Abstract
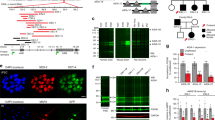
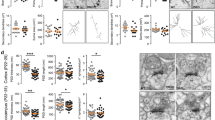
Gain-of-function mutations in some genes underlie neurodegenerative conditions, whereas loss-of-function mutations in the same genes have distinct phenotypes. This appears to be the case with the protein ataxin 1 (ATXN1), which forms a transcriptional repressor complex with capicua (CIC). Gain of function of the complex leads to neurodegeneration, but ATXN1–CIC is also essential for survival. We set out to understand the functions of the ATXN1–CIC complex in the developing forebrain and found that losing this complex results in hyperactivity, impaired learning and memory, and abnormal maturation and maintenance of upper-layer cortical neurons. We also found that CIC activity in the hypothalamus and medial amygdala modulates social interactions. Informed by these neurobehavioral features in mouse mutants, we identified five individuals with de novo heterozygous truncating mutations in CIC who share similar clinical features, including intellectual disability, attention deficit/hyperactivity disorder (ADHD), and autism spectrum disorder. Our study demonstrates that loss of ATXN1–CIC complexes causes a spectrum of neurobehavioral phenotypes.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Accession codes
References
Orr, H.T. et al. Expansion of an unstable trinucleotide CAG repeat in spinocerebellar ataxia type 1. Nat. Genet. 4, 221–226 (1993).
Burright, E.N. et al. SCA1 transgenic mice: a model for neurodegeneration caused by an expanded CAG trinucleotide repeat. Cell 82, 937–948 (1995).
Matilla, A. et al. Mice lacking ataxin-1 display learning deficits and decreased hippocampal paired-pulse facilitation. J. Neurosci. 18, 5508–5516 (1998).
Watase, K. et al. A long CAG repeat in the mouse Sca1 locus replicates SCA1 features and reveals the impact of protein solubility on selective neurodegeneration. Neuron 34, 905–919 (2002).
Asher, M., Johnson, A., Zecevic, B., Pease, D. & Cvetanovic, M. Ataxin-1 regulates proliferation of hippocampal neural precursors. Neuroscience 322, 54–65 (2016).
Lin, X., Antalffy, B., Kang, D., Orr, H.T. & Zoghbi, H.Y. Polyglutamine expansion down-regulates specific neuronal genes before pathologic changes in SCA1. Nat. Neurosci. 3, 157–163 (2000).
Lam, Y.C. et al. ATAXIN-1 interacts with the repressor Capicua in its native complex to cause SCA1 neuropathology. Cell 127, 1335–1347 (2006).
Bowman, A.B. et al. Duplication of Atxn1l suppresses SCA1 neuropathology by decreasing incorporation of polyglutamine-expanded ataxin-1 into native complexes. Nat. Genet. 39, 373–379 (2007).
Lee, Y. et al. ATXN1 protein family and CIC regulate extracellular matrix remodeling and lung alveolarization. Dev. Cell 21, 746–757 (2011).
Fryer, J.D. et al. Exercise and genetic rescue of SCA1 via the transcriptional repressor Capicua. Science 334, 690–693 (2011).
Celestino-Soper, P.B. et al. Deletions in chromosome 6p22.3-p24.3, including ATXN1, are associated with developmental delay and autism spectrum disorders. Mol. Cytogenet. 5, 17 (2012).
Barøy, T. et al. Haploinsufficiency of two histone modifier genes on 6p22.3, ATXN1 and JARID2, is associated with intellectual disability. Orphanet J. Rare Dis. 8, 3 (2013).
Di Benedetto, D. et al. 6p22.3 deletion: report of a patient with autism, severe intellectual disability and electroencephalographic anomalies. Mol. Cytogenet. 6, 4 (2013).
Vissers, L.E. et al. A de novo paradigm for mental retardation. Nat. Genet. 42, 1109–1112 (2010).
Athanasakis, E. et al. Next generation sequencing in nonsyndromic intellectual disability: from a negative molecular karyotype to a possible causative mutation detection. Am. J. Med. Genet. A. 164A, 170–176 (2014).
Giedd, J.N. & Rapoport, J.L. Structural MRI of pediatric brain development: what have we learned and where are we going? Neuron 67, 728–734 (2010).
Ecker, C., Bookheimer, S.Y. & Murphy, D.G. Neuroimaging in autism spectrum disorder: brain structure and function across the lifespan. Lancet Neurol. 14, 1121–1134 (2015).
Gorski, J.A. et al. Cortical excitatory neurons and glia, but not GABAergic neurons, are produced in the Emx1-expressing lineage. J. Neurosci. 22, 6309–6314 (2002).
Sotnikoca, T.D. & Gainedinov, R.R. in Behavioral Genetics of the Mouse Vol. 2 (eds. Pietropaolo, S., Sluyter, F. & Crusio, W.E.) 164–172 (Cambridge University Press, 2014).
Jonas, P. & Lisman, J. Structure, function, and plasticity of hippocampal dentate gyrus microcircuits. Front. Neural Circuits 8, 107 (2014).
Fame, R.M., MacDonald, J.L. & Macklis, J.D. Development, specification, and diversity of callosal projection neurons. Trends Neurosci. 34, 41–50 (2011).
Hevner, R.F. et al. Beyond laminar fate: toward a molecular classification of cortical projection/pyramidal neurons. Dev. Neurosci. 25, 139–151 (2003).
Goebbels, S. et al. Genetic targeting of principal neurons in neocortex and hippocampus of NEX-Cre mice. Genesis 44, 611–621 (2006).
Cubelos, B. et al. Cux1 and Cux2 regulate dendritic branching, spine morphology, and synapses of the upper layer neurons of the cortex. Neuron 66, 523–535 (2010).
Doan, R.N. et al. Mutations in human accelerated regions disrupt cognition and social behavior. Cell 167, 341–354 (2016).
Kim, J.Y. et al. Viral transduction of the neonatal brain delivers controllable genetic mosaicism for visualising and manipulating neuronal circuits in vivo. Eur. J. Neurosci. 37, 1203–1220 (2013).
Acampora, D. et al. Progressive impairment of developing neuroendocrine cell lineages in the hypothalamus of mice lacking the Orthopedia gene. Genes Dev. 13, 2787–2800 (1999).
Díaz, C., Morales-Delgado, N. & Puelles, L. Ontogenesis of peptidergic neurons within the genoarchitectonic map of the mouse hypothalamus. Front. Neuroanat. 8, 162 (2015).
Zhou, P. et al. Interrogating translational efficiency and lineage-specific transcriptomes using ribosome affinity purification. Proc. Natl. Acad. Sci. USA 110, 15395–15400 (2013).
Alvarez-Bolado, G., Grinevich, V. & Puelles, L. Development of the hypothalamus. Front. Neuroanat. 9, 83 (2015).
Kim, Y. et al. Mapping social behavior–induced brain activation at cellular resolution in the mouse. Cell Rep. 10, 292–305 (2015).
Kas, M.J., Modi, M.E., Saxe, M.D. & Smith, D.G. Advancing the discovery of medications for autism spectrum disorder using new technologies to reveal social brain circuitry in rodents. Psychopharmacology (Berl.) 231, 1147–1165 (2014).
Mellén, M., Ayata, P., Dewell, S., Kriaucionis, S. & Heintz, N. MeCP2 binds to 5hmC enriched within active genes and accessible chromatin in the nervous system. Cell 151, 1417–1430 (2012).
Lek, M. et al. Analysis of protein-coding genetic variation in 60,706 humans. Nature 536, 285–291 (2016).
Shaw, P. et al. Attention-deficit/hyperactivity disorder is characterized by a delay in cortical maturation. Proc. Natl. Acad. Sci. USA 104, 19649–19654 (2007).
Shaw, P. et al. Longitudinal mapping of cortical thickness and clinical outcome in children and adolescents with attention-deficit/hyperactivity disorder. Arch. Gen. Psychiatry 63, 540–549 (2006).
Sobreira, N., Schiettecatte, F., Valle, D. & Hamosh, A. GeneMatcher: a matching tool for connecting investigators with an interest in the same gene. Hum. Mutat. 36, 928–930 (2015).
Warming, S., Costantino, N., Court, D.L., Jenkins, N.A. & Copeland, N.G. Simple and highly efficient BAC recombineering using galK selection. Nucleic Acids Res. 33, e36 (2005).
Chao, H.T. et al. Dysfunction in GABA signalling mediates autism-like stereotypies and Rett syndrome phenotypes. Nature 468, 263–269 (2010).
Han, K. et al. SHANK3 overexpression causes manic-like behaviour with unique pharmacogenetic properties. Nature 503, 72–77 (2013).
Crespo-Barreto, J., Fryer, J.D., Shaw, C.A., Orr, H.T. & Zoghbi, H.Y. Partial loss of ataxin-1 function contributes to transcriptional dysregulation in spinocerebellar ataxia type 1 pathogenesis. PLoS Genet. 6, e1001021 (2010).
Park, J. et al. RAS–MAPK–MSK1 pathway modulates ataxin 1 protein levels and toxicity in SCA1. Nature 498, 325–331 (2013).
Schindelin, J. et al. Fiji: an open-source platform for biological-image analysis. Nat. Methods 9, 676–682 (2012).
Yaylaoglu, M.B. et al. Comprehensive expression atlas of fibroblast growth factors and their receptors generated by a novel robotic in situ hybridization platform. Dev. Dyn. 234, 371–386 (2005).
Schmittgen, T.D. & Livak, K.J. Analyzing real-time PCR data by the comparative C T method. Nat. Protoc. 3, 1101–1108 (2008).
Yuan, J.S., Reed, A., Chen, F. & Stewart, C.N. Jr. Statistical analysis of real-time PCR data. BMC Bioinformatics 7, 85 (2006).
Kim, D. et al. TopHat2: accurate alignment of transcriptomes in the presence of insertions, deletions and gene fusions. Genome Biol. 14, R36 (2013).
Anders, S., Pyl, P.T. & Huber, W. HTSeq—a Python framework to work with high-throughput sequencing data. Bioinformatics 31, 166–169 (2015).
Wang, J., Duncan, D., Shi, Z. & Zhang, B. WEB-based GEne SeT AnaLysis Toolkit (WebGestalt): update 2013. Nucleic Acids Res. 41, W77–W83 (2013).
Zhang, B., Kirov, S. & Snoddy, J. WebGestalt: an integrated system for exploring gene sets in various biological contexts. Nucleic Acids Res. 33, W741–W748 (2005).
Acknowledgements
We thank the families for their participation in the study and H. Zheng (Baylor College of Medicine) for providing Neurod6-Cre (NEX-Cre) mice. This project was partly supported by the Genomic and RNA Profiling Core at Baylor College of Medicine and the expert assistance of the core director, L.D. White. We would like to thank the Mayo Clinic Center for Individualized Medicine Investigative and Functional Genomics Program for funding and support. The project was supported by grants NIH/NICHD R01 HD081216 and HD083809 to R.H.F.; NIH/NHGRI 1UM1 HG008898-01 to M.N.B.; NIH/NINDS R37 NS22920 to H.T.O.; NIH/NINDS R01 NS089664 to R.V.S.; NIH/NIGMS R01 GM120033, NSF DMS-1263932 and CPRIT RP170387 to Z.L.; and NIH/NINDS R01 NS027699-26 and R37 NS027699-28 to H.Y.Z. Q.T. was supported by NIH/NINDS F32 NS083091, and M.W.C.R. received support from Canadian Institutes of Health Research Fellowship 201210MFE-290072–173743. H.Y.Z. is an investigator of the Howard Hughes Medical Institute. We thank the RNA In Situ Hybridization Core (supported by Shared Instrumentation grant 1S10OD016167), the Microscopy Core, and the Neuropathology Core at Baylor College of Medicine. All three cores are supported by NIH IDDRC grant U54 HD083092 from the Eunice Kennedy Shriver National Institute of Child Health and Human Development. The content is solely the responsibility of the authors and does not necessarily represent the official views of the Eunice Kennedy Shriver National Institute of Child Health and Human Development or the National Institutes of Health.
Author information
Authors and Affiliations
Contributions
H.-C.L., Q.T., and H.Y.Z. conceived and designed the experiments. H.T.O. and R.V.S. provided critical input to the project. H.L., Q.T., M.W.C.R., W.W., J.-Y.K., R.R., S.-Y.Y., J.M.P., T.L., and M.C.L. performed the experiments. X.L., Y.L., J.D.F., J.H., and M.C. assisted in generating the mouse models. Y.-W.W. and Z.L. performed RNA–seq analysis. M.E.Z.-J. cultured fibroblasts for patient 1. Q.T. performed human fibroblast RNA and protein analysis. H.L., Q.T., R.H.F., Y.L., P.A., K.A.-Y., L.V.M., D.L., N.J.-M., A.-L.M.-B., J.T., M.A.C., D.E.B., B.C.L., E.W.K., N.A., M.N.B., C.P.S., and H.Y.Z. recruited patients, acquired clinical data, or analyzed whole-exome or Sanger sequencing results. H.L., Q.T., and H.Y.Z. drafted the original manuscript, and all authors assisted in editing the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–23 and Supplementary Note (PDF 3573 kb)
Supplementary Table 1
Gene expression analysis of Otp-Cre; Cicflox/flox; ROSAfsTRAP and Otp-Cre; ROSAfsTRAP neurons. (XLSX 59 kb)
Supplementary Table 2
Gene set expression analysis of differentially expressed genes between Otp-Cre; Cicflox/flox; ROSAfsTRAP and Otp-Cre; ROSAfsTRAP neurons. (XLSX 13 kb)
Supplementary Table 3
Details of statistical analysis. (XLSX 31 kb)
Supplementary Table 4
Primer sequences used in this study. (XLSX 31 kb)
Rights and permissions
About this article
Cite this article
Lu, HC., Tan, Q., Rousseaux, M. et al. Disruption of the ATXN1–CIC complex causes a spectrum of neurobehavioral phenotypes in mice and humans. Nat Genet 49, 527–536 (2017). https://doi.org/10.1038/ng.3808
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.3808
This article is cited by
-
Exploring genetic testing requests, genetic alterations and clinical associations in a cohort of children with autism spectrum disorder
European Child & Adolescent Psychiatry (2024)
-
Molecular Determinants of Neurocognitive Deficits in Glioma: Based on 2021 WHO Classification
Journal of Molecular Neuroscience (2024)
-
Association between birth by caesarian section and anxiety, self-harm: a gene-environment interaction study using UK Biobank data
BMC Psychiatry (2023)
-
Functional implications of paralog genes in polyglutamine spinocerebellar ataxias
Human Genetics (2023)
-
Identification of the ataxin-1 interaction network and its impact on spinocerebellar ataxia type 1
Human Genomics (2022)