Abstract
Transcription factor Foxp3 is critical for generating regulatory T cells (Treg cells). Transforming growth factor-β (TGF-β) induces Foxp3 and suppressive Treg cells from naive T cells, whereas interleukin 6 (IL-6) inhibits the generation of inducible Treg cells. Here we show that IL-4 blocked the generation of TGF-β-induced Foxp3+ Treg cells and instead induced a population of T helper cells that produced IL-9 and IL-10. The IL-9+IL-10+ T cells demonstrated no regulatory properties despite producing abundant IL-10. Adoptive transfer of IL-9+IL-10+ T cells into recombination-activating gene 1–deficient mice induced colitis and peripheral neuritis, the severity of which was aggravated if the IL-9+IL-10+ T cells were transferred with CD45RBhi CD4+ effector T cells. Thus IL-9+IL-10+ T cells lack suppressive function and constitute a distinct population of helper-effector T cells that promote tissue inflammation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
09 April 2009
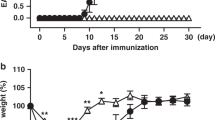
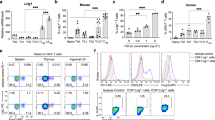
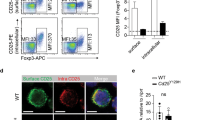
In the version of this article initially published, graph axes are mislabeled in Figures 1e, 2d and 2e, and a gene symbol is misidentified in the legend to Figure 2. The correct axis labels should be as follows: Figure 1e, middle right and far right vertical axes should end “(relative x 103)”; Figure 2d,e, left vertical axes should read “Il9 mRNA (relative x 102)”; Figure 2e, right vertical axis should read “Il10 mRNA (relative)”; and Figure 2e, horizontal axes should read “WT” (in place of “Foxp3-GFP”) and “GATA3-KO” (in place of “STAT6-KO.Fox3-GFP”). The legend for Figure 2d should state “relative to Hprt1 mRNA” and the legend to Figure 2e should have that phrase removed. The errors have been corrected in the HTML and PDF versions of the article.
References
Szabo, S.J. et al. A novel transcription factor, T-bet, directs Th1 lineage commitment. Cell 100, 655–669 (2000).
Zheng, W. & Flavell, R.A. The transcription factor GATA-3 is necessary and sufficient for Th2 cytokine gene expression in CD4 T cells. Cell 89, 587–596 (1997).
Ivanov, I.I. et al. The orphan nuclear receptor RORγt directs the differentiation program of proinflammatory IL-17+ T helper cells. Cell 126, 1121–1133 (2006).
Ouyang, W., Kolls, J.K. & Zheng, Y. The biological functions of T helper 17 cell effector cytokines in inflammation. Immunity 28, 454–467 (2008).
Korn, T., Oukka, M., Kuchroo, V. & Bettelli, E. Th17 cells: effector T cells with inflammatory properties. Semin. Immunol. 19, 362–371 (2007).
Battaglia, M., Gregori, S., Bacchetta, R. & Roncarolo, M.G. Tr1 cells: from discovery to their clinical application. Semin. Immunol. 18, 120–127 (2006).
Hori, S., Takahashi, T. & Sakaguchi, S. Control of autoimmunity by naturally arising regulatory CD4+ T cells. Adv. Immunol. 81, 331–371 (2003).
Hori, S., Nomura, T. & Sakaguchi, S. Control of regulatory T cell development by the transcription factor Foxp3. Science 299, 1057–1061 (2003).
Kretschmer, K. et al. Inducing and expanding regulatory T cell populations by foreign antigen. Nat. Immunol. 6, 1219–1227 (2005).
Apostolou, I. & von Boehmer, H. In vivo instruction of suppressor commitment in naive T cells. J. Exp. Med. 199, 1401–1408 (2004).
Chen, W. et al. Conversion of peripheral CD4+CD25− naive T cells to CD4+CD25+ regulatory T cells by TGF-β induction of transcription factor Foxp3. J. Exp. Med. 198, 1875–1886 (2003).
Bettelli, E. et al. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature 441, 235–238 (2006).
Wan, Y.Y. & Flavell, R.A. Regulatory T-cell functions are subverted and converted owing to attenuated Foxp3 expression. Nature 445, 766–770 (2007).
Wei, J. et al. Antagonistic nature of T helper 1/2 developmental programs in opposing peripheral induction of Foxp3+ regulatory T cells. Proc. Natl. Acad. Sci. USA 104, 18169–18174 (2007).
Mantel, P.Y. et al. GATA3-driven Th2 responses inhibit TGF-β1-induced FOXP3 expression and the formation of regulatory T cells. PLoS Biol. 5, e329 (2007).
Awasthi, A. et al. A dominant function for interleukin 27 in generating interleukin 10–producing anti-inflammatory T cells. Nat. Immunol. 8, 1380–1389 (2007).
Stumhofer, J.S. et al. Interleukins 27 and 6 induce STAT3-mediated T cell production of interleukin 10. Nat. Immunol. 8, 1363–1371 (2007).
Ishida, H. et al. Continuous administration of anti-interleukin 10 antibodies delays onset of autoimmunity in NZB/W F1 mice. J. Exp. Med. 179, 305–310 (1994).
Llorente, L. et al. Role of interleukin 10 in the B lymphocyte hyperactivity and autoantibody production of human systemic lupus erythematosus. J. Exp. Med. 181, 839–844 (1995).
Van Snick, J. et al. Cloning and characterization of a cDNA for a new mouse T cell growth factor (P40). J. Exp. Med. 169, 363–368 (1989).
Faulkner, H., Humphreys, N., Renauld, J.C., Van Snick, J. & Grencis, R. Interleukin-9 is involved in host protective immunity to intestinal nematode infection. Eur. J. Immunol. 27, 2536–2540 (1997).
Kamanaka, M. et al. Expression of interleukin-10 in intestinal lymphocytes detected by an interleukin-10 reporter knockin tiger mouse. Immunity 25, 941–952 (2006).
Bettelli, E., Korn, T., Oukka, M. & Kuchroo, V.K. Induction and effector functions of TH17 cells. Nature 453, 1051–1057 (2008).
Wurster, A.L., Withers, D.J., Uchida, T., White, M.F. & Grusby, M.J. Stat6 and IRS-2 cooperate in interleukin 4 (IL-4)-induced proliferation and differentiation but are dispensable for IL-4-dependent rescue from apoptosis. Mol. Cell. Biol. 22, 117–126 (2002).
Li, M.O. & Flavell, R.A. Contextual regulation of inflammation: a duet by transforming growth factor-β and interleukin-10. Immunity 28, 468–476 (2008).
Izcue, A., Coombes, J.L. & Powrie, F. Regulatory T cells suppress systemic and mucosal immune activation to control intestinal inflammation. Immunol. Rev. 212, 256–271 (2006).
Uhlig, H.H. et al. Characterization of Foxp3+CD4+CD25+ and IL-10-secreting CD4+CD25+ T cells during cure of colitis. J. Immunol. 177, 5852–5860 (2006).
Read, S., Malmstrom, V. & Powrie, F. Cytotoxic T lymphocyte-associated antigen 4 plays an essential role in the function of CD25+CD4+ regulatory cells that control intestinal inflammation. J. Exp. Med. 192, 295–302 (2000).
Kaplan, M.H. & Grusby, M.J. Regulation of T helper cell differentiation by STAT molecules. J. Leukoc. Biol. 64, 2–5 (1998).
Ziegler-Heitbrock, L. et al. IFN-α induces the human IL-10 gene by recruiting both IFN regulatory factor 1 and Stat3. J. Immunol. 171, 285–290 (2003).
Zhou, L. et al. TGF-β-induced Foxp3 inhibits TH17 cell differentiation by antagonizing RORγt function. Nature 453, 236–240 (2008).
Hoffmann, K.F., Cheever, A.W. & Wynn, T.A. IL-10 and the dangers of immune polarization: excessive type 1 and type 2 cytokine responses induce distinct forms of lethal immunopathology in murine schistosomiasis. J. Immunol. 164, 6406–6416 (2000).
Li, C., Corraliza, I. & Langhorne, J. A defect in interleukin-10 leads to enhanced malarial disease in Plasmodium chabaudi chabaudi infection in mice. Infect. Immun. 67, 4435–4442 (1999).
Gazzinelli, R.T., Oswald, I.P., James, S.L. & Sher, A. IL-10 inhibits parasite killing and nitrogen oxide production by IFN-γ-activated macrophages. J. Immunol. 148, 1792–1796 (1992).
Boussiotis, V.A. et al. IL-10-producing T cells suppress immune responses in anergic tuberculosis patients. J. Clin. Invest. 105, 1317–1325 (2000).
Carvalho, E.M. et al. Restoration of IFN-γ production and lymphocyte proliferation in visceral leishmaniasis. J. Immunol. 152, 5949–5956 (1994).
King, C.L. et al. Cytokine control of parasite-specific anergy in human urinary schistosomiasis. IL-10 modulates lymphocyte reactivity. J. Immunol. 156, 4715–4721 (1996).
Makela, M.J. et al. IL-10 is necessary for the expression of airway hyperresponsiveness but not pulmonary inflammation after allergic sensitization. Proc. Natl. Acad. Sci. USA 97, 6007–6012 (2000).
Rubtsov, Y.P. et al. Regulatory T cell-derived interleukin-10 limits inflammation at environmental interfaces. Immunity 28, 546–558 (2008).
Temann, U.A., Geba, G.P., Rankin, J.A. & Flavell, R.A. Expression of interleukin 9 in the lungs of transgenic mice causes airway inflammation, mast cell hyperplasia, and bronchial hyperresponsiveness. J. Exp. Med. 188, 1307–1320 (1998).
Temann, U.A., Ray, P. & Flavell, R.A. Pulmonary overexpression of IL-9 induces Th2 cytokine expression, leading to immune pathology. J. Clin. Invest. 109, 29–39 (2002).
Lu, L.F. et al. Mast cells are essential intermediaries in regulatory T-cell tolerance. Nature 442, 997–1002 (2006).
Tepper, R.I. et al. IL-4 induces allergic-like inflammatory disease and alters T cell development in transgenic mice. Cell 62, 457–467 (1990).
Annacker, O. et al. Essential role for CD103 in the T cell-mediated regulation of experimental colitis. J. Exp. Med. 202, 1051–1061 (2005).
Fahlen, L. et al. T cells that cannot respond to TGF-β escape control by CD4+CD25+ regulatory T cells. J. Exp. Med. 201, 737–746 (2005).
Acknowledgements
We thank D. Kozoriz, R. Chandwaskar and D. Lee for cell sorting and technical assistance; and R. Flavell (Yale University School of Medicine) for IL-10–GFP mice. Supported by the US National Institutes of Health (R01AI073542-01 to M.O., and 1R01NS045937-01, 2R01NS35685-06, 2R37NS30843-11, 1R01A144880-03, 2P01A139671-07, 1P01NS38037-04, 1R01NS046414 and a Javits Neuroscience Investigator Award to V.K.K.), the National Multiple Sclerosis Society (RG-2571-D-9 to V.K.K. and RG-3882-A-1 to M.O.), the Juvenile Diabetes Research Foundation Center for Immunological Tolerance at Harvard Medical School, and the National Multiple Sclerosis Society, New York (A.A. and V.D.).
Author information
Authors and Affiliations
Contributions
V.D. and A.A. performed and designed the experiments, collected the data and contributed to the writing of the manuscript; H.K. and G.G. helped with the immunoprecipitation assays; W.G. and T.B.S. checked Foxp3 expression in IL-4-transgenic T cells; R.A.S. did the pathology analysis; M.M. diagnosed the neurological signs found in RAG-1–reconstituted mice; W.E. and S.K. helped in the studies using anti-IL-9; I.-C.H. generated and provided conditional GATA-3-deficient mice; M.O. designed experiments; V.K.K. designed the experiments, supervised the study and edited the manuscript.
Corresponding authors
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–3 (PDF 5097 kb)
Rights and permissions
About this article
Cite this article
Dardalhon, V., Awasthi, A., Kwon, H. et al. IL-4 inhibits TGF-β-induced Foxp3+ T cells and, together with TGF-β, generates IL-9+ IL-10+ Foxp3− effector T cells. Nat Immunol 9, 1347–1355 (2008). https://doi.org/10.1038/ni.1677
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1677
This article is cited by
-
Immune modulation in malignant pleural effusion: from microenvironment to therapeutic implications
Cancer Cell International (2024)
-
Haemonchus contortus HcL6 promoted the Th9 immune response in goat PBMCs by activating the STAT6/PU.1/NF-κB pathway
Veterinary Research (2023)
-
Regulatory T cells in autoimmune kidney diseases and transplantation
Nature Reviews Nephrology (2023)
-
IL-9-Producing Th9 Cells Participate in the Occurrence and Development of Iodine-Induced Autoimmune Thyroiditis
Biological Trace Element Research (2023)
-
The Dysregulation of Inflammatory Pathways Triggered by Copper Exposure
Biological Trace Element Research (2023)