Abstract
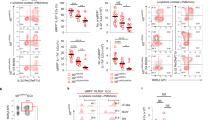
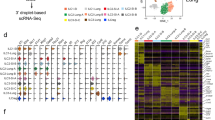
Type 2 innate lymphoid cells (ILC2 cells) participate in host defense against helminth parasites and in allergic inflammation. Given their functional relatedness to type 2 helper T cells (TH2 cells), we explored whether Gfi1 acts as a shared transcriptional determinant in ILC2 cells. Gfi1 promoted the development of ILC2 cells and controlled their responsiveness during infection with Nippostrongylus brasiliensis and protease allergen–induced lung inflammation. Gfi1 'preferentially' regulated the responsiveness of ILC2 cells to interleukin 33 (IL-33) by directly activating Il1rl1, which encodes the IL-33 receptor (ST2). Loss of Gfi1 in activated ILC2 cells resulted in impaired expression of the transcription factor GATA-3 and a dysregulated genome-wide effector state characterized by coexpression of IL-13 and IL-17. Our findings establish Gfi1 as a shared determinant that reciprocally regulates the type 2 and IL-17 effector states in cells of the innate and adaptive immune systems.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Spits, H. & Cupedo, T. Innate lymphoid cells: emerging insights in development, lineage relationships, and function. Annu. Rev. Immunol. 30, 647–675 (2012).
Zeng, H., Yucel, R., Kosan, C., Klein-Hitpass, L. & Moroy, T. Transcription factor Gfi1 regulates self-renewal and engraftment of hematopoietic stem cells. EMBO J. 23, 4116–4125 (2004).
Hock, H. et al. Gfi-1 restricts proliferation and preserves functional integrity of haematopoietic stem cells. Nature 431, 1002–1007 (2004).
Karsunky, H. et al. Inflammatory reactions and severe neutropenia in mice lacking the transcriptional repressor Gfi1. Nat. Genet. 30, 295–300 (2002).
Hock, H. et al. Intrinsic requirement for zinc finger transcription factor Gfi-1 in neutrophil differentiation. Immunity 18, 109–120 (2003).
Laslo, P. et al. Multilineage transcriptional priming and determination of alternate hematopoietic cell fates. Cell 126, 755–766 (2006).
Spooner, C.J., Cheng, J.X., Pujadas, E., Laslo, P. & Singh, H. A recurrent network involving the transcription factors PU.1 and Gfi1 orchestrates innate and adaptive immune cell fates. Immunity 31, 576–586 (2009).
Yücel, R., Karsunky, H., Klein-Hitpass, L. & Moroy, T. The transcriptional repressor Gfi1 affects development of early, uncommitted c-Kit+ T cell progenitors and CD4/CD8 lineage decision in the thymus. J. Exp. Med. 197, 831–844 (2003).
Zhu, J., Jankovic, D., Grinberg, A., Guo, L. & Paul, W.E. Gfi-1 plays an important role in IL-2-mediated Th2 cell expansion. Proc. Natl. Acad. Sci. USA 103, 18214–18219 (2006).
Zhu, J. et al. Down-regulation of Gfi-1 expression by TGF-β is important for differentiation of Th17 and CD103+ inducible regulatory T cells. J. Exp. Med. 206, 329–341 (2009).
Wong, S.H. et al. Transcription factor RORα is critical for nuocyte development. Nat. Immunol. 13, 229–236 (2012).
Yang, Q., Saenz, S.A., Zlotoff, D.A., Artis, D. & Bhandoola, A. Cutting edge: Natural helper cells derive from lymphoid progenitors. J. Immunol. 187, 5505–5509 (2011).
Yücel, R., Kosan, C., Heyd, F. & Moroy, T. Gfi1:green fluorescent protein knock-in mutant reveals differential expression and autoregulation of the growth factor independence 1 (Gfi1) gene during lymphocyte development. J. Biol. Chem. 279, 40906–40917 (2004).
Hoyler, T. et al. The transcription factor GATA-3 controls cell fate and maintenance of type 2 innate lymphoid cells. Immunity 37, 634–648 (2012).
Price, A.E. et al. Systemically dispersed innate IL-13-expressing cells in type 2 immunity. Proc. Natl. Acad. Sci. USA 107, 11489–11494 (2010).
Neill, D.R. et al. Nuocytes represent a new innate effector leukocyte that mediates type-2 immunity. Nature 464, 1367–1370 (2010).
Fallon, P.G. et al. Identification of an interleukin (IL)-25-dependent cell population that provides IL-4, IL-5, and IL-13 at the onset of helminth expulsion. J. Exp. Med. 203, 1105–1116 (2006).
Voehringer, D., Reese, T.A., Huang, X., Shinkai, K. & Locksley, R.M. Type 2 immunity is controlled by IL-4/IL-13 expression in hematopoietic non-eosinophil cells of the innate immune system. J. Exp. Med. 203, 1435–1446 (2006).
Shroyer, N.F., Wallis, D., Venken, K.J., Bellen, H.J. & Zoghbi, H.Y. Gfi1 functions downstream of Math1 to control intestinal secretory cell subtype allocation and differentiation. Genes Dev. 19, 2412–2417 (2005).
Halim, T.Y., Krauss, R.H., Sun, A.C. & Takei, F. Lung natural helper cells are a critical source of Th2 cell-type cytokines in protease allergen-induced airway inflammation. Immunity 36, 451–463 (2012).
Yasuda, K. et al. Contribution of IL-33-activated type II innate lymphoid cells to pulmonary eosinophilia in intestinal nematode-infected mice. Proc. Natl. Acad. Sci. USA 109, 3451–3456 (2012).
Kim, B.S. et al. TSLP elicits IL-33-independent innate lymphoid cell responses to promote skin inflammation. Sci. Transl. Med. 5, 170ra116 (2013).
Moro, K. et al. Innate production of TH2 cytokines by adipose tissue-associated c-Kit+Sca-1+ lymphoid cells. Nature 463, 540–544 (2010).
Smith, D.E. The biological paths of IL-1 family members IL-18 and IL-33. J. Leukoc. Biol. 89, 383–392 (2011).
Shinnakasu, R. et al. Gfi1-mediated stabilization of GATA3 protein is required for Th2 cell differentiation. J. Biol. Chem. 283, 28216–28225 (2008).
Zhu, J. et al. Growth factor independent-1 induced by IL-4 regulates Th2 cell proliferation. Immunity 16, 733–744 (2002).
Duan, Z. & Horwitz, M. Targets of the transcriptional repressor oncoprotein Gfi-1. Proc. Natl. Acad. Sci. USA 100, 5932–5937 (2003).
Malhotra, N. et al. A network of high-mobility group box transcription factors programs innate interleukin-17 production. Immunity 38, 681–693 (2013).
Duan, Z., Zarebski, A., Montoya-Durango, D., Grimes, H.L. & Horwitz, M. Gfi1 coordinates epigenetic repression of p21Cip/WAF1 by recruitment of histone lysine methyltransferase G9a and histone deacetylase 1. Mol. Cell Biol. 25, 10338–10351 (2005).
Lehnertz, B. et al. Activating and inhibitory functions for the histone lysine methyltransferase G9a in T helper cell differentiation and function. J. Exp. Med. 207, 915–922 (2010).
Allan, R.S. et al. An epigenetic silencing pathway controlling T helper 2 cell lineage commitment. Nature 487, 249–253 (2012).
Doe, C. et al. Expression of the T helper 17-associated cytokines IL-17A and IL-17F in asthma and COPD. Chest 138, 1140–1147 (2010).
Wang, Y.H. et al. A novel subset of CD4+ TH2 memory/effector cells that produce inflammatory IL-17 cytokine and promote the exacerbation of chronic allergic asthma. J. Exp. Med. 207, 2479–2491 (2010).
Cosmi, L. et al. Identification of a novel subset of human circulating memory CD4+ T cells that produce both IL-17A and IL-4. J. Allergy Clin. Immunol. 125, 222–230, e221–224 (2010).
Walker, J.A., Barlow, J.L. & McKenzie, A.N. Innate lymphoid cells–how did we miss them? Nat. Rev. Immunol. 13, 75–87 (2013).
Wilhelm, C. et al. An IL-9 fate reporter demonstrates the induction of an innate IL-9 response in lung inflammation. Nat. Immunol. 12, 1071–1077 (2011).
Glasmacher, E. et al. A genomic regulatory element that directs assembly and function of immune-specific AP-1-IRF complexes. Science 338, 975–980 (2012).
Acknowledgements
We thank members of the Singh laboratory, as well as H. Xi, M. van Lookeren Campagne, D. Holmes, S. Lutz and W. Ouyang, for discussions; and the Mouse Genetics Department, FACS, microarray, DNA sequencing, microscopy, and oligonucleotide synthesis facilities at Genentech for assistance and reagents.
Author information
Authors and Affiliations
Contributions
C.J.S. and H.S. designed and interpreted the experiments and wrote the manuscript; C.J.S., D.Y. and M.X. used the N. brasiliensis infection model; C.J.S., J.L., and J.D. did the experiments with administration of IL-25 and IL-33; C.J.S, M.Z. and W.P.L. used the papain lung-inflammation model; J.E.-A. and L.D. assisted with the animal pathology; V.R.-C. and R.P. assisted with the in vitro ILC2 cell cultures; R.S. and Z.M. did the microarray analysis; and A.A. and A.A.K. did the computational and statistical analysis of the microarray and ChIP-seq data, respectively.
Corresponding authors
Ethics declarations
Competing interests
C.J.S., J.L., D.Y., A.A., V.R.-C., M.Z., R.S., J.E.-A., L.D., W.P.L., Z.M., R.P., M.X. and J.D. are employees of Genentech.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 and Supplementary Tables 1–2 (PDF 1578 kb)
Rights and permissions
About this article
Cite this article
Spooner, C., Lesch, J., Yan, D. et al. Specification of type 2 innate lymphocytes by the transcriptional determinant Gfi1. Nat Immunol 14, 1229–1236 (2013). https://doi.org/10.1038/ni.2743
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.2743
This article is cited by
-
Update: Innate Lymphoid Cells in Inflammatory Bowel Disease
Digestive Diseases and Sciences (2022)
-
Sprouty2 limits intestinal tuft and goblet cell numbers through GSK3β-mediated restriction of epithelial IL-33
Nature Communications (2021)
-
TGF-β induces ST2 and programs ILC2 development
Nature Communications (2020)
-
Innate lymphoid cells control signaling circuits to regulate tissue-specific immunity
Cell Research (2020)
-
Plasticity of innate lymphoid cell subsets
Nature Reviews Immunology (2020)