Abstract
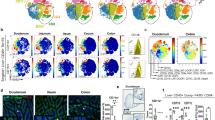
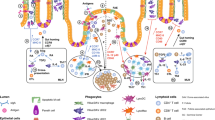
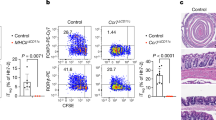
The intestinal cell types responsible for defense against pathogenic organisms remain incompletely characterized. Here we identify a subset of CD11chiCD11bhi lamina propria dendritic cells (LPDCs) that expressed Toll-like receptor 5 (TLR5) in the small intestine. When stimulated by the TLR5 ligand flagellin, TLR5+ LPDCs induced the differentiation of naive B cells into immunoglobulin A–producing plasma cells by a mechanism independent of gut-associated lymphoid tissue. In addition, by a mechanism dependent on TLR5 stimulation, these LPDCs promoted the differentiation of antigen-specific interleukin 17–producing T helper cells and type 1 T helper cells. Unlike spleen DCs, the LPDCs specifically produced retinoic acid, which, in a dose-dependent way, supported the generation and retention of immunoglobulin A–producing cells in the lamina propria and positively regulated the differentiation interleukin 17–producing T helper cells. Our findings demonstrate unique properties of LPDCs and the importance of TLR5 for adaptive immunity in the intestine.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Steinman, R.M., Hawiger, D. & Nussenzweig, M.C. Tolerogenic dendritic cells. Annu. Rev. Immunol. 21, 685–711 (2003).
Kelsall, B.L. & Leon, F. Involvement of intestinal dendritic cells in oral tolerance, immunity to pathogens, and inflammatory bowel disease. Immunol. Rev. 206, 132–148 (2005).
Bilsborough, J., George, T.C., Norment, A. & Viney, J.L. Mucosal CD8α+ DC, with a plasmacytoid phenotype, induce differentiation and support function of T cells with regulatory properties. Immunology 108, 481–492 (2003).
Asselin-Paturel, C., Brizard, G., Pin, J.J., Briere, F. & Trinchieri, G. Mouse strain differences in plasmacytoid dendritic cell frequency and function revealed by a novel monoclonal antibody. J. Immunol. 171, 6466–6477 (2003).
Iwasaki, A. & Kelsall, B.L. Freshly isolated Peyer's patch, but not spleen, dendritic cells produce interleukin 10 and induce the differentiation of T helper type 2 cells. J. Exp. Med. 190, 229–239 (1999).
Macpherson, A.J. & Uhr, T. Induction of protective IgA by intestinal dendritic cells carrying commensal bacteria. Science 303, 1662–1665 (2004).
Mora, J.R. et al. Generation of gut-homing IgA-secreting B cells by intestinal dendritic cells. Science 314, 1157–1160 (2006).
Coombes, J.L. & Maloy, K.J. Control of intestinal homeostasis by regulatory T cells and dendritic cells. Semin. Immunol. 19, 116–126 (2007).
Annacker, O. et al. Essential role for CD103 in the T cell-mediated regulation of experimental colitis. J. Exp. Med. 202, 1051–1061 (2005).
Sun, C.M. et al. Small intestine lamina propria dendritic cells promote de novo generation of Foxp3 T reg cells via retinoic acid. J. Exp. Med. 204, 1775–1785 (2007).
MacPherson, G.G., Jenkins, C.D., Stein, M.J. & Edwards, C. Endotoxin-mediated dendritic cell release from the intestine. Characterization of released dendritic cells and TNF dependence. J. Immunol. 154, 1317–1322 (1995).
Jang, M.H. et al. CCR7 is critically important for migration of dendritic cells in intestinal lamina propria to mesenteric lymph nodes. J. Immunol. 176, 803–810 (2006).
Johansson-Lindbom, B. et al. Functional specialization of gut CD103+ dendritic cells in the regulation of tissue-selective T cell homing. J. Exp. Med. 202, 1063–1073 (2005).
Coombes, J.L. et al. A functionally specialized population of mucosal CD103+ DCs induces Foxp3+ regulatory T cells via a TGF-β and retinoic acid-dependent mechanism. J. Exp. Med. 204, 1757–1764 (2007).
Denning, T.L., Wang, Y.C., Patel, S.R., Williams, I.R. & Pulendran, B. Lamina propria macrophages and dendritic cells differentially induce regulatory and interleukin 17–producing T cell responses. Nat. Immunol. 8, 1086–1094 (2007).
Akira, S., Uematsu, S. & Takeuchi, O. Pathogen recognition and innate immunity. Cell 124, 783–801 (2006).
Abreu, M.T., Fukata, M. & Arditi, M. TLR signaling in the gut in health and disease. J. Immunol. 174, 4453–4460 (2005).
Uematsu, S. et al. Detection of pathogenic intestinal bacteria by Toll-like receptor 5 on intestinal CD11c+ lamina propria cells. Nat. Immunol. 7, 868–874 (2006).
Pavli, P., Woodhams, C.E., Doe, W.F. & Hume, D.A. Isolation and characterization of antigen-presenting dendritic cells from the mouse intestinal lamina propria. Immunology 70, 40–47 (1990).
van Egmond, M. et al. IgA and the IgA Fc receptor. Trends Immunol. 22, 205–211 (2001).
Suzuki, K., Ha, S.A., Tsuji, M. & Fagarasan, S. Intestinal IgA synthesis: a primitive form of adaptive immunity that regulates microbial communities in the gut. Semin. Immunol. 19, 127–135 (2007).
Tseng, J. Transfer of lymphocytes of Peyer's patches between immunoglobulin allotype congenic mice: repopulation of the IgA plasma cells in the gut lamina propria. J. Immunol. 127, 2039–2043 (1981).
Tseng, J. A population of resting IgM-IgD double-bearing lymphocytes in Peyer's patches: the major precursor cells for IgA plasma cells in the gut lamina propria. J. Immunol. 132, 2730–2735 (1984).
Fagarasan, S., Kinoshita, K., Muramatsu, M., Ikuta, K. & Honjo, T. In situ class switching and differentiation to IgA-producing cells in the gut lamina propria. Nature 413, 639–643 (2001).
Suzuki, K., Meek, B., Doi, Y., Honjo, T. & Fagarasan, S. Two distinctive pathways for recruitment of naive and primed IgM+ B cells to the gut lamina propria. Proc. Natl. Acad. Sci. USA 102, 2482–2486 (2005).
Kunisawa, J. et al. Sphingosine 1-phosphate regulates peritoneal B-cell trafficking for subsequent intestinal IgA production. Blood 109, 3749–3756 (2007).
Macpherson, A.J. et al. A primitive T cell-independent mechanism of intestinal mucosal IgA responses to commensal bacteria. Science 288, 2222–2226 (2000).
Litinskiy, M.B. et al. DCs induce CD40-independent immunoglobulin class switching through BLyS and APRIL. Nat. Immunol. 3, 822–829 (2002).
Tezuka, H. et al. Regulation of IgA production by naturally occurring TNF/iNOS-producing dendritic cells. Nature 448, 929–933 (2007).
Muramatsu, M. et al. Class switch recombination and hypermutation require activation-induced cytidine deaminase (AID), a potential RNA editing enzyme. Cell 102, 553–563 (2000).
Kang, H.S. et al. Signaling via LTβR on the lamina propria stromal cells of the gut is required for IgA production. Nat. Immunol. 3, 576–582 (2002).
Hieshima, K. et al. CC chemokine ligands 25 and 28 play essential roles in intestinal extravasation of IgA antibody-secreting cells. J. Immunol. 173, 3668–3675 (2004).
Ivanov, I.I. et al. The orphan nuclear receptor RORγt directs the differentiation program of proinflammatory IL-17+ T helper cells. Cell 126, 1121–1133 (2006).
Korn, T. et al. IL-21 initiates an alternative pathway to induce proinflammatory TH17 cells. Nature 448, 484–487 (2007).
Nurieva, R. et al. Essential autocrine regulation by IL-21 in the generation of inflammatory T cells. Nature 448, 480–483 (2007).
Zhou, L. et al. IL-6 programs TH-17 cell differentiation by promoting sequential engagement of the IL-21 and IL-23 pathways. Nat. Immunol. 8, 967–974 (2007).
Bettelli, E. et al. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature 441, 235–238 (2006).
Mucida, D. et al. Reciprocal TH17 and regulatory T cell differentiation mediated by retinoic acid. Science 317, 256–260 (2007).
Elias, K.M. et al. Retinoic acid inhibits Th17 polarization and enhances FoxP3 expression through a Stat-3/Stat-5 independent signaling pathway. Blood 111, 1013–1020 (2008).
Schambach, F., Schupp, M., Lazar, M.A. & Reiner, S.L. Activation of retinoic acid receptor-α favours regulatory T cell induction at the expense of IL-17-secreting T helper cell differentiation. Eur. J. Immunol. 37, 2396–2399 (2007).
Iwata, M. et al. Retinoic acid imprints gut-homing specificity on T cells. Immunity 21, 527–538 (2004).
Viney, J.L., Mowat, A.M., O'Malley, J.M., Williamson, E. & Fanger, N.A. Expanding dendritic cells in vivo enhances the induction of oral tolerance. J. Immunol. 160, 5815–5825 (1998).
Williamson, E., Westrich, G.M. & Viney, J.L. Modulating dendritic cells to optimize mucosal immunization protocols. J. Immunol. 163, 3668–3675 (1999).
Macpherson, A.J. & Harris, N.L. Interactions between commensal intestinal bacteria and the immune system. Nat. Rev. Immunol. 4, 478–485 (2004).
Niess, J.H. et al. CX3CR1-mediated dendritic cell access to the intestinal lumen and bacterial clearance. Science 307, 254–258 (2005).
Chieppa, M., Rescigno, M., Huang, A.Y. & Germain, R.N. Dynamic imaging of dendritic cell extension into the small bowel lumen in response to epithelial cell TLR engagement. J. Exp. Med. 203, 2841–2852 (2006).
Kolls, J.K. & Linden, A. Interleukin-17 family members and inflammation. Immunity 21, 467–476 (2004).
Yokota, Y. et al. Development of peripheral lymphoid organs and natural killer cells depends on the helix-loop-helix inhibitor Id2. Nature 397, 702–706 (1999).
Kopf, M. et al. Impaired immune and acute-phase responses in interleukin-6-deficient mice. Nature 368, 339–342 (1994).
Barnden, M.J., Allison, J., Heath, W.R. & Carbone, F.R. Defective TCR expression in transgenic mice constructed using cDNA-based α- and β-chain genes under the control of heterologous regulatory elements. Immunol. Cell Biol. 76, 34–40 (1998).
Acknowledgements
We thank N. Kitagaki for technical assistance; T. Kawai and C. Coban for discussions; and A. Shigeta and M. Matsumoto for the distribution of OT-II-transgenic mice. Il6−/− mice (C57BL/6) were provided by M. Kopf (Swiss Federal Institute of Technology), and OT-II transgenic mice (C57BL/6) were provided by W.R. Heath (The Walter and Eliza Hall Institute of Medical Research). Supported by the Ministry of Education, Culture, Sports, Science and Technology in Japan (S.A.), the Ministry of Health, Labour and Welfare in Japan (S.A.), the 21st Century Center of Excellence Program of Japan (S.A.), the World Premier International Research Center (S.A.) and the National Institutes of Health (AI070167 to S.A.).
Author information
Authors and Affiliations
Contributions
K.F. and S.U. did most of the experiments; S.U., K.J.I. and M.H.J. designed all the experiments; B.-G.Y. helped with the immunohistochemical analysis; Y.-J.J. and M.N. helped to isolate cells; S.S., T.T. and M.Y. provided advice for the experiments; Y.Y. provided Id2−/− mice; H.K. and M.M. provided advice for the experiments and manuscript; S.U. and S.A. prepared the manuscript; and S.A. directed the research.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–9 and Supplementary Table 1 (PDF 3389 kb)
Rights and permissions
About this article
Cite this article
Uematsu, S., Fujimoto, K., Jang, M. et al. Regulation of humoral and cellular gut immunity by lamina propria dendritic cells expressing Toll-like receptor 5. Nat Immunol 9, 769–776 (2008). https://doi.org/10.1038/ni.1622
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1622
This article is cited by
-
Mucosal TLR5 activation controls healthspan and longevity
Nature Communications (2024)
-
Recombinant lipidated FLIPr effectively enhances mucosal and systemic immune responses for various vaccine types
npj Vaccines (2023)
-
Investigation of TLR4 Antagonists for Prevention of Intestinal Inflammation
Inflammation (2023)
-
The role of retinoic acid in the production of immunoglobulin A
Mucosal Immunology (2022)
-
Dendritic cell functions in the inductive and effector sites of intestinal immunity
Mucosal Immunology (2022)