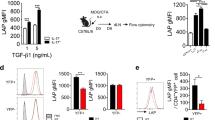
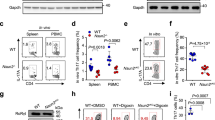
Abstract
CD4+ interleukin 17 (IL-17)-producing helper T cells (TH17 cells) are instrumental in the immune response to pathogens. However, an overactive TH17 response results in tissue inflammation and autoimmunity, and therefore it is important to identify the molecular mechanisms that control the development of TH17 cells. IL-2 suppresses such development, but how IL-2 production is actively suppressed during TH7 differentiation is not understood. Here we report that under TH17-polarizing conditions, the transcription factors STAT3 and AhR upregulated the expression of Aiolos, a member of the Ikaros family of transcription factors. Using Aiolos-deficient mice, we demonstrated that Aiolos silenced the Il2 locus, promoting TH17 differentiation in vitro and in vivo. Thus, we have identified a module in the transcriptional program of TH17 cells that actively limits IL-2 production and promotes their differentiation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Change history
21 September 2012
In the version of this article initially published, some data in the bottom right plot in Figure 3d were partially obscured. The error has been corrected in the HTML and PDF versions of the article.
References
Korn, T., Bettelli, E., Oukka, M. & Kuchroo, V.K. IL-17 and Th17 cells. Annu. Rev. Immunol. 27, 485–517 (2009).
Bettelli, E. et al. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature 441, 235–238 (2006).
Korn, T. et al. IL-21 initiates an alternative pathway to induce proinflammatory TH17 cells. Nature 448, 484–487 (2007).
Yang, X.O. et al. STAT3 regulates cytokine-mediated generation of inflammatory helper T cells. J. Biol. Chem. 282, 9358–9363 (2007).
McGeachy, M.J. et al. The interleukin 23 receptor is essential for the terminal differentiation of interleukin 17-producing effector T helper cells in vivo. Nat. Immunol. 10, 314–324 (2009).
Ivanov, I.I. et al. The orphan nuclear receptor RORγt directs the differentiation program of proinflammatory IL-17+ T helper cells. Cell 126, 1121–1133 (2006).
Yang, X.O. et al. T helper 17 lineage differentiation is programmed by orphan nuclear receptors RORα and RORγ. Immunity 28, 29–39 (2008).
Kimura, A., Naka, T., Nohara, K., Fujii-Kuriyama, Y. & Kishimoto, T. Aryl hydrocarbon receptor regulates Stat1 activation and participates in the development of Th17 cells. Proc. Natl. Acad. Sci. USA 105, 9721–9726 (2008).
Quintana, F.J. et al. Control of Treg and TH17 cell differentiation by the aryl hydrocarbon receptor. Nature 453, 65–71 (2008).
Veldhoen, M. et al. The aryl hydrocarbon receptor links TH17-cell-mediated autoimmunity to environmental toxins. Nature 453, 106–109 (2008).
Chen, Y. et al. Foxp3+ regulatory T cells promote T helper 17 cell development in vivo through regulation of interleukin-2. Immunity 34, 409–421 (2011).
Laurence, A. et al. Interleukin-2 signaling via STAT5 constrains T helper 17 cell generation. Immunity 26, 371–381 (2007).
Liao, W., Lin, J.X., Wang, L., Li, P. & Leonard, W.J. Modulation of cytokine receptors by IL-2 broadly regulates differentiation into helper T cell lineages. Nat. Immunol. 12, 551–559 (2011).
Pandiyan, P. et al. CD4+CD25+Foxp3+ regulatory T cells promote Th17 cells in vitro and enhance host resistance in mouse Candida albicans Th17 cell infection model. Immunity 34, 422–434 (2011).
Yang, X.P. et al. Opposing regulation of the locus encoding IL-17 through direct, reciprocal actions of STAT3 and STAT5. Nat. Immunol. 12, 247–254 (2011).
Georgopoulos, K. Haematopoietic cell-fate decisions, chromatin regulation and ikaros. Nat. Rev. Immunol. 2, 162–174 (2002).
Gandhi, R. et al. Activation of the aryl hydrocarbon receptor induces human type 1 regulatory T cell-like and Foxp3+ regulatory T cells. Nat. Immunol. 11, 846–853 (2010).
Pan, F. et al. Eos mediates Foxp3-dependent gene silencing in CD4+ regulatory T cells. Science 325, 1142–1146 (2009).
Thornton, A.M. et al. Expression of Helios, an Ikaros transcription factor family member, differentiates thymic-derived from peripherally induced Foxp3+ T regulatory cells. J. Immunol. 184, 3433–3441 (2010).
Umetsu, S.E. & Winandy, S. Ikaros is a regulator of Il10 expression in CD4+ T cells. J. immunol. 183, 5518–5525 (2009).
Wang, J.H. et al. Aiolos regulates B cell activation and maturation to effector state. Immunity 9, 543–553 (1998).
Bandyopadhyay, S. et al. Interleukin 2 gene transcription is regulated by Ikaros-induced changes in histone acetylation in anergic T cells. Blood 109, 2878–2886 (2007).
Depper, J.M. et al. Interleukin 2 (IL-2) augments transcription of the IL-2 receptor gene. Proc. Natl. Acad. Sci. USA 82, 4230–4234 (1985).
Kimura, A., Naka, T. & Kishimoto, T. IL-6-dependent and -independent pathways in the development of interleukin 17-producing T helper cells. Proc. Natl. Acad. Sci. USA 104, 12099–12104 (2007).
Villarino, A.V., Gallo, E. & Abbas, A.K. STAT1-activating cytokines limit Th17 responses through both T-bet-dependent and -independent mechanisms. J. immunol. 185, 6461–6471 (2010).
Thomas, R.M. et al. Ikaros enforces the costimulatory requirement for IL2 gene expression and is required for anergy induction in CD4+ T lymphocytes. J. Immunol. 179, 7305–7315 (2007).
Koipally, J., Renold, A., Kim, J. & Georgopoulos, K. Repression by Ikaros and Aiolos is mediated through histone deacetylase complexes. EMBO J. 18, 3090–3100 (1999).
Durant, L. et al. Diverse targets of the transcription factor STAT3 contribute to T cell pathogenicity and homeostasis. Immunity 32, 605–615 (2010).
Wei, L., Laurence, A., Elias, K.M. & O'Shea, J.J. IL-21 is produced by Th17 cells and drives IL-17 production in a STAT3-dependent manner. J. Biol. Chem. 282, 34605–34610 (2007).
Wei, L. et al. Discrete roles of STAT4 and STAT6 transcription factors in tuning epigenetic modifications and transcription during T helper cell differentiation. Immunity 32, 840–851 (2010).
Quintana, F.J. et al. Antigen microarrays identify unique serum autoantibody signatures in clinical and pathologic subtypes of multiple sclerosis. Proc. Natl. Acad. Sci. USA 105, 18889–18894 (2008).
Dooms, H., Wolslegel, K., Lin, P. & Abbas, A.K. Interleukin-2 enhances CD4+ T cell memory by promoting the generation of IL-7Rα-expressing cells. J. Exp. Med. 204, 547–557 (2007).
Kühn, R., Lohler, J., Rennick, D., Rajewsky, K. & Muller, W. Interleukin-10-deficient mice develop chronic enterocolitis. Cell 75, 263–274 (1993).
Ivanov, I.I. et al. Induction of intestinal Th17 cells by segmented filamentous bacteria. Cell 139, 485–498 (2009).
O'Connor, W. Jr. et al. A protective function for interleukin 17A in T cell-mediated intestinal inflammation. Nat. Immunol. 10, 603–609 (2009).
Kameyama, K. et al. IL-2 is positively involved in the development of colitogenic CD4+ IL-7Rα high memory T cells in chronic colitis. Eur. J. Immunol. 40, 2423–2436 (2010).
Cox, J.H. et al. IL-27 promotes T cell-dependent colitis through multiple mechanisms. J. Exp. Med. 208, 115–123 (2011).
Villarino, A.V. et al. Helper T cell IL-2 production is limited by negative feedback and STAT-dependent cytokine signals. J. Exp. Med. 204, 65–71 (2007).
Oh, H.-M. et al. STAT3 promotes T cell survival and inhibits IL-2 production through up-regulation of class O forkhead transcription factors. J. Biol. Chem. 286, 30888–30897 (2011).
Yamanouchi, J. et al. Interleukin-2 gene variation impairs regulatory T cell function and causes autoimmunity. Nat. Genet. 39, 329–337 (2007).
Liu, S.M. et al. Differential IL-21 signaling in APCs leads to disparate Th17 differentiation in diabetes-susceptible NOD and diabetes-resistant NOD.Idd3 mice. J. Clin. Invest. 121, 4303–4310 (2011).
Fontenot, J.D., Rasmussen, J.P., Gavin, M.A. & Rudensky, A.Y. A function for interleukin 2 in Foxp3-expressing regulatory T cells. Nat. Immunol. 6, 1142–1151 (2005).
Wilson, C.B., Rowell, E. & Sekimata, M. Epigenetic control of T-helper-cell differentiation. Nat. Rev. Immunol. 9, 91–105 (2009).
Jeon, M.S. & Esser, C. The murine IL-2 promoter contains distal regulatory elements responsive to the Ah receptor, a member of the evolutionarily conserved bHLH-PAS transcription factor family. J. Immunol. 165, 6975–6983 (2000).
Apetoh, L. et al. The aryl hydrocarbon receptor interacts with c-Maf to promote the differentiation of type 1 regulatory T cells induced by IL-27. Nat. Immunol. 11, 854–861 (2010).
Acknowledgements
We thank D. Kozoriz for cell sorting, and B. Waksman and I.R. Cohen for discussions. Supported by the US National Institutes of Health (P01AI073748, P01NS038037, P01AI056299 and R01NS030843 to V.K.K.; and AI075285 and AI093903 to F.J.Q.), the National Multiple Sclerosis Society (RG4111A1 to F.J.Q.), the Harvard Medical School Office for Diversity and Community Partnership (F.J.Q.) and the National Multiple Sclerosis Society (FG1850-A-1 to H.J.).
Author information
Authors and Affiliations
Contributions
H.J., F.J.Q., M.N. and M.R. did in vitro and in vivo experiments; M.N., A.Y., E.J.B., D.K., C.Z. and S.X. did in vitro experiments; K.G. and J.S. provided reagents; F.J.Q. wrote the manuscript; and V.K.K. and F.J.Q. supervised the study and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 (PDF 624 kb)
Rights and permissions
About this article
Cite this article
Quintana, F., Jin, H., Burns, E. et al. Aiolos promotes TH17 differentiation by directly silencing Il2 expression. Nat Immunol 13, 770–777 (2012). https://doi.org/10.1038/ni.2363
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.2363
This article is cited by
-
The transcription factor Aiolos restrains the activation of intestinal intraepithelial lymphocytes
Nature Immunology (2024)
-
Aiolos represses CD4+ T cell cytotoxic programming via reciprocal regulation of TFH transcription factors and IL-2 sensitivity
Nature Communications (2023)
-
Regulation of activated T cell survival in rheumatic autoimmune diseases
Nature Reviews Rheumatology (2022)
-
Cancer therapies based on targeted protein degradation — lessons learned with lenalidomide
Nature Reviews Clinical Oncology (2021)
-
Environmental pollutants and the immune response
Nature Immunology (2020)