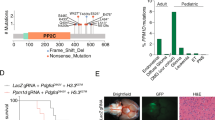
Abstract
Diffuse intrinsic pontine glioma (DIPG) is a fatal brain cancer that arises in the brainstem of children, with no effective treatment and near 100% fatality. The failure of most therapies can be attributed to the delicate location of these tumors and to the selection of therapies on the basis of assumptions that DIPGs are molecularly similar to adult disease. Recent studies have unraveled the unique genetic makeup of this brain cancer, with nearly 80% found to harbor a p.Lys27Met histone H3.3 or p.Lys27Met histone H3.1 alteration. However, DIPGs are still thought of as one disease, with limited understanding of the genetic drivers of these tumors. To understand what drives DIPGs, we integrated whole-genome sequencing with methylation, expression and copy number profiling, discovering that DIPGs comprise three molecularly distinct subgroups (H3-K27M, silent and MYCN) and uncovering a new recurrent activating mutation affecting the activin receptor gene ACVR1 in 20% of DIPGs. Mutations in ACVR1 were constitutively activating, leading to SMAD phosphorylation and increased expression of the downstream activin signaling targets ID1 and ID2. Our results highlight distinct molecular subgroups and novel therapeutic targets for this incurable pediatric cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Stiller, C.A. Population based survival rates for childhood cancer in Britain, 1980–1991. Br. Med. J. 309, 1612–1616 (1994).
Freeman, C.R. & Perilongo, G. Chemotherapy for brain stem gliomas. Childs Nerv. Syst. 15, 545–553 (1999).
Maria, B.L. et al. Brainstem glioma: I. Pathology, clinical features, and therapy. J. Child Neurol. 8, 112–128 (1993).
Hargrave, D., Bartels, U. & Bouffet, E. Diffuse brainstem glioma in children: critical review of clinical trials. Lancet Oncol. 7, 241–248 (2006).
Donaldson, S.S., Laningham, F. & Fisher, P.G. Advances toward an understanding of brainstem gliomas. J. Clin. Oncol. 24, 1266–1272 (2006).
Buczkowicz, P. et al. Aurora kinase B is a potential therapeutic target in pediatric diffuse intrinsic pontine glioma. Brain Pathol. 23, 244–253 (2013).
Paugh, B.S. et al. Genome-wide analyses identify recurrent amplifications of receptor tyrosine kinases and cell-cycle regulatory genes in diffuse intrinsic pontine glioma. J. Clin. Oncol. 29, 3999–4006 (2011).
Paugh, B.S. et al. Integrated molecular genetic profiling of pediatric high-grade gliomas reveals key differences with the adult disease. J. Clin. Oncol. 28, 3061–3068 (2010).
Zarghooni, M. et al. Whole-genome profiling of pediatric diffuse intrinsic pontine gliomas highlights platelet-derived growth factor receptor α and poly (ADP-ribose) polymerase as potential therapeutic targets. J. Clin. Oncol. 28, 1337–1344 (2010).
Khuong-Quang, D.A. et al. K27M mutation in histone H3.3 defines clinically and biologically distinct subgroups of pediatric diffuse intrinsic pontine gliomas. Acta Neuropathol. 124, 439–447 (2012).
Schwartzentruber, J. et al. Driver mutations in histone H3.3 and chromatin remodelling genes in paediatric glioblastoma. Nature 482, 226–231 (2012).
Wu, G. et al. Somatic histone H3 alterations in pediatric diffuse intrinsic pontine gliomas and non-brainstem glioblastomas. Nat. Genet. 44, 251–253 (2012).
Herbert, B.S., Shay, J.W. & Wright, W.E. Analysis of telomeres and telomerase. Curr. Protoc. Cell Biol. Chapter 18, Unit 18.6 (2003).
Henson, J.D. et al. DNA C-circles are specific and quantifiable markers of alternative-lengthening-of-telomeres activity. Nat. Biotechnol. 27, 1181–1185 (2009).
Song, G.A. et al. Molecular consequences of the ACVR1R206H mutation of fibrodysplasia ossificans progressiva. J. Biol. Chem. 285, 22542–22553 (2010).
Kaplan, F.S. et al. Classic and atypical fibrodysplasia ossificans progressiva (FOP) phenotypes are caused by mutations in the bone morphogenetic protein (BMP) type I receptor ACVR1. Hum. Mutat. 30, 379–390 (2009).
Petrie, K.A. et al. Novel mutations in ACVR1 result in atypical features in two fibrodysplasia ossificans progressiva patients. PLoS ONE 4, e5005 (2009).
Kowanetz, M., Valcourt, U., Bergstrom, R., Heldin, C.H. & Moustakas, A. Id2 and Id3 define the potency of cell proliferation and differentiation responses to transforming growth factor β and bone morphogenetic protein. Mol. Cell. Biol. 24, 4241–4254 (2004).
Kurooka, H., Nakahiro, T., Mori, K., Sano, K. & Yokota, Y. BMP signaling is responsible for serum-induced Id2 expression. Biochem. Biophys. Res. Commun. 420, 281–287 (2012).
Shin, M. et al. Identification of a novel bone morphogenetic protein (BMP)-inducible transcript, BMP-inducible transcript-1, by utilizing the conserved BMP-responsive elements in the Id genes. J. Bone Miner. Metab. 31, 34–43 (2013).
Chen, X.S., Zhang, Y.H., Cai, Q.Y. & Yao, Z.X. ID2: a negative transcription factor regulating oligodendroglia differentiation. J. Neurosci. Res. 90, 925–932 (2012).
Liu, Z. et al. Overexpressed DNA-binding protein inhibitor 2 as an unfavorable prognosis factor promotes cell proliferation in nasopharyngeal carcinoma. Acta Biochim. Biophys. Sin. (Shanghai) 44, 503–512 (2012).
Rollin, J. et al. The intracellular localization of ID2 expression has a predictive value in non small cell lung cancer. PLoS ONE 4, e4158 (2009).
Wazir, U., Jiang, W.G., Sharma, A.K., Newbold, R.F. & Mokbel, K. The mRNA expression of inhibitors of DNA binding-1 and -2 is associated with advanced tumour stage and adverse clinical outcome in human breast cancer. Anticancer Res. 33, 2179–2183 (2013).
Li, H. & Durbin, R. Fast and accurate long-read alignment with Burrows-Wheeler transform. Bioinformatics 26, 589–595 (2010).
Jiang, Y., Wang, Y. & Brudno, M. PRISM: pair-read informed split-read mapping for base-pair level detection of insertion, deletion and structural variants. Bioinformatics 28, 2576–2583 (2012).
Mills, R.E. et al. Natural genetic variation caused by small insertions and deletions in the human genome. Genome Res. 21, 830–839 (2011).
Fiume, M. et al. Savant Genome Browser 2: visualization and analysis for population-scale genomics. Nucleic Acids Res. 40, W615–W621 (2012).
Medvedev, P., Fiume, M., Dzamba, M., Smith, T. & Brudno, M. Detecting copy number variation with mated short reads. Genome Res. 20, 1613–1622 (2010).
Sasai, K. et al. O6-methylguanine-DNA methyltransferase is downregulated in transformed astrocyte cells: implications for anti-glioma therapies. Mol. Cancer 6, 36 (2007).
Bland, J.M. & Altman, D.G. Statistics notes. The odds ratio. Br. Med. J. 320, 1468 (2000).
Acknowledgements
We would like to thank all of the patients and families for donating tissue for this research. This work was supported by the Canadian Institutes of Health Research (CIHR, MOP 115004) and was funded in part by a Genome Canada/CIHR grant (cofunding from Genome BC, Génome Québec, CIHR-ICR (Institute for Cancer Research) and C17, through the Genome Canada/CIHR joint ATID Competition (project title: The Canadian Paediatric Cancer Genome Consortium (CPCGC): Translating Next-Generation Sequencing Technologies into Improved Therapies for High-Risk Childhood Cancer)). P.B. is a recipient of a CIHR Doctoral Frederick Banting and Charles Best Canada Graduate Scholarships award. O.B. is a Damon Runyon Clinical Investigator and is supported by the US Department of Defense and the Pediatric Brain Tumor Foundation. C.J., A. Mackay and K.R.T. acknowledge National Health Service (NHS) funding to the Biomedical Research Centre and support from the Stavros Niarchos Foundation. Sample collection for M.A.K. and D.Z. was supported in part by grant UL1TR000038 from the National Center for Research Resources, US National Institutes of Health and by grant 5P30CA016087-32 from the National Cancer Institute.
Author information
Authors and Affiliations
Contributions
E.B., U.B., P.B., O.B. and C. Hawkins designed the study. P.B., C. Hoeman, F.C., P.R., S.P., A. Morrison, J.Z., S.A., S.R., M. Barszczyk, Y.C., P.C.-B., K.C.H. and J. Mangerel performed experiments. P.B., C. Hoeman, F.C., P.R., L.L., M.D., M. Bourgey, G.B. and A. Montpetit collected and analyzed data. O.B., C. Hawkins, C.J., K.R.T., A. Mackay, A.E.B., J.N., J.R.F., M.A.K., D.Z., N.K.F., A.D., J.V.H., A.S., J. Chan, L.L.-C., S.D., J.H., C.D., K.S., J. Michaud, S.Z., D.R., J. Cain, M.M.S., E.B., U.T. and U.B. provided reagents, tissue and mice. P.B., P.R., S.P., M.D., O.B. and C. Hawkins wrote the manuscript. P.L., C.B., C.D.A., M. Brudno, A.H. and U.T. gave technical support and conceptual advice. All authors approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 MYCN subgroup tumors show recurrent structural variants involving high-level amplification and rearrangement of MYCN, ID2 and KIDINS220 on chromosome 2p.
SNP6.0 copy number profiles of chromosome 2p focal, high-level amplifications in (a) DIPG38, (b) DIPG49, (c) DIPG01 and (d) DIPG29. These amplifications always involve the genes MYCN, ID2 and KIDINS220. Circos plots of structural variants in (e) DIPG01 and (f) DIPG29 as determined from WGS data.
Supplementary Figure 2 Event graph and Circos plot of chromothripsis in DIPG29.
(a) The bidirected event graph for a chromothripsis event in DIPG29. Red edges represent the genomic interval of their respective nodes. Blue edges represent groups of discordant read pairs supporting the same breakpoint. Arc width is proportional to the maximal likely copy count. (b) A Circos plot of chromosomes 1 and 2 from MYCN group patient DIPG29. Only those clusters larger than 1,000 bp are shown between chr. 1 and chr. 2. The width of each arc is proportional to the log of its estimated copy count. The highest estimated copy count is 61.
Supplementary Figure 3 Structural variant validation in DIPG29.
(a) Structural variant spanning six breakpoints as predicted by discordant read-pair mappings in DIPG29. (b) PCR validation of DDX1-KIDINS220 (left primer, TCTATGCCAGTGCTTTACTCCTT; right primer, CTGTTCCACCAAAGCCAAAT) and DDX1-MYCN (left primer, TGAGCAGATTTTCTGTATATTTTCCA; right primer, GTCTCCCAGGCTGCAGTG) in tumour and matched normal show product band only in tumor DNA. (c) Sanger sequencing through breakpoints in DDX1-KIDINS220 and DDX1-MYCN structural variants.
Supplementary Figure 4 DIPG patients with ALT phenotype have longer telomeres and are diagnosed at an older age.
All ALT-positive DIPG patients were found in the H3-K27M subgroup. By WGS, these patients had significantly longer telomeres; (a) 2.28 times longer than their matched normal vs. non-ALT DIPG patients who had telomeres that were 1.47 times shorter than their matched normal (P < 0.0001). (b) There was a significant difference in age of diagnosis between ALT-negative (5.89 ± 2.82 years) vs. ALT-positive (10.08 ± 3.61 years) patients; P <0.0001). Error bars represent s.e.m.
Supplementary Figure 5 H3-H27M DIPG tumors exhibit structural variants in the PDGFRA and PVT1/MYC loci.
H3-K27M DIPG patients often show gains and amplifications as well as structural variants in (a) PDGFRA and (b) PVT1/MYC.
Supplementary Figure 6 K27M-H3.3 tumors exhibit a global decrease in H3K27me3 levels compared to WT-H3.3 in vitro and in vivo.
(a) Protein blot of Flag-tagged WT-H3.3 and K27M-H3.3 reveals both clones expressing similar levels of protein. No expression was detected in untransfected NHA and NHA cells transfected with empty vector control. (b) Immunofluorescence staining of NHA cells shows nuclear localization of Flag-tagged WT-H3.3 and K27M-H3.3 protein. (c) K27M-H3.3–expressing NHA cells have a decreased growth rate compared to both WT-H3.3 and empty vector control (P < 0.0001). (d) By western blot, H3K27me3 levels are decreased in K27M-H3.3 NHA cells compared to WT-H3.3 and empty vector NHA cells by 52% (P = 0.01). (e) Immortalized NHAs transfected with K27M-H3.3 show phenotypic changes compared to empty vector and WT-H3.3 controls, forming cell clusters at high density when seeded in DMEM and growing semi-adherently in neural stem cell medium. (f) K27M-H3.3 NHAs have different methylation and expression profiles than controls. The association of decreased H3K27me3 levels and mutant histone H3 is also seen by immunohistochemical staining of DIPG tissue microarray, where patients with K27M-H3.3 (g) show decreased H3K27me3 levels. DIPG patients that are WT-H3.3 (h) show more positive staining by immunohistochemistry for H3K27me3.
Supplementary Figure 7 K27M-H3.3 semi-adherent cells exhibit higher SOX2 expression.
Immunofluorescence staining of empty vector, K27M-H3.3 adherent and K27M-H3.3 semi-adherent iNHA cells reveals increased SOX2 expression in the semi-adherent cells but no changes in GFAP, nestin, TUJ1 or O4 levels. Images were taken at 400× magnification.
Supplementary Figure 8 Discordant read-pair clustering.
(a) Schematic of discordant read pairs supporting a translocation event (1) and clipped mappings narrowing in breakpoint location (2). (b) Read pairs as viewed by translocated region aligned to tumor genome. (c) The spanning reads in the normal sample provide an expected arrival count for the Poisson distribution when used to determine maximum-likelihood copy counts.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 and Supplementary Tables 5, 6 and 8 (PDF 1296 kb)
Supplementary Tables
Supplementary Tables 1–4 and 7 (XLSX 2202 kb)
Rights and permissions
About this article
Cite this article
Buczkowicz, P., Hoeman, C., Rakopoulos, P. et al. Genomic analysis of diffuse intrinsic pontine gliomas identifies three molecular subgroups and recurrent activating ACVR1 mutations. Nat Genet 46, 451–456 (2014). https://doi.org/10.1038/ng.2936
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.2936
This article is cited by
-
Current and future therapeutic strategies for high-grade gliomas leveraging the interplay between epigenetic regulators and kinase signaling networks
Journal of Experimental & Clinical Cancer Research (2024)
-
PRMT5 inhibition shows in vitro efficacy against H3K27M-altered diffuse midline glioma, but does not extend survival in vivo
Scientific Reports (2024)
-
TP-0184 inhibits FLT3/ACVR1 to overcome FLT3 inhibitor resistance and hinder AML growth synergistically with venetoclax
Leukemia (2024)
-
MYCN immunohistochemistry as surrogate marker for MYCN-amplified spinal ependymomas
Human Cell (2024)
-
BMP signaling in cancer stemness and differentiation
Cell Regeneration (2023)