Abstract
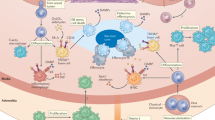
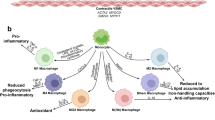
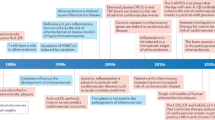
Cardiovascular disease, a leading cause of mortality worldwide, is caused mainly by atherosclerosis, a chronic inflammatory disease of blood vessels. Lesions of atherosclerosis contain macrophages, T cells and other cells of the immune response, together with cholesterol that infiltrates from the blood. Targeted deletion of genes encoding costimulatory factors and proinflammatory cytokines results in less disease in mouse models, whereas interference with regulatory immunity accelerates it. Innate as well as adaptive immune responses have been identified in atherosclerosis, with components of cholesterol-carrying low-density lipoprotein triggering inflammation, T cell activation and antibody production during the course of disease. Studies are now under way to develop new therapies based on these concepts of the involvement of the immune system in atherosclerosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Katie Vicari

Katie Vicari

Katie Vicari

Katie Vicari

Katie Vicari
Similar content being viewed by others
References
Dahlof, B. Cardiovascular disease risk factors: epidemiology and risk assessment. Am. J. Cardiol. 105, 3A–9A (2010).
Lloyd-Jones, D.M. Cardiovascular risk prediction: basic concepts, current status, and future directions. Circulation 121, 1768–1777 (2010).
Hansson, G.K. Inflammation, atherosclerosis, and coronary artery disease. N. Engl. J. Med. 352, 1685–1695 (2005).
Hansson, G.K., Robertson, A.K.L. & Söderberg-Nauclér, C. Inflammation and atherosclerosis. Annu. Rev. Pathol. 1, 297–329 (2006).
Tabas, I., Williams, K.J. & Boren, J. Subendothelial lipoprotein retention as the initiating process in atherosclerosis: update and therapeutic implications. Circulation 116, 1832–1844 (2007).
Skålen, K. et al. Subendothelial retention of atherogenic lipoproteins in early atherosclerosis. Nature 417, 750–754 (2002).
Bochkov, V.N. et al. Oxidized phospholipids stimulate tissue factor expression in human endothelial cells via activation of ERK/EGR-1 and Ca++/NFAT. Blood 99, 199–206 (2002).
Gharavi, N.M. et al. Role of the Jak/STAT pathway in the regulation of interleukin-8 transcription by oxidized phospholipids in vitro and in atherosclerosis in vivo. J. Biol. Chem. 282, 31460–31468 (2007).
Gargalovic, P.S. et al. The unfolded protein response is an important regulator of inflammatory genes in endothelial cells. Arterioscler. Thromb. Vasc. Biol. 26, 2490–2496 (2006).
Binder, C.J. et al. Pneumococcal vaccination decreases atherosclerotic lesion formation: molecular mimicry between Streptococcus pneumoniae and oxidized LDL. Nat. Med. 9, 736–743 (2003).
Caligiuri, G. et al. Phosphorylcholine-targeting immunization reduces atherosclerosis. J. Am. Coll. Cardiol. 50, 540–546 (2007).
Schiopu, A. et al. Recombinant human antibodies against aldehyde-modified apolipoprotein B-100 peptide sequences inhibit atherosclerosis. Circulation 110, 2047–2052 (2004).
Zernecke, A., Shagdarsuren, E. & Weber, C. Chemokines in atherosclerosis: an update. Arterioscler. Thromb. Vasc. Biol. 28, 1897–1908 (2008).
Smith, J.D. et al. Decreased atherosclerosis in mice deficient in both macrophage colony-stimulating factor (op) and apolipoprotein E. Proc. Natl. Acad. Sci. USA 92, 8264–8268 (1995).
Niessner, A. et al. Pathogen-sensing plasmacytoid dendritic cells stimulate cytotoxic T-cell function in the atherosclerotic plaque through interferon-alpha. Circulation 114, 2482–2489 (2006).
Niessner, A. & Weyand, C.M. Dendritic cells in atherosclerotic disease. Clin. Immunol. 134, 25–32 (2010).
Tedgui, A. & Mallat, Z. Cytokines in atherosclerosis: pathogenic and regulatory pathways. Physiol. Rev. 86, 515–581 (2006).
Kovanen, P.T. Mast cells: multipotent local effector cells in atherothrombosis. Immunol. Rev. 217, 105–122 (2007).
Grabner, R. et al. Lymphotoxin beta receptor signaling promotes tertiary lymphoid organogenesis in the aorta adventitia of aged ApoE-/- mice. J. Exp. Med. 206, 233–248 (2009).
Lundberg, A.M. & Hansson, G.K. Innate immune signals in atherosclerosis. Clin. Immunol. 134, 5–24 (2010).
Greaves, D.R. & Gordon, S. The macrophage scavenger receptor at 30 years of age: current knowledge and future challenges. J. Lipid Res. 50 Suppl, S282–S286 (2009).
Tall, A.R. Cholesterol efflux pathways and other potential mechanisms involved in the athero-protective effect of high density lipoproteins. J. Intern. Med. 263, 256–273 (2008).
Yvan-Charvet, L. et al. ABCA1 and ABCG1 protect against oxidative stress-induced macrophage apoptosis during efferocytosis. Circ. Res. 106, 1861–1869 (2010).
Edfeldt, K., Swedenborg, J., Hansson, G.K. & Yan, Z.Q. Expression of toll-like receptors in human atherosclerotic lesions: a possible pathway for plaque activation. Circulation 105, 1158–1161 (2002).
Curtiss, L.K. & Tobias, P.S. Emerging role of toll-like receptors in atherosclerosis. J. Lipid Res. 50, 5340–5345 (2009).
Michelsen, K.S. et al. Lack of Toll-like receptor 4 or myeloid differentiation factor 88 reduces atherosclerosis and alters plaque phenotype in mice deficient in apolipoprotein E. Proc. Natl. Acad. Sci. USA 101, 10679–10684 (2004).
Bjorkbacka, H. et al. Reduced atherosclerosis in MyD88-null mice links elevated serum cholesterol levels to activation of innate immunity signaling pathways. Nat. Med. 10, 416–421 (2004).
Kirii, H. et al. Lack of interleukin-1b decreases the severity of atherosclerosis in ApoE-deficient mice. Arterioscler. Thromb. Vasc. Biol. 23, 656–660 (2003).
Elhage, R. et al. Reduced atherosclerosis in interleukin-18 deficient apolipoprotein E-knockout mice. Cardiovasc. Res. 59, 234–240 (2003).
Seimon, T.A. et al. Atherogenic lipids and lipoproteins trigger CD36–TLR2-dependent apoptosis in macrophages undergoing endoplasmic reticulum stress. Cell Metab. 12, 467–482 (2010).
West, X.Z. et al. Oxidative stress induces angiogenesis by activating TLR2 with novel endogenous ligands. Nature 467, 972–976 (2010).
Miller, Y.I. et al. Minimally modified LDL binds to CD14, induces macrophage spreading via TLR4/MD-2, and inhibits phagocytosis of apoptotic cells. J. Biol. Chem. 278, 1561–1568 (2003).
Mullick, A.E., Tobias, P.S. & Curtiss, L.K. Modulation of atherosclerosis in mice by Toll-like receptor 2. J. Clin. Invest. 115, 3149–3156 (2005).
Schroder, K. & Tschopp, J. The inflammasomes. Cell 140, 821–832 (2010).
Duewell, P. et al. NLRP3 inflammasomes are required for atherogenesis and activated by cholesterol crystals. Nature 464, 1357–1361 (2010).
Rajamaki, K. et al. Cholesterol crystals activate the NLRP3 inflammasome in human macrophages: a novel link between cholesterol metabolism and inflammation. PLoS ONE 5, e11765 (2010).
Edfeldt, K. et al. Involvement of the antimicrobial peptide LL-37 in human atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 26, 1551–1557 (2006).
Samuelsson, B., Morgenstern, R. & Jakobsson, P.J. Membrane prostaglandin E synthase-1: a novel therapeutic target. Pharmacol. Rev. 59, 207–224 (2007).
Hui, Y. et al. Targeted deletions of cyclooxygenase-2 and atherogenesis in mice. Circulation 121, 2654–2660 (2010).
Bäck, M. et al. Leukotriene B4 signaling through NF-kB-dependent BLT1 receptors on vascular smooth muscle cells in atherosclerosis and intimal hyperplasia. Proc. Natl. Acad. Sci. USA 102, 17501–17506 (2005).
Heller, E.A. et al. Inhibition of atherogenesis in BLT1-deficient mice reveals a role for LTB4 and BLT1 in smooth muscle cell recruitment. Circulation 112, 578–586 (2005).
Mehrabian, M. et al. Identification of 5-lipoxygenase as a major gene contributing to atherosclerosis susceptibility in mice. Circ. Res. 91, 120–126 (2002).
Spanbroek, R. et al. Expanding expression of the 5-lipoxygenase pathway within the arterial wall during human atherogenesis. Proc. Natl. Acad. Sci. USA 100, 1238–1243 (2003).
Qiu, H. et al. Expression of 5-lipoxygenase and leukotriene A4 hydrolase in human atherosclerotic lesions correlates with symptoms of plaque instability. Proc. Natl. Acad. Sci. USA 103, 8161–8166 (2006).
Helgadottir, A. et al. The gene encoding 5-lipoxygenase activating protein confers risk of myocardial infarction and stroke. Nat. Genet. 36, 233–239 (2004).
Andersson, J., Libby, P. & Hansson, G.K. Adaptive immunity and atherosclerosis. Clin. Immunol. 134, 33–46 (2010).
Reardon, C.A. et al. Effect of immune deficiency on lipoproteins and atherosclerosis in male apolipoprotein E-deficient mice. Arterioscler. Thromb. Vasc. Biol. 21, 1011–1016 (2001).
Zhou, X., Nicoletti, A., Elhage, R. & Hansson, G.K. Transfer of CD4+ T cells aggravates atherosclerosis in immunodeficient apolipoprotein E knockout mice. Circulation 102, 2919–2922 (2000).
Caligiuri, G., Nicoletti, A., Poirier, B. & Hansson, G.K. Protective immunity against atherosclerosis carried by B cells of hypercholesterolemic mice. J. Clin. Invest. 109, 745–753 (2002).
Major, A.S., Fazio, S. & Linton, M.F. B-lymphocyte deficiency increases atherosclerosis in LDL receptor-null mice. Arterioscler. Thromb. Vasc. Biol. 22, 1892–1898 (2002).
Binder, C.J. et al. IL-5 links adaptive and natural immunity specific for epitopes of oxidized LDL and protects from atherosclerosis. J. Clin. Invest. 114, 427–437 (2004).
Ait-Oufella, H. et al. B cell depletion reduces the development of atherosclerosis in mice. J. Exp. Med. 207, 1579–1587 (2010).
Kyaw, T. et al. Conventional B2 B cell depletion ameliorates whereas its adoptive transfer aggravates atherosclerosis. J. Immunol. 185, 4410–4419 (2010).
Palinski, W., Miller, E. & Witztum, J.L. Immunization of low density lipoprotein (LDL) receptor-deficient rabbits with homologous malondialdehyde-modified LDL reduces atherogenesis. Proc. Natl. Acad. Sci. USA 92, 821–825 (1995).
Ameli, S. et al. Effect of immunization with homologous LDL and oxidized LDL on early atherosclerosis in hypercholesterolemic rabbits. Arterioscler. Thromb. Vasc. Biol. 16, 1074–1079 (1996).
Nilsson, J., Hansson, G.K. & Shah, P.K. Immunomodulation of atherosclerosis: implications for vaccine development. Arterioscler. Thromb. Vasc. Biol. 25, 18–28 (2005).
Hulthe, J. et al. Antibody titers against oxidized LDL are not elevated in patients with familial hypercholesterolemia. Arterioscler. Thromb. Vasc. Biol. 18, 1203–1211 (1998).
Tornvall, P., Waeg, G., Nilsson, J., Hamsten, A. & Regnstrom, J. Autoantibodies against modified low-density lipoproteins in coronary artery disease. Atherosclerosis 167, 347–353 (2003).
Fredrikson, G.N. et al. Association between IgM against an aldehyde-modified peptide in apolipoprotein B-100 and progression of carotid disease. Stroke 38, 1495–1500 (2007).
Sjogren, P. et al. High plasma concentrations of autoantibodies against native peptide 210 of apoB-100 are related to less coronary atherosclerosis and lower risk of myocardial infarction. Eur. Heart J. 29, 2218–2226 (2008).
Tsimikas, S. et al. Relationship of IgG and IgM autoantibodies to oxidized low density lipoprotein with coronary artery disease and cardiovascular events. J. Lipid Res. 48, 425–433 (2007).
Paulsson, G., Zhou, X., Tornquist, E. & Hansson, G.K. Oligoclonal T cell expansions in atherosclerotic lesions of apolipoprotein E-deficient mice. Arterioscler. Thromb. Vasc. Biol. 20, 10–17 (2000).
Liuzzo, G. et al. Monoclonal T-cell proliferation and plaque instability in acute coronary syndromes. Circulation 101, 2883–2888 (2000).
Mach, F., Schönbeck, U., Sukhova, G.K., Atkinson, E. & Libby, P. Reduction of atherosclerosis in mice by inhibition of CD40 signalling. Nature 394, 200–203 (1998).
Olofsson, P.S. et al. CD137 is expressed in human atherosclerosis and promotes development of plaque inflammation in hypercholesterolemic mice. Circulation 117, 1292–1301 (2008).
Ludewig, B. et al. Linking immune-mediated arterial inflammation and cholesterol-induced atherosclerosis in a transgenic mouse model. Proc. Natl. Acad. Sci. USA 97, 12752–12757 (2000).
Gotsman, I. et al. Proatherogenic immune responses are regulated by the PD-1/PD-L pathway in mice. J. Clin. Invest. 117, 2974–2982 (2007).
Gupta, S. et al. IFN-g potentiates atherosclerosis in ApoE knock-out mice. J. Clin. Invest. 99, 2752–2761 (1997).
Whitman, S.C., Ravisankar, P., Elam, H. & Daugherty, A. Exogenous interferon-g enhances atherosclerosis in apolipoprotein E-/- mice. Am. J. Pathol. 157, 1819–1824 (2000).
Whitman, S.C., Ravisankar, P. & Daugherty, A. IFN-g deficiency exerts gender-specific effects on atherogenesis in apolipoprotein E-/- mice. J. Interferon Cytokine Res. 22, 661–670 (2002).
Buono, C. et al. Influence of interferon-g on the extent and phenotype of diet-induced atherosclerosis in the LDLR-deficient mouse. Arterioscler. Thromb. Vasc. Biol. 23, 454–460 (2003).
Lee, T.S., Yen, H.C., Pan, C.C. & Chau, L.Y. The role of interleukin 12 in the development of atherosclerosis in ApoE-deficient mice. Arterioscler. Thromb. Vasc. Biol. 19, 734–742 (1999).
Davenport, P. & Tipping, P.G. The role of interleukin-4 and interleukin-12 in the progression of atherosclerosis in apolipoprotein E-deficient mice. Am. J. Pathol. 163, 1117–1125 (2003).
Hauer, A.D. et al. Blockade of interleukin-12 function by protein vaccination attenuates atherosclerosis. Circulation 112, 1054–1062 (2005).
Whitman, S.C., Ravisankar, P. & Daugherty, A. Interleukin-18 enhances atherosclerosis in apolipoprotein E−/− mice through release of interferon-gamma. Circ. Res. 90, E34–E38 (2002).
Buono, C. et al. T-bet deficiency reduces atherosclerosis and alters plaque antigen-specific immune responses. Proc. Natl. Acad. Sci. USA 102, 1596–1601 (2005).
Frostegard, J. et al. Cytokine expression in advanced human atherosclerotic plaques: dominance of pro-inflammatory (Th1) and macrophage-stimulating cytokines. Atherosclerosis 145, 33–43 (1999).
King, V.L., Szilvassy, S.J. & Daugherty, A. Interleukin-4 deficiency decreases atherosclerotic lesion formation in a site-specific manner in female LDL receptor-/- mice. Arterioscler. Thromb. Vasc. Biol. 22, 456–461 (2002).
Huber, S.A., Sakkinen, P., David, C., Newell, M.K. & Tracy, R.P. T helper-cell phenotype regulates atherosclerosis in mice under conditions of mild hypercholesterolemia. Circulation 103, 2610–2616 (2001).
King, V.L., Cassis, L.A. & Daugherty, A. Interleukin-4 does not influence development of hypercholesterolemia or angiotensin II-induced atherosclerotic lesions in mice. Am. J. Pathol. 171, 2040–2047 (2007).
Miller, A.M. et al. IL-33 reduces the development of atherosclerosis. J. Exp. Med. 205, 339–346 (2008).
de Boer, O.J. et al. Differential expression of interleukin-17 family cytokines in intact and complicated human atherosclerotic plaques. J. Pathol. 220, 499–508 (2010).
Eid, R.E. et al. Interleukin-17 and interferon-g are produced concomitantly by human coronary artery-infiltrating T cells and act synergistically on vascular smooth muscle cells. Circulation 119, 1424–1432 (2009).
Erbel, C. et al. Inhibition of IL-17A attenuates atherosclerotic lesion development in apoE-deficient mice. J. Immunol. 183, 8167–8175 (2009).
van Es, T. et al. Attenuated atherosclerosis upon IL-17R signaling disruption in LDLr deficient mice. Biochem. Biophys. Res. Commun. 388, 261–265 (2009).
Smith, E. et al. Blockade of interleukin-17A results in reduced atherosclerosis in apolipoprotein E-deficient mice. Circulation 121, 1746–1755 (2010).
Taleb, S. et al. Loss of SOCS3 expression in T cells reveals a regulatory role for interleukin-17 in atherosclerosis. J. Exp. Med. 206, 2067–2077 (2009).
Veillard, N.R., Steffens, S., Burger, F., Pelli, G. & Mach, F. Differential expression patterns of proinflammatory and antiinflammatory mediators during atherogenesis in mice. Arterioscler. Thromb. Vasc. Biol. 24, 2339–2344 (2004).
de Boer, O.J., van der Meer, J.J., Teeling, P., van der Loos, C.M. & van der Wal, A.C. Low numbers of FOXP3 positive regulatory T cells are present in all developmental stages of human atherosclerotic lesions. PLoS ONE 2, e779 (2007).
Ait-Oufella, H. et al. Natural regulatory T cells control the development of atherosclerosis in mice. Nat. Med. 12, 178–180 (2006).
Mor, A. et al. Role of naturally occurring CD4+CD25+ regulatory T cells in experimental atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 27, 893–900 (2007).
Klingenberg, R. et al. Intranasal immunization with an apolipoprotein B-100 fusion protein induces antigen-specific regulatory T cells and reduces atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 30, 946–952 (2010).
Sasaki, N. et al. Oral anti-CD3 antibody treatment induces regulatory T cells and inhibits the development of atherosclerosis in mice. Circulation 120, 1996–2005 (2009).
Vliegen, I., Herngreen, S.B., Grauls, G.E., Bruggeman, C.A. & Stassen, F.R. Mouse cytomegalovirus antigenic immune stimulation is sufficient to aggravate atherosclerosis in hypercholesterolemic mice. Atherosclerosis 181, 39–44 (2005).
Hansson, G.K. & Libby, P. The immune response in atherosclerosis: a double-edged sword. Nat. Rev. Immunol. 6, 508–519 (2006).
Wick, G., Knoflach, M. & Xu, Q. Autoimmune and inflammatory mechanisms in atherosclerosis. Annu. Rev. Immunol. 22, 361–403 (2004).
Afek, A. et al. Immunization of low-density lipoprotein receptor deficient (LDL-RD) mice with heat shock protein 65 (HSP-65) promotes early atherosclerosis. J. Autoimmun. 14, 115–121 (2000).
Harats, D., Yacov, N., Gilburd, B., Shoenfeld, Y. & George, J. Oral tolerance with heat shock protein 65 attenuates Mycobacterium tuberculosis-induced and high-fat-diet-driven atherosclerotic lesions. J. Am. Coll. Cardiol. 40, 1333–1338 (2002).
Maron, R. et al. Mucosal administration of heat shock protein-65 decreases atherosclerosis and inflammation in aortic arch of low-density lipoprotein receptor-deficient mice. Circulation 106, 1708–1715 (2002).
Tsan, M.F. & Gao, B. Heat shock proteins and immune system. J. Leukoc. Biol. 85, 905–910 (2009).
Steinberg, D. The LDL modification hypothesis of atherogenesis: an update. J. Lipid Res. 50 Suppl, S376–S381 (2009).
Stemme, S. et al. T lymphocytes from human atherosclerotic plaques recognize oxidized low density lipoprotein. Proc. Natl. Acad. Sci. USA 92, 3893–3897 (1995).
Zhou, X., Robertson, A.K., Hjerpe, C. & Hansson, G.K. Adoptive transfer of CD4+ T cells reactive to modified low-density lipoprotein aggravates atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 26, 864–870 (2006).
Fredrikson, G.N. et al. Identification of immune responses against aldehyde-modified peptide sequences in apoB associated with cardiovascular disease. Arterioscler. Thromb. Vasc. Biol. 23, 872–878 (2003).
Nicoletti, A. et al. The macrophage scavenger receptor type A directs modified proteins to antigen presentation. Eur. J. Immunol. 29, 512–521 (1999).
Hjerpe, C., Johansson, D., Hermansson, A., Hansson, G.K. & Zhou, X. Dendritic cells pulsed with malondialdehyde modified low density lipoprotein aggravate atherosclerosis in Apoe−/− mice. Atherosclerosis 209, 436–441 (2010).
Hermansson, A. et al. Immunotherapy with tolerogenic apolipoprotein B-100 loaded dendritic cells attenuates atherosclerosis in hypercholesterolemic mice. Circulation (in the press).
Hermansson, A. et al. Inhibition of T cell response to native low-density lipoprotein reduces atherosclerosis. J. Exp. Med. 207, 1081–1093 (2010).
Huang, W. & Glass, C.K. Nuclear receptors and inflammation control: molecular mechanisms and pathophysiological relevance. Arterioscler. Thromb. Vasc. Biol. 30, 1542–1549 (2010).
Glass, C.K. & Saijo, K. Nuclear receptor transrepression pathways that regulate inflammation in macrophages and T cells. Nat. Rev. Immunol. 10, 365–376 (2010).
Liao, J.K. & Laufs, U. Pleiotropic effects of statins. Annu. Rev. Pharmacol. Toxicol. 45, 89–118 (2005).
Jury, E.C., Isenberg, D.A., Mauri, C. & Ehrenstein, M.R. Atorvastatin restores Lck expression and lipid raft-associated signaling in T cells from patients with systemic lupus erythematosus. J. Immunol. 177, 7416–7422 (2006).
Hansson, G.K. & Bjorkholm, M. Tackling two diseases with HDL. Science 328, 1641–1642 (2010).
Youssef, S. et al. The HMG-CoA reductase inhibitor, atorvastatin, promotes a Th2 bias and reverses paralysis in central nervous system autoimmune disease. Nature 420, 78–84 (2002).
Klareskog, L. & Hamsten, A. Statins in rheumatoid arthritis–two birds with one stone? Lancet 363, 2011–2012 (2004).
Wang, X. et al. Positional identification of TNFSF4, encoding OX40 ligand, as a gene that influences atherosclerosis susceptibility. Nat. Genet. 37, 365–372 (2005).
Swanberg, M. et al. MHC2TA is associated with differential MHC molecule expression and susceptibility to rheumatoid arthritis, multiple sclerosis and myocardial infarction. Nat. Genet. 37, 486–494 (2005).
Dwyer, J.H. et al. Arachidonate 5-lipoxygenase promoter genotype, dietary arachidonic acid, and atherosclerosis. N. Engl. J. Med. 350, 29–37 (2004).
Teslovich, T.M. et al. Biological, clinical and population relevance of 95 loci for blood lipids. Nature 466, 707–713 (2010).
Gabriel, S.E. Cardiovascular morbidity and mortality in rheumatoid arthritis. Am. J. Med. 121, S9–S14 (2008).
Holmqvist, M.E. et al. No increased occurrence of ischemic heart disease prior to the onset of rheumatoid arthritis: results from two Swedish population-based rheumatoid arthritis cohorts. Arthritis Rheum. 60, 2861–2869 (2009).
Dixon, W.G. et al. Reduction in the incidence of myocardial infarction in patients with rheumatoid arthritis who respond to anti-tumor necrosis factor alpha therapy: results from the British Society for Rheumatology Biologics Register. Arthritis Rheum. 56, 2905–2912 (2007).
Muller-Ehmsen, J. & Schwinger, R.H. TNF and congestive heart failure: therapeutic possibilities. Expert Opin. Ther. Targets 8, 203–209 (2004).
Zink, A. et al. European biologicals registers: methodology, selected results and perspectives. Ann. Rheum. Dis. 68, 1240–1246 (2009).
Ridker, P.M., Hennekens, C.H., Buring, J.E. & Rifai, N. C-reactive protein and other markers of inflammation in the prediction of cardiovascular disease in women. N. Engl. J. Med. 342, 836–843 (2000).
Acknowledgements
We thank J. Andersson and A.-K. Robertson for critical reading of the manuscript. Supported by the Swedish Research Council, Foundation for Strategic Research, VINNOVA, the Swedish Heart-Lung Foundation, the Leducq Foundation and the European Union (AtheroRemo project).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
TG.K.H. and A.H. have submitted patent applications in the area reviewed.
Rights and permissions
About this article
Cite this article
Hansson, G., Hermansson, A. The immune system in atherosclerosis. Nat Immunol 12, 204–212 (2011). https://doi.org/10.1038/ni.2001
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.2001