014).
Chiriac, M.T. et al. activation of epithelial signal transducer and activator of transcription 1 by interleukin 28 controls mucosal healing in mice with colitis and is increased in mucosa of patients with inflammatory bowel disease. Gastroenterology 153, 123–138 (2017).
Mócsai, A. et al. Kinase pathways in chemoattractant-induced degranulation of neutrophils: the role of p38 mitogen-activated protein kinase activated by Src family kinases. J. Immunol. 164, 4321–4331 (2000).
Chassaing, B., Aitken, J.D., Malleshappa, M. & Vijay-Kumar, M. Dextran sulfate sodium (DSS)-induced colitis in mice. Curr. Protoc. Immunol. 104, Unit 15 25 (2014).
Acknowledgements
We thank S.B. Snapper (Harvard Medical School) for Stat1−/− and LysMcreStat3fl/fl mice; the Wellcome Trust Sanger Institute Mouse Genetics Project (Sanger MGP) and its funders for the mutant mouse line Ifnlr1tm1a(EUCOMM)Wtsi; H.W. Virgin (Washington University) for the Ifnlr1tm1a(EUCOMM)Wtsi mouse line backcrossed to Villincre-recombinase-expressing mice; and J.C. Kagan, L.R. Marek-Kagan, the Kagan Lab, S.B. Snapper, the Snapper Lab, A. Mocsai and H.W. Virgin for discussions, help and support. Supported by the US National Institutes of Health (1R01DK115217, 1R01AI121066-01A1 to I.Z.), the Harvard Digestive Diseases Center (P30 DK034854 to I.Z.), the Harvard Medical School Milton Fund (to I.Z.), Crohn's and Colitis Foundation of America (I.Z.), the Cariplo Foundation (I.Z. and F.G.), the Associazione Italiana per la Ricerca sul Cancro (F.G.), Fondazione Italiana di Ricerca per la SLA–Sclerosi Laterale Amiotrofica (ARISLA ) (F.G.), Fondazione Regionale per la Ricerca Biomedica (F.G.) and the Jane Coffin Child's Memorial Fund for Medical Research (Y.T.).
Author information
Authors and Affiliations
Contributions
A.B., F.G. and I.Z. designed the study; A.B. conducted most of the experiments; Y.T. prepared the IFNLR-expressing RAW cell line; F.G. and I.Z. conceived of the study; and I.Z. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Mature neutrophils are unique among immune cells in their capacity to respond to IFN-λ.
a) Representative cytofluorimetry plot of neutrophils purified from mouse BM on a percoll gradient. b) Bone marrow neutrophils were sorted based on the expression of the differentiation marker Ly6g and SSC intensity. Representative plots of sorted Ly6ghiSSChi mature neutrophils (left panel, black-line gate) and immature neutrophil precursors Ly6gint-hiSSCint (right panel, dotted gate) are shown. c) Expression of Ifnlr1 mRNA in different neutrophil populations: neutrophil purified as in (a) (percoll PMN), neutrophil sorted as depicted in (b) (Ly6ghi SSChi, PMN Ly6gint-hiSSCint), neutrophils sorted from blood (blood PMN), neutrophil sorted from the peritoneum after thioglycollate treatment (pPMN). Means and s.e.m. of 3 biological replicates are shown. Data are representative of one out of three experiments. d) Viperin induction in response to 100U/ml of either IFN-λ or IFN-β has been evaluated in BM neutrophils sorted as in b). Means and s.e.m. of 3 technical replicates are shown. Data are representative of one out of three experiments. e) Expression of mRNA encoding the two components of the IFN-λ receptor (Ifnlr1, left axis; Il10rb, right axis) was evaluated in BM neutrophils (PMN), conventional splenic dendritic cells (cDCs), CD4+ T cells, and peritoneal macrophages (pM0). Means and s.e.m. of 3 technical replicates are shown. Data are representative of one out of three experiments. f) Neutrophils from WT mice were stimulated either with IFN-λ or IFN-β (100U/ml). STAT1, STAT2 and STAT3 phosphorylation was evaluated by western blot at the indicated time points.
Supplementary Figure 2 IFN-λ modulates neutrophil function in a dose- and time-dependent manner.
a) Superoxide release was measured in bone marrow-derived neutrophils pre-treated or not with different doses of IFN-λ (20, 50 or 100 U/ml) and then stimulated with 10 μg/ml LPS. % of inhibition, compared to neutrophils treated with LPS alone, 30 minutes after stimulation is shown (b). Superoxide release in response to 100 ng/ml TNF has been measured in bone marrow derive neutrophils pre-treated or not with two different doses of IFN-λ (50 or 100 U/ml). % of inhibition 30 min after stimulation (left panel) and representative kinetic analysis (right panel) are shown (c). Neutrophils were pre-treated for 30 minutes (pre) with IFN-λ and then stimulated with TNF or co-treated with IFN-λ and TNF (co). % of inhibition, compared to neutrophils treated with TNF alone, 30 minutes after stimulation (left panel) and representative kinetic analysis (right panel) are shown. Mean and s.e.m. of three biological replicates are shown. Representative results of one out of three experiments are shown. d) Neutrophil degranulation of tertiary granules was evaluated by zymography, measuring MMP-9 activity in cell supernatants of neutrophils treated with either 100 U/ml IFN-λ or 100 U/ml IFN-β and stimulated with 10 μg/ml LPS for the indicated time. Relative intensity to cells treated with LPS alone are shown. Mean and s.e.m. of three biological replicates are shown. Representative data of one out of three independent experiments are shown. e) LDH release was evaluated in supernatants of neutrophils treated with 100 U/ml IFN-λ and stimulated with 10 μg/ml LPS for 30 min, in the presence or absence of 10 μg/ml cyclohexamide (CHX). LDH released is expressed as % of LDH content of the same number of lysed cells. Mean and s.e.m. of three biological replicates are shown. Representative plot of three independent experiment is shown.
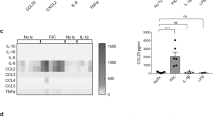
Supplementary Figure 3 Several neutrophil functions are similarly regulated by IFN-λ and IFN-β.
(a) Neutrophils were treated or not with IFN-λ or IFN-β (100 U/ml) and stimulated or not with LPS. Neutrophil apoptosis was evaluated by flow cytometry 6 hours after treatment. Cells were stained with Annexin V and 7-AAD. The percentage of live cells (Annexin V- 7-AAD-) is shown. b) Neutrophils were treated with either IFN-λ or IFN-β (100 U/ml) and incubated with eGFP expessing E. coli, either non-opsonized (E. coli) or opsonized (E. coli ops) with 5% murine serum, at a multiplicity of infection (M.O.I.) of 1, for 30 minutes. The percentage of neutrophils that have engulfed E. coli (E. coli+ neutrophils) is shown. c) Neutrophils were treated with either IFN-λ or IFN-β and subsequently stimulated over night with 10 μg/ml LPS. Cytokine production was measured by ELISA. a-c) Mean and s.e.m. of three biological replicates are shown. Data are representative of one out of three experiments. d) Neutrophils were treated with IFN-λ or IFN-β and stimulated with LPS (10 μg/ml). Gene transcription was measured by Nanostring technology using the nCounter Mouse Inflammation v2 panel. Genes modulated more than 2 folds that obtained a P<0.05 are shown.
Supplementary Figure 4 IFN-λ diminishes neutrophil function without involving the classic Jak–STAT transcriptional pathway.
a) Neutrophils were treated or not for 10 min with 10 μg/ml cycloheximide (CHX), and/or IFN-λ, and subsequently incubated for 30 min in the presence of puromycin. Total protein extracts were run in a SDS page gel and incorporation of a puromycin in newly synthetized proteins was evaluated by WB. Whole gel picture is displayed. b-d, f,g) Neutrophils were treated with IFN-λ in the presence of cycloheximide (CHX) (b,c), the STAT1 inhibitor fludarabine (1 mM) (d), the pan-Jak inhibitor Pyridone-6 (Py-6) 50μM (f), or the Jak-2 inhibitor AG490, 100 μM (g) and stimulated as indicated. ROS production (b,d,f,g) and MMP-9 secretion (c) were evaluated as described in Figure 2. b, d, e, g) Left panel: % inhibition of ROS production by neutrophils pre-treated with IFN-λ (100 U/ml) and stimulated with either TNFor LPS, compared to neutrophils stimulated with TNF or LPS alone, in the presence or absence of the indicated inhibitors, 30 min after stimulation. Mean and s.e.m. of three biological replicates are shown. Data representative of one out of three independent experiments. Right panel: representative kinetic analysis of the respiratory burst in the presence of the indicated inhibitors. e) Jak-2 phosphorylation was evaluated by WB in RAW cells expressing a tagged IFNLR1, in response to 100 U/ml of IFN-λ, in the presence or absence of the Jak-2 inhibitor HBC 10 μM. Data are representative of three independent experiments. h) Viperin induction was evaluated by qPCR on neutrophil treated with either the pan-Jak inhibitor Py-6 (50nM), or the Jak-2 inhibitor HBC (10μM) and stimulated with 100 U/ml IFN-λ, or IFN-β, for 3 hours. Mean and s.e.m. of three replicates of one out of three experiments are shown. i) Neutrophils were treated or not with 100 U/ml of IFN-λ and stimulated with 6mM PMA. ROS production was evaluated as described in Figure 2. Representative plot of three independent experiments is shown. j) Neutrophils were pretreated, or not, with 100U/ml IFN-λ and stimulated, or not, with 10μg/ml LPS. Immunoprecipitation for p47phox was performed. The presence of associated p67phox and RAC has been evaluated by WB. Representative data of three independent experiments is shown. e,j) blots have been cropped to show appropriate molecular weight bands. Statistical significance was evaluated by unpaired t test (b-d,f,g) or Two-way ANOVA (h) *: P < 0.05; **: P < 0.01; ***: P < 0,001.
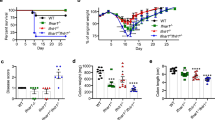
Supplementary Figure 5 Neutrophil migration and transcription of genes encoding inflammatory molecules are regulated similarly in wild-type and Ifnlr1–/– mice.
a) Neutrophils were treated or not with IFN-λ or IFN-β (100U/ml), seeded on top of a 3 μm transwell and 1mM fMLP was placed at the bottom of the transwell. Migration towards fMLP was measured as number of neutrophil recovered at the bottom of the transwell. Mean and s.e.m. of three replicates of one out of three experiments are shown. b-e) Wild type (WT) or IFNLR1-deficient (Ifnlr1-/-) animals were administered 2.5% DSS for 7 days and followed for 9 days. At days 5, 7 and 9, colons were harvested and the absolute number of CD45+, Ly6G+, CD11b+ neutrophils in the lamina propria was evaluated by flow cytometry (b). mRNA expression of Il-6 and Tnfa in the colons was evaluated by qPCR (c,d). The % of Annexin V- 7-AAD- neutrophils from the lamina propria of either WT or Ifnlr1-/- mice was quantified by flow cytometry (e). Representative data of one out of three independent experiments are shown. Mean and s.e.m. of 5 mice per group are depicted. Statistical significance was evaluated by One-way ANOVA (a), Two-way ANOVA (b) or unpaired t test (c-e) *: P < 0.05; **: P < 0.01; ***: P<0,001. f) Gene expression was evaluated on 100 ng of mRNA purified from total colon lysates using Nanostring technology with the nCounter Mouse Inflammation v2 panel. mRNA counts are depicted for genes that generated a count greater than 500 for each genotype.
Supplementary Figure 6 The protective effect of IFN-λ in DSS-induced colitis is dependent on its non-transcriptional regulation of neutrophil function.
a) Expression of Ifnlr1 was evaluated in immune cells populations sorted from the lamina propria of WT mice treated for 7 days with 2.5% DSS in the drinking water. B cells, CD4+ and CD8+ T cells, conventional dendritic cells (DCs), NK1.1+ cells, macrophages (MFs), Type 3 innate lymphoid cells (ILC3s, identified as Lin-, CD45+, CD90+, CD127+ cells), and neutrophils (PMN) were analyzed. Mean and s.e.m. of three biological replicates of one out of three experiments are shown. b,c) Bone marrow chimeras in which IFNLR1-deficient irradiated hosts were reconstituted with wild type bone marrow (Ifnlr1-/-→ WT) or control chimeras in which wild type hosts were reconstituted with wild type bone marrow (WT → WT) were created. The percentage of chimerism was evaluated by flow cytometry as % of cells of donor origin (CD45.2) vs. cells of recipient origin (CD45.1) in the blood of WT → WT (b), or Ifnlr1-/-→ WT (c) chimeric mice. Bulk leukocyte population (CD45+ cells) and neutrophil population (CD45+Ly6G+) were evaluated. d,e). WT → WT and WT → Ifnlr1-/- bone marrow chimeras were treated with 2.5% DSS of the drinking water for 7 days. Weight loss was evaluated in (d) and oxidative stress was evaluated as presence of 8-oxo guanine in (e). f) Expression of Ifnlr1 was evaluated by qPCR in BM neutrophils and IECs derived from Ifnlr1fl/flMRP8cre and Ifnlr1fl/fl control mice. Mean and s.e.m. of 3 biological replicates are shown. Data are representative of one out of three experiments. g) Viperin induction was evaluated by qPCR in neutrophils derived from Ifnlr1fl/flMRP8cre and Ifnlr1fl/fl mice, following treatment with 100 U/ml of IFN-λ, or IFN-β, for 3h. Mean and s.e.m. of 3 biological replicates are shown. Data are representative of one out of three experiments. h,i) Ifnlr1fl/flMRP8cre and Ifnlr1fl/fl littermates were treated with 2,5% DSS in the drinking water for 7 days. Weight loss (h) and PMN infiltration (i) (left panel: absolute numbers, Right panel: representative dot plots) were evaluated. Mean and s.e.m. of data obtained from 5 mice are shown. Data are representative of one out of three experiments. j) Gene expression was evaluated on 10,000 cells sorted from lamina propria (LP) as Ly6ghiCD11bhi (PMN) or (IEC) using Nanostring technology with the nCounter Mouse Inflammation v2 panel. mRNA counts are depicted. k,l) % of cells of donor origin (CD45.1) vs. cells of recipient origin (CD45.2) in the blood of WT → WT (k) or WT → Ifnlr1-/- (l) chimeric mice was evaluated by flow cytometry. Bulk leukocyte population (CD45+ cells) and neutrophil population (CD45+Ly6G+) were evaluated. m) Expression of the Ifnlr1 mRNA was evaluated by qPCR in IEC derived from either Ifnlr1fl/fl vilcre or Ifnlr1fl/fl mice. n) Ifnlr1fl/flvilcre and Ifnlr1fl/fl littermates were treated with 2,5% DSS in the drinking water for 7 days and weight loss was evaluated. Mean and s.e.m. of 5 animals/group is depicted. Statistical significance was evaluated with Two-way ANOVA (d,f,g) or nonparametric t test (e,h,i). *: P < 0.05; **: P < 0.01; ***: P < 0,001.
Supplementary Figure 7 After the induction of DSS-induced colitis, Ifnar–/– mice are as sensitive as wild-type mice to depletion of enteric virus.
a,b) Wild type (WT) mice were treated intra-gastrically (i.g.) with an anti-viral (AV) drug cocktail for 10 days. a) mRNA expression of Ifnl2-Ifnl3 by LP cells derived from the colon was determined by qPCR. Mean and s.e.m. of data collected from 5 mice are shown. One out of three experiments is shown. b) The presence of virus like particles (VLP) in the stool of mice treated (AV) or not (NT) with AV cocktail was evaluated by fluorescence microscopy. The number of VLP/field of view positive for either the DNA specific dye SYBR Gold (left panel) or the RNA specific dye SYBR Green II (right panel) is shown. Mean and s.e.m. of 10 mice per group are shown. One out of three independent experiments is shown. c-i) WT or IFNAR deficient (Ifnar -/-) mice were administered i.g. a cocktail of anti-viral drugs (AV), or PBS as control, for 10 days and given 2.5% DSS in the drinking water. After seven days mice were sacrificed and gene expression and colitis development was evaluated measuring colon length (c), disease activity at day 7 (d), histological features (e,g) and total weight loss at day 7 (f). Induction of Hmox1 (h) and Tnf (i) was also evaluated in IEC cells by qPCR. Representative data of one out of three independent experiments are shown. Mean and s.e.m. of 5 mice per group is depicted. Statistical significance was evaluated by Two-way ANOVA (c,d,e,f,h,i), or nonparametric t test (a). *: P < 0.05; **: P < 0.01; ***: P < 0,001.
Supplementary Figure 8 Administration of recombinant IFN-λ protects mice from DSS-induced colitis.
a-c) WT mice received 2.5% DSS in the drinking water for 7 days, then switched to water for up to 10 days. During DSS administration, the mice were treated daily with 1mg/kg of recombinant IFN-λ i.p. Colitis development was evaluated monitoring weight loss (a), colon length (b) and by histology (c). d) Schematic of the activity of IFN-λs vs. type I IFNs in neutrophils and its effect on intestinal inflammation. Mean and s.e.m. of 10 mice per group is depicted. One out of three experiments is shown. Statistical significance was evaluated with Two-way ANOVA(a) or One-way ANOVA (b) *: P < 0.05; **: P < 0.01; ***: P < 0,001.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 and Supplementary Tables 1–2.
Rights and permissions
About this article
Cite this article
Broggi, A., Tan, Y., Granucci, F. et al. IFN-λ suppresses intestinal inflammation by non-translational regulation of neutrophil function. Nat Immunol 18, 1084–1093 (2017). https://doi.org/10.1038/ni.3821
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.3821
This article is cited by
-
NLRP3 exacerbates EAE severity through ROS-dependent NET formation in the mouse brain
Cell Communication and Signaling (2024)
-
Interferon antagonists encoded by SARS-CoV-2 at a glance
Medical Microbiology and Immunology (2023)
-
A mammalian lung's immune system minimizes tissue damage by initiating five major sequential phases of defense
Clinical and Experimental Medicine (2023)
-
Mechanisms involved in controlling RNA virus-induced intestinal inflammation
Cellular and Molecular Life Sciences (2022)
-
Critical role of interferons in gastrointestinal injury repair
Nature Communications (2021)