Abstract
Pediatric midline high-grade astrocytomas (mHGAs) are incurable with few treatment targets identified. Most tumors harbor mutations encoding p.Lys27Met in histone H3 variants. In 40 treatment-naive mHGAs, 39 analyzed by whole-exome sequencing, we find additional somatic mutations specific to tumor location. Gain-of-function mutations in ACVR1 occur in tumors of the pons in conjunction with histone H3.1 p.Lys27Met substitution, whereas FGFR1 mutations or fusions occur in thalamic tumors associated with histone H3.3 p.Lys27Met substitution. Hyperactivation of the bone morphogenetic protein (BMP)-ACVR1 developmental pathway in mHGAs harboring ACVR1 mutations led to increased levels of phosphorylated SMAD1, SMAD5 and SMAD8 and upregulation of BMP downstream early-response genes in tumor cells. Global DNA methylation profiles were significantly associated with the p.Lys27Met alteration, regardless of the mutant histone H3 variant and irrespective of tumor location, supporting the role of this substitution in driving the epigenetic phenotype. This work considerably expands the number of potential treatment targets and further justifies pretreatment biopsy in pediatric mHGA as a means to orient therapeutic efforts in this disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
References
Khuong-Quang, D.A. et al. K27M mutation in histone H3.3 defines clinically and biologically distinct subgroups of pediatric diffuse intrinsic pontine gliomas. Acta Neuropathol. 124, 439–447 (2012).
Schwartzentruber, J. et al. Driver mutations in histone H3.3 and chromatin remodelling genes in paediatric glioblastoma. Nature 482, 226–231 (2012).
Wu, G. et al. Somatic histone H3 alterations in pediatric diffuse intrinsic pontine gliomas and non-brainstem glioblastomas. Nat. Genet. 44, 251–253 (2012).
Sturm, D. et al. Hotspot mutations in H3F3A and IDH1 define distinct epigenetic and biological subgroups of glioblastoma. Cancer Cell 22, 425–437 (2012).
Fontebasso, A.M. et al. Mutations in SETD2 and genes affecting histone H3K36 methylation target hemispheric high-grade gliomas. Acta Neuropathol. 125, 659–669 (2013).
Paugh, B.S. et al. Genome-wide analyses identify recurrent amplifications of receptor tyrosine kinases and cell-cycle regulatory genes in diffuse intrinsic pontine glioma. J. Clin. Oncol. 29, 3999–4006 (2011).
Zarghooni, M. et al. Whole-genome profiling of pediatric diffuse intrinsic pontine gliomas highlights platelet-derived growth factor receptor α and poly (ADP-ribose) polymerase as potential therapeutic targets. J. Clin. Oncol. 28, 1337–1344 (2010).
Jones, D.T. et al. Recurrent somatic alterations of FGFR1 and NTRK2 in pilocytic astrocytoma. Nat. Genet. 45, 927–932 (2013).
Shore, E.M. et al. A recurrent mutation in the BMP type I receptor ACVR1 causes inherited and sporadic fibrodysplasia ossificans progressiva. Nat. Genet. 38, 525–527 (2006).
Fukuda, T. et al. Generation of a mouse with conditionally activated signaling through the BMP receptor, ALK2. Genesis 44, 159–167 (2006).
Chaikuad, A. et al. Structure of the bone morphogenetic protein receptor ALK2 and implications for fibrodysplasia ossificans progressiva. J. Biol. Chem. 287, 36990–36998 (2012).
Fukuda, T. et al. A unique mutation of ALK2, G356D, found in a patient with fibrodysplasia ossificans progressiva is a moderately activated BMP type I receptor. Biochem. Biophys. Res. Commun. 377, 905–909 (2008).
Mishina, Y., Crombie, R., Bradley, A. & Behringer, R.R. Multiple roles for activin-like kinase-2 signaling during mouse embryogenesis. Dev. Biol. 213, 314–326 (1999).
Kishigami, S. et al. BMP signaling through ACVRI is required for left-right patterning in the early mouse embryo. Dev. Biol. 276, 185–193 (2004).
Miyazono, K., Maeda, S. & Imamura, T. BMP receptor signaling: transcriptional targets, regulation of signals, and signaling cross-talk. Cytokine Growth Factor Rev. 16, 251–263 (2005).
Korchynskyi, O. & ten Dijke, P. Identification and functional characterization of distinct critically important bone morphogenetic protein–specific response elements in the Id1 promoter. J. Biol. Chem. 277, 4883–4891 (2002).
Singh, D. et al. Transforming fusions of FGFR and TACC genes in human glioblastoma. Science 337, 1231–1235 (2012).
Zhang, J. et al. Whole-genome sequencing identifies genetic alterations in pediatric low-grade gliomas. Nat. Genet. 45, 602–612 (2013).
Paugh, B.S. et al. Integrated molecular genetic profiling of pediatric high-grade gliomas reveals key differences with the adult disease. J. Clin. Oncol. 28, 3061–3068 (2010).
Lewis, P.W. et al. Inhibition of PRC2 activity by a gain-of-function H3 H3 mutation found in pediatric glioblastoma. Science 340, 857–861 (2013).
Shen, Q. et al. The fibrodysplasia ossificans progressiva R206H ACVR1 mutation activates BMP-independent chondrogenesis and zebrafish embryo ventralization. J. Clin. Invest. 119, 3462–3472 (2009).
Lahti, L., Peltopuro, P., Piepponen, T.P. & Partanen, J. Cell-autonomous FGF signaling regulates anteroposterior patterning and neuronal differentiation in the mesodiencephalic dopaminergic progenitor domain. Development 139, 894–905 (2012).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
McKenna, A. et al. The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 20, 1297–1303 (2010).
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Wang, K., Li, M. & Hakonarson, H. ANNOVAR: functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res. 38, e164 (2010).
McPherson, A. et al. deFuse: an algorithm for gene fusion discovery in tumor RNA-Seq data. PLoS Comput. Biol. 7, e1001138 (2011).
Horn, S. et al. TERT promoter mutations in familial and sporadic melanoma. Science 339, 959–961 (2013).
Maksimovic, J., Gordon, L. & Oshlack, A. SWAN: subset-quantile within array normalization for Illumina Infinium HumanMethylation450 BeadChips. Genome Biol. 13, R44 (2012).
Suzuki, R. & Shimodaira, H. Pvclust: an R package for assessing the uncertainty in hierarchical clustering. Bioinformatics 22, 1540–1542 (2006).
Monje, M. et al. Hedgehog-responsive candidate cell of origin for diffuse intrinsic pontine glioma. Proc. Natl. Acad. Sci. USA 108, 4453–4458 (2011).
Siegel, P.M., Ryan, E.D., Cardiff, R.D. & Muller, W.J. Elevated expression of activated forms of Neu/ErbB-2 and ErbB-3 are involved in the induction of mammary tumors in transgenic mice: implications for human breast cancer. EMBO J. 18, 2149–2164 (1999).
Schmierer, B. & Hill, C.S. TGFβ-SMAD signal transduction: molecular specificity and functional flexibility. Nat. Rev. Mol. Cell Biol. 8, 970–982 (2007).
Kleinman, C.L. et al. Fusion of TTYH1 with the C19MC microRNA cluster drives expression of a brain-specific DNMT3B isoform in the embryonal brain tumor ETMR. Nat. Genet. 46, 39–44 (2014).
Ramkissoon, L.A. et al. Genomic analysis of diffuse pediatric low-grade gliomas identifies recurrent oncogenic truncating rearrangements in the transcription factor MYBL1. Proc. Natl. Acad. Sci. USA 110, 8188–8193 (2013).
Firestein, R. et al. CDK8 is a colorectal cancer oncogene that regulates β-catenin activity. Nature 455, 547–551 (2008).
Acknowledgements
The authors would like to express their sincere gratitude toward all staff at the McGill University and Génome Québec Innovation Centre for excellent technical expertise, library preparation and sequencing. The authors are very grateful to J.-J. Lebrun (McGill University) for primer sequences and materials for SMAD signaling studies. This work was performed within the context of the I-CHANGE (International Childhood Astrocytoma Integrated Genomics and Epigenomics) Consortium and was supported by funding from Genome Canada, Génome Québec, the Institute for Cancer Research of the Canadian Institutes for Health Research (CIHR), McGill University and the Montreal Children's Hospital Foundation. This work was also supported by Hungarian Scientific Research Fund (OTKA) contract T-04639, National Research and Development Fund (NKFP) contract 1A/002/2004 (P.H. and M.G.) and TÁMOP-4.2.2A-11/1/KONV-2012-0025 (A.K. and L.B.). N.J. is a member of the Penny Cole laboratory and the recipient of a Chercheur Clinicien Senior Award. J. Majewski holds a Canada Research Chair (tier 2). L.G., K.L.L. and M.W.K. are supported by NCI P01CA142536. We acknowledge the support of the Zach Carson DIPG Fund at the Dana-Farber Cancer Institute (DFCI), the Ellie Kavalieros Fund (DFCI), the Mikey Czech Foundation, the Prayer From Maria Foundation, the Hope for Caroline Fund (DFCI), the Ryan Harvey DIPG Fund (DFCI), the Stop&Shop Pediatric Brain Tumor Program (DFCI) and the Pediatric Brain Tumor Clinical and Research Fund (DFCI). A.M.F. is supported by a studentship from CIHR, as well as by an award from the CIHR Systems Biology Training Program at McGill University. D.B. is supported by a studentship from the T.D. Trust/Montreal Children's Hospital Foundation, and N. Gerges is supported by a studentship from the Cedars Cancer Institute. N.D.J. is supported by an award from the McGill Integrated Cancer Research Training Program.
Author information
Authors and Affiliations
Contributions
A.M.F., N. Gerges, P.-O.F., D.B., D.F., L.A.R., A.C., A.H.L., S.A. and Z.D. performed experiments. A.M.F., S.P.-C., J.S., H.N., N.D.J., A.H.L., S.A., Z.D. and P.M.S. analyzed the data and produced figures and tables. D.T.W.J., D.S., P.J., T.T., S.G., M.N., A.B., L.G., D.C.B., J.R.L., J.B.R., T.A., S.B., J.R.G., G.J., K.C., N. Gupta, M.D.P., A.-S.C., B.E., L.C., A.K., L.B., P.H., M.G., J. Myseros, H.M., S.A. and S.M.P. provided tissue samples. K.L.L., J. Majewski, N.J. and M.W.K. provided project leadership and designed the study. All authors contributed to the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Patterns of K27M-associated mutations among midline pediatric high-grade astrocytomas.
a, Neuroanatomical distribution of mutations and alterations of interest in pediatric midline high-grade astrocytomas. b, The age of patients harboring H3.3 and H3.1 mutations is significantly different (P = 0.0280; two-tailed t test). c, Mutual exclusivity and associative statistical analyses (represented as P values) of mutations in H3F3A, HIST1H3B/HIST1H3C, TP53, FGFR1, ACVR1, PDGFRA and PI3K pathway genes PIK3CA/PIK3R1/PTEN demonstrates predilection for co-occurrence of and exclusion of particular mutations as unique mutational groups within 39 midline high-grade astrocytomas profiled by whole-exome sequencing. P values were calculated utilizing Fisher's exact test (two-sided) with highlighted values indicating statistically significant (P < 0.05) preferences for co-occurrence or mutual exclusivity.
Supplementary Figure 2 PDGFRA amplification is present in a minority of cells in DIPG.
Representative images from FISH assay for PDGFRA amplification reveal substantial increase in the number of PDFRA-positive nuclei following treatment compared with samples from pretreatment biopsies. Green, PDGFRA; red, Chr4/CEN4.
Supplementary Figure 3 Copy number variant analysis using biopsy material from five different anatomical loci in a single tumor.
a, Tumor biopsies (n = 5) show highly similar CNV patterns across the genome. b, Plots depicting 20-Mb zoom-in of chromosome 4, showing significant PDGFRA, KIT and KDR amplification in only the deep tissue biopsy and not others.
Supplementary Figure 4 H3.1 and H3.3 K27M variants demonstrate similar patterns of global DNA methylation.
Heat map of hierarchical clustering analysis of DNA methylation array data from the top 10,000 most variable β values from 27 K27M-mutant high-grade gliomas demonstrates that H3.3 and H3.1 K27M mutant tumors show similar patterns of global epigenomic dysregulation.
Supplementary Figure 5 Multiscale bootstrapping of the high-grade astrocytoma methylation cluster.
Dendrogram of multiscale bootstrapping of DNA methylation profiles of patient high-grade tumors. P values, represented as the red values at the internal nodes, are the approximately unbiased (AU) P value computed by the R package pvclust.
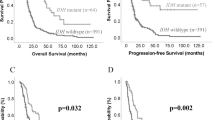
Supplementary Figure 6 Mutation subgroup-specific overall survival of midline high-grade astrocytomas (mHGAs) and diffuse intrinsic pontine gliomas (DIPGs).
Kaplan-Meier analysis of overall survival (months) of mHGAs with available outcome data, with indicated P values from Mantel-Cox (log-rank) and Gehan-Breslow-Wilcoxon testing for comparison based on a, ACVR1 mutation status (n = 27), b, H3.3/H3.1 K27M mutation status (n = 27) and overall survival restricted to tumors of the pontine area based on c, ACVR1 mutation status (n = 23), d, H3.3/H3.1 K27M mutation status (n = 22).
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 (PDF 2686 kb)
Supplementary Tables 1–8
Supplementary Tables 1–8 (XLSX 130 kb)
Rights and permissions
About this article
Cite this article
Fontebasso, A., Papillon-Cavanagh, S., Schwartzentruber, J. et al. Recurrent somatic mutations in ACVR1 in pediatric midline high-grade astrocytoma. Nat Genet 46, 462–466 (2014). https://doi.org/10.1038/ng.2950
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.2950