Abstract
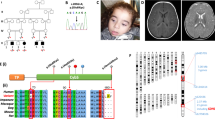
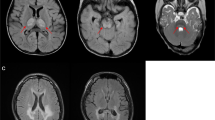
An isolated defect of respiratory chain complex I activity is a frequent biochemical abnormality in mitochondrial disorders. Despite intensive investigation in recent years, in most instances, the molecular basis underpinning complex I defects remains unknown. We report whole-exome sequencing of a single individual with severe, isolated complex I deficiency. This analysis, followed by filtering with a prioritization of mitochondrial proteins, led us to identify compound heterozygous mutations in ACAD9, which encodes a poorly understood member of the mitochondrial acyl-CoA dehydrogenase protein family. We demonstrated the pathogenic role of the ACAD9 variants by the correction of the complex I defect on expression of the wildtype ACAD9 protein in fibroblasts derived from affected individuals. ACAD9 screening of 120 additional complex I–defective index cases led us to identify two additional unrelated cases and a total of five pathogenic ACAD9 alleles.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Ng, S.B. et al. Targeted capture and massively parallel sequencing of 12 human exomes. Nature 461, 272–276 (2009).
Hedges, D.J. et al. Exome sequencing of a multigenerational human pedigree. PLoS ONE 4, e8232 (2009).
Ng, S.B. et al. Exome sequencing identifies the cause of a mendelian disorder. Nat. Genet. 42, 30–35 (2010).
Hoischen, A. et al. De novo mutations of SETBP1 cause Schinzel-Giedion syndrome. Nat. Genet. 42, 483–485 (2010).
Choi, M. et al. Genetic diagnosis by whole exome capture and massively parallel DNA sequencing. Proc. Natl. Acad. Sci. USA 106, 19096–19101 (2009).
Triepels, R.H., Van Den Heuvel, L.P., Trijbels, J.M. & Smeitink, J.A. Respiratory chain complex I deficiency. Am. J. Med. Genet. 106, 37–45 (2001).
Thorburn, D.R. et al. Biochemical and molecular diagnosis of mitochondrial respiratory chain disorders. Biochim. Biophys. Acta 1659, 121–128 (2004).
Elstner, M., Andreoli, C., Klopstock, T., Meitinger, T. & Prokisch, H. The mitochondrial proteome database: MitoP2. Methods Enzymol. 457, 3–20 (2009).
Gobin-Limballe, S. et al. Genetic basis for correction of very-long-chain acyl-coenzyme A dehydrogenase deficiency by bezafibrate in patient fibroblasts: toward a genotype-based therapy. Am. J. Hum. Genet. 81, 1133–1143 (2007).
Saijo, T. & Tanaka, K. Isoalloxazine ring of FAD is required for the formation of the core in the Hsp60-assisted folding of medium chain acyl-CoA dehydrogenase subunit into the assembly competent conformation in mitochondria. J. Biol. Chem. 270, 1899–1907 (1995).
Zhang, J. et al. Cloning and functional characterization of ACAD-9, a novel member of human acyl-CoA dehydrogenase family. Biochem. Biophys. Res. Commun. 297, 1033–1042 (2002).
Ensenauer, R. et al. Human acyl-CoA dehydrogenase-9 plays a novel role in the mitochondrial beta-oxidation of unsaturated fatty acids. J. Biol. Chem. 280, 32309–32316 (2005).
He, M. et al. A new genetic disorder in mitochondrial fatty acid beta-oxidation: ACAD9 deficiency. Am. J. Hum. Genet. 81, 87–103 (2007).
Feng, D., Witkowski, A. & Smith, S. Down-regulation of mitochondrial acyl carrier protein in mammalian cells compromises protein lipoylation and respiratory complex I and results in cell death. J. Biol. Chem. 284, 11436–11445 (2009).
Nouws, J. et al. Acyl-CoA dehydrogenase 9 is required for the biogenesis of oxidative phosphorylation complex I. Cell Metab. 12, 283–294 (2010).
Ohtsuka, T., Nishijima, M., Suzuki, K. & Akamatsu, Y. Mitochondrial dysfunction of a cultured Chinese hamster ovary cell mutant deficient in cardiolipin. J. Biol. Chem. 268, 22914–22919 (1993).
Ghezzi, D. et al. Severe X-linked mitochondrial encephalomyopathy associated with a mutation in apoptosis-inducing factor. Am. J. Hum. Genet. 86, 639–649 (2010).
Gempel, K. et al. The myopathic form of coenzyme Q10 deficiency is caused by mutations in the electron-transferring-flavoprotein dehydrogenase (ETFDH) gene. Brain 130, 2037–2044 (2007).
Homer, N., Merriman, B. & Nelson, S.F. BFAST: an alignment tool for large scale genome resequencing. PLoS ONE 4, e7767 (2009).
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
International HapMap Consortium. The International HapMap Project. Nature 426, 789–796 (2003).
Sherry, S.T. et al. dbSNP: the NCBI database of genetic variation. Nucleic Acids Res. 29, 308–311 (2001).
Meisinger, C. et al. A genome-wide association study identifies three loci associated with mean platelet volume. Am. J. Hum. Genet. 84, 66–71 (2009).
Kuznetsov, A.V. et al. Analysis of mitochondrial function in situ in permeabilized muscle fibers, tissues and cells. Nat. Protoc. 3, 965–976 (2008).
Sgobbo, P., Pacelli, C., Grattagliano, I., Villani, G. & Cocco, T. Carvedilol inhibits mitochondrial complex I and induces resistance to H2O2-mediated oxidative insult in H9C2 myocardial cells. Biochim. Biophys. Acta 1767, 222–232 (2007).
Wittig, I., Braun, H.P. & Schagger, H. Blue native PAGE. Nat. Protoc. 1, 418–428 (2006).
Schägger, H. Tricine-SDS-PAGE. Nat. Protoc. 1, 16–22 (2006).
Schwarz, J.M., Rödelsperger, C., Schuelke, M. & Seelow, D. MutationTaster evaluates disease-causing potential of sequence alterations. Nat. Methods 7, 575–576 (2010).
Acknowledgements
We are indebted to the subjects and their families involved in the study. We gratefully acknowledge the support of R. Hellinger, A. Löschner, M. Bada and F. Carrara in genotyping and cell culture work. Thanks to C. Fratter, M. Pike and S. Olpin. T.M. and H.P. were supported by the Impulse and Networking Fund of the Helmholtz Association in the framework of the Helmholtz Alliance for Mental Health in an Ageing Society (HA-215), the German Federal Ministry of Education and Research (BMBF) funded German National Research Network (NGFNplus #01GS08134), German Network for Mitochondrial Disorders (mitoNET #01GM0862 and 01GM0867), German Center for Diabetes Research (DZD e.V.) and grant RF-INN-2007-634163 of the Italian Ministry of Health. H.-W.M., T.M. and H.P. were supported by the Systems Biology of Metabotypes (SysMBo #0315494A). I.W. was supported by the Deutsche Forschungsgemeinschaft, Sonderforschungsbereich 815, Project Z1 (Redox-Proteomics), by the German Network for Mitochondrial Disorders (mitoNET #01GM0863) and by the Cluster of Excellence 'Macromolecular Complexes' at the Goethe-University (EXC 115). M.Z. was supported by the Pierfranco and Luisa Mariani Foundation Italy, Fondazione Telethon Italy grant number GGP07019, Fondazione Giuseppe Tomasello–ONLUS and grant RF-INN-2007-634163 of the Italian Ministry of Health. J.P. was supported by the Medical Research Council (UK) and the National Commissioning Group.
Author information
Authors and Affiliations
Contributions
Project planning: T.M., M.Z., H.P. Experimental design: H.P. Review of phenotypes and sample collection: G.U., E.L., F.I., J.P. and B.R. Mutation screening: T.B.H., D.B. and S.B. Data analysis: T.B.H., J.H., T.S., H.-W.M. and H.P. Cell biology experiments: K.D., B.H., V.S., I.W. and A.I. Manuscript writing: T.B.H., T.M., M.Z. and H.P. Critical revision of the manuscript: all authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Tables 1–4 and Supplementary Note (PDF 110 kb)
Rights and permissions
About this article
Cite this article
Haack, T., Danhauser, K., Haberberger, B. et al. Exome sequencing identifies ACAD9 mutations as a cause of complex I deficiency. Nat Genet 42, 1131–1134 (2010). https://doi.org/10.1038/ng.706
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.706
This article is cited by
-
New insights into the nutritional genomics of adult-onset riboflavin-responsive diseases
Nutrition & Metabolism (2023)
-
Transcriptional profiles of pulmonary artery endothelial cells in pulmonary hypertension
Scientific Reports (2023)
-
Diagnostic yield of clinical exome sequencing as a first-tier genetic test for the diagnosis of genetic disorders in pediatric patients: results from a referral center study
Human Genetics (2022)
-
Ophthalmic genetic counselling: emerging trends in practice perspectives in Asia
Journal of Community Genetics (2022)
-
“Transcriptomics”: molecular diagnosis of inborn errors of metabolism via RNA‐sequencing
Journal of Inherited Metabolic Disease (2018)