Abstract
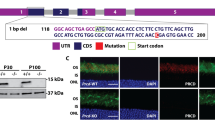
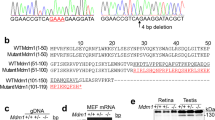
The homologous membrane proteins Rom-1 and peripherin-2 are localized to the disk rims of photoreceptor outer segments (OSs), where they associate as tetramers and larger oligomers1,2,3. Disk rims are thought to be critical for disk morphogenesis, OS renewal4 and the maintenance of OS structure5, but the molecules which regulate these processes are unknown. Although peripherin-2 is known to be required for OS formation (because Prph2−/− mice do not form OSs; ref. 6), and mutations in RDS (the human homologue of Prph2) cause retinal degeneration7, the relationship of Rom-1 to these processes is uncertain. Here we show that Rom1−/− mice form OSs in which peripherin-2 homotetramers are localized to the disk rims, indicating that peripherin-2 alone is sufficient for both disk and OS morphogenesis. The disks produced in Rom1−/− mice were large, rod OSs were highly disorganized (a phenotype which largely normalized with age) and rod photoreceptors died slowly by apoptosis. Furthermore, the maximal photoresponse of Rom1−/− rod photoreceptors was lower than that of controls. We conclude that Rom-1 is required for the regulation of disk morphogenesis and the viability of mammalian rod photoreceptors, and that mutations in human ROM1 may cause recessive photoreceptor degeneration.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Bascom, R.A. et al. Cloning of the cDNA for a novel photoreceptor membrane protein (rom-1) identifies a disk rim protein family implicated in human retinopathies . Neuron 8, 1171–1184 (1992).
Kedzierski, W., Weng, J. & Travis, G.H. Analysis of the rds/peripherin:rom1 complex in transgenic photoreceptors that express a chimeric protein. J. Biol. Chem. 274, 29181–29187 (1999).
Loewen, C.J.R. & Molday, R.S. Disulfide-mediated oligomerization of peripherin/rds and rom-1 from photoreceptor disk membranes. Implications for photoreceptor outer segment morphogenesis and degeneration. J. Biol. Chem. 275, 5370–5378 (2000).
Young, R.W. Visual cells and the concept of renewal. Invest. Ophthalmol. Vis. Sci. 15, 700–725 ( 1976).
Steinberg, R.H., Fisher, S.K. & Anderson, D.H. Disc morphogenesis in vertebrate photoreceptors. J. Comp. Neurol. 190, 501–519 (1980).
Sanyal, S., De Ruiter, A. & Hawkins, R.K. Development and degeneration of retina in rds mutant mice: light microscopy. J. Comp. Neurol. 194, 193–207 (1980).
Kohl, S. et al. The role of the peripherin/RDS gene in retinal dystrophies . Acta Anat. 162, 75–84 (1998).
Travis, G.H. Mechanisms of cell death in the inherited retinal degenerations. Am. J. Hum. Genet. 62, 503–508 (1998).
Hawkins, R.K., Jansen, H.G. & Sanyal, S. Development and degeneration of retina in rds mutant mice: photoreceptor abnormalities in the heterozygotes. Exp. Eye Res. 41, 701–720 (1985).
Carter-Dawson, L.D. & LaVail, M.M. Rods and cones in the mouse retina. I. Structural analysis using light and electron microscopy . J. Comp. Neurol. 188, 245– 262 (1979).
Williams, D.S. Actin filaments and photoreceptor membrane turnover. Bioessays 13, 171–178 ( 1991).
Arikawa, K., Molday, L.L., Molday, R.S. & Williams, D.S. Localization of peripherin/rds in disk membranes of cone and rod photoreceptors: relationship to disk membrane morphogenesis and retinal degeneration. J. Cell Biol. 116, 659–667 (1992).
Williams, D.S., Linberg, K.A., Vaughan, D.K., Fariss, R.N. & Fisher, S.K. Disruption of microfilament organization and deregulation of disk membrane morphogenesis by cytochalasin D in rod and cone photoreceptors. J. Comp. Neurol. 272, 161–176 (1988).
Boesze-Battaglia, K., Lamba, O.P., Napoli, A.A. Jr, Sinha, S. & Guo, Y. Fusion between retinal rod outer segment membranes and model membranes: a role for photoreceptor peripherin/rds. Biochemistry 37, 9477– 9487 (1998).
Goldberg, A.F.X., Moritz, O.L. & Molday, R.S. Heterologous expression of photoreceptor peripherin/rds and rom-1 in COS-1 cells: assembly, interactions, and localization of multisubunit complexes. Biochemistry 34, 14213– 14219 (1995).
Illing, M., Molday, L.L. & Molday, R.S. The 220-kDa rim protein of retinal rod outer segments is a member of the ABC transporter superfamily. J. Biol. Chem. 272, 10303–10310 (1997).
Moritz, O.L. & Molday, R.S. Molecular cloning, membrane topology, and localization of bovine rom-1 in rod and cone photoreceptor cells. Invest. Ophthalmol. Vis. Sci. 37, 352– 362 (1996).
Bascom, R.A., Schappert, K. & McInnes, R.R. Cloning of the human and murine ROM1 genes: genomic orgnization and sequence conservation. Hum. Mol. Genet . 2, 385–391 ( 1993).
Dryja, T.P., Hahn, L.B., Kajiwara, K. & Berson, E.L. Dominant and digenic mutations in the Peripherin/RDS and ROM1 genes in retinitis pigmentosa. Invest. Ophthalmol. Vis. Sci. 38, 1972–1982 (1997).
Tybulewicz, V.L.J., Crawford, C.E., Jackson, P.K., Bronson, R.T. & Mulligan, R.C. Neonatal lethality and lymphopenia in mice with a homozygous disruption of the c-abl proto-oncogene. Cell 65, 1153–1163 ( 1991).
Nagy, A., Rossant, J., Nagy, R., Abramow-Newerly, W. & Roder, J.C. Derivation of completely cell culture-derived mice from early-passage embryonic stem cells. Proc. Natl Acad. Sci. USA 90, 8424–8428 ( 1993).
Sambrook, J., Fritsch, E.F. & Maniatis, T. Molecular Cloning: A Laboratory Manual (Cold Spring Harbor Laboratory Press, New York, 1989).
Nagy, A. & Rossant, J. in Gene Targeting: A Practical Approach (ed. Joyner, A.L.) 147–179 (Oxford University Press, Oxford, 1993).
Connell, G. et al. Photoreceptor peripherin is the normal product of the gene responsible for retinal degeneration slow in the rds mouse. Proc. Natl Acad. Sci. USA 88, 723– 726 (1991).
Molday, R.S. & MacKenzie, D. Monoclonal antibodies to rhodopsin: characterization, cross-reactivity, and application as structural probes. Biochemistry 22, 653–660 (1983).
Röhlich, P. & Szél, Á. Binding sites of photoreceptor-specific antibodies COS-1, OS-2 and AO. Exp. Eye Res. 12, 935–944 (1993).
Michon, J.J., Li, Z.-L., Shioura, N., Anderson, R.J. & Tso, M.O.M. A comparative study of methods of photoreceptor morphometry. Invest. Ophthalmol. Vis. Sci. 32, 280–284 (1991).
Kedzierski, W., Lloyd, M., Birch, D.G., Bok, D. & Travis, G.H. Generation and analysis of transgenic mice expressing P216L-substituted rds/peripherin in rod photoreceptors. Invest. Ophthalmol. Vis. Sci. 38, 498–509 (1997).
Goldberg, A.F.X. & Molday, R.S. Subunit composition of the peripher/rds-rom-1 disk rim complex from rod photoreceptors: hydrodynamic evidence for a tetrameric quarternary structure. Biochemistry 35, 6144–6149 (1996).
Goldberg, A.F.X., Loewen, C.J.R. & Molday, R.S. Cysteine residues of photoreceptor peripherin/rds: role in subunit assembly and autosomal dominant retinitis pigmentosa. Biochemistry 37, 680–685 (1998).
Acknowledgements
We thank J.G. Hollyfield, D. Bok, A. Aguirre and S. Fisher for helpful comments; J. Saari for the RDH-E6 anti-peripherin-2 antibody; G.H. Travis for the anti-peripherin-2 antibody D2P4; J. Nathans for the 2379 antibody to the Abcr protein; and B. Calvieri, D. Gross and I. Diplock for technical assistance. This work was supported by the Foundation Fighting Blindness (R.R.M.), the RP Eye Research Foundation of Canada (R.R.M. and R.S.M.), the NEI (EY02422) (R.S.M.), the Medical Research Council (Canada) (R.S.M. and J.R.) and the Canadian Genetic Disease Network (R.R.M.). G.C. was a recipient of a RP Eye Research Foundation of Canada Studentship. R.R.M. and J.R. are International Research Scholars of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Clarke, G., Goldberg, A., Vidgen, D. et al. Rom-1 is required for rod photoreceptor viability and the regulation of disk morphogenesis. Nat Genet 25, 67–73 (2000). https://doi.org/10.1038/75621
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/75621
This article is cited by
-
Glaucoma related Proteomic Alterations in Human Retina Samples
Scientific Reports (2016)
-
Retinal remodeling
Japanese Journal of Ophthalmology (2012)
-
Structural and functional relationships between photoreceptor tetraspanins and other superfamily members
Cellular and Molecular Life Sciences (2012)
-
Analysis of a zebrafish dync1h1mutant reveals multiple functions for cytoplasmic dynein 1 during retinal photoreceptor development
Neural Development (2010)