Abstract
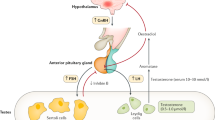
Late-onset hypogonadism (formerly called the andropause) is a clinical and biochemical syndrome associated with advancing age, which is characterized by typical signs and symptoms and a deficiency in serum testosterone levels. Age-related hypoandrogenism in the male is a result of the interaction of hypothalamopituitary and testicular factors. The hypothalamic pulsatile secretion of gonadotropin-releasing hormone is blunted, due to increased hypothalamic sensitivity to inhibition by steroids, but the responsiveness of the pituitary gonadotrophs seems to be intact. In addition, testicular volume as well as Leydig cell mass and reserve function are diminished. Taken together, these mechanisms result in reduced testosterone secretion and the loss of nycthemeral variability.
Key Points
-
Late-onset hypogonadism (LOH) results from the interaction of hypothalamic, pituitary and testicular factors
-
In men with LOH, the hypothalamic pulsatile secretion of gonadotropin-releasing hormone is reduced
-
Testicular Leydig cell mass and reserve function are diminished in men with LOH
-
Underlying endocrine mechanisms result in reduced testosterone secretion and the loss of nycthemeral variability
-
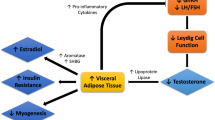
Many systemic, pathologic conditions are epidemiologically linked to LOH; whether these are a cause or a result of LOH is unclear, and in some cases the relationship may be bidirectional (e.g. obesity)
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Nieschlag E et al. (2005) Investigation, treatment and monitoring of late-onset hypogonadism in males: ISA, ISSAM, and EAU recommendations. Int J Androl 28: 125–127
Comhaire F and Vermeulen A (1975) Plasma testosterone in patients with varicocele and sexual inadequacy. J Clin Endocrinol Metab 40: 824–829
Canales BK et al. (2005) Prevalence and effect of varicoceles in an elderly population. Urology 66: 627–631
Vermeulen A et al. (1996) Influence of some biological indexes on sex hormone-binding globulin and androgen levels in aging or obese males. J Clin Endocrinol Metab 81: 1821–1826
Tsai EC et al. (2000) Low serum testosterone level as a predictor of increased visceral fat in Japanese-American men. Int J Obes Relat Metab Disord 24: 485–491
T'Sjoen G et al. (2003) Self-referred patients in an aging male clinic: much more than androgen deficiency alone. Aging Male 6: 157–165
Comhaire FH (2000) Andropause: hormone replacement therapy in the ageing male. Eur Urol 38: 655–662
Isom-Batz G et al. (2005) Testosterone as a predictor of pathological stage in clinically localized prostate cancer. J Urol 173: 1935–1937
Imamoto T et al. (2005) Pretreatment serum testosterone level as a predictive factor of pathological stage in localized prostate cancer patients treated with radical prostatectomy. Eur Urol 47: 308–312
Feneley MR and Carruthers M (2005) PSA monitoring during testosterone replacement therapy: low long-term risk of prostate cancer with improved opportunity for cure. In Proceedings of the 4th international workshop on molecular andrology: 2005 October 7–9; Giessen, Germany. Andrologia 37: 249
Rhoden EL and Morgentaler A (2003) Testosterone replacement therapy in hypogonadal men at high risk for prostate cancer: results of 1 year of treatment in men with prostatic intraepithelial neoplasia. J Urol 170: 2348–2351
Kaufman J (2006) A rational approach to androgen therapy for hypogonadal men with prostate cancer. Int J Impot Res 18: 26–31
Hammond GL et al. (1979) Serum steroids and pituitary hormones in infants with particular reference to testicular activity. J Clin Endocrinol Metab 49: 40–45
Dufau ML (1988) Endocrine regulation and communicating functions of the Leydig cell. Annu Rev Physiol 50: 483–508
Schally AV et al. (1971) Isolation and properties of the FSH and LH-releasing hormone. Biochem Biophys Res Commun 43: 393–399
Crowley WF Jr et al. (1991) Neuroendocrine control of human reproduction in the male. Recent Prog Horm Res 47: 27–62
Knobil E (1990) The GnRH pulse generator. Am J Obstet Gynecol 163: 1721–1727
Kaufman JM et al. (1985) Electrophysiological manifestation of luteinizing hormone-releasing hormone pulse generator activity in the rhesus monkey: influence of alpha-adrenergic and dopaminergic blocking agents. Endocrinology 116: 1327–1333
Knobil E (1980) The neuroendocrine control of the menstrual cycle. Recent Prog Horm Res 36: 53–88
Marshall JC and Kelch RP (1986) Gonadotropin-releasing hormone: role of pulsatile secretion in the regulation of reproduction. N Engl J Med 315: 1459–1468
Plant TM and Dubey AK (1984) Evidence from the rhesus monkey (Macaca mulatta) for the view that negative feedback control of luteinizing hormone secretion by the testis is mediated by a deceleration of hypothalamic gonadotropin-releasing hormone pulse frequency. Endocrinology 115: 2145–2153
Matsumoto AM and Bremner WJ (1984) Modulation of pulsatile gonadotropin secretion by testosterone in man. J Clin Endocrinol Metab 58: 609–614
Opstad PK (1992) Androgenic hormones during prolonged physical stress, sleep, and energy deficiency. J Clin Endocrinol Metab 74: 1176–1183
MacConnie SE et al. (1986) Decreased hypothalamic gonadotropin-releasing hormone secretion in male marathon runners. N Engl J Med 315: 411–417
Aitken RJ et al. (1985) Relationship between the movement characteristics of human spermatozoa and their ability to penetrate cervical mucus and zona-free hamster oocytes. J Reprod Fertil 73: 441–449
Warren MP (1983) Effects of undernutrition on reproductive function in the human. Endocr Rev 4: 363–377
Vescovi PP et al. (1992) Chronic effects of marihuana smoking on luteinizing hormone, follicle-stimulating hormone and prolactin levels in human males. Drug Alcohol Depend 30: 59–63
Kesner JS et al. (1986) The effect of morphine on the electrophysiological activity of the hypothalamic luteinizing hormone-releasing hormone pulse generator in the rhesus monkey. Neuroendocrinology 43: 686–688
Veldhuis JD et al. (1984) Role of endogenous opiates in the expression of negative feedback actions of androgen and estrogen on pulsatile properties of luteinizing hormone secretion in man. J Clin Invest 74: 47–55
Dudas B and Merchenthaler I (2006) Three-dimensional representation of the neurotransmitter systems of the human hypothalamus: inputs of the gonadotrophin hormone-releasing hormone neuronal system. J Neuroendocrinol 18: 79–95
Clayton RN (1989) Gonadotrophin-releasing hormone: its actions and receptors. J Endocrinol 120: 11–19
Schurmeyer T et al. (1984) Suppression of pituitary and testicular function in normal men by constant gonadotropin-releasing hormone agonist infusion. J Clin Endocrinol Metab 59: 19–24
Matsumoto AM et al. (1991) The physiological significance of pulsatile LHRH secretion in man: gonadotrophin responses to physiological doses of pulsatile versus continuous LHRH administration. Int J Androl 14: 23–32
Conn PM and Crowley WF Jr (1991) Gonadotropin-releasing hormone and its analogs. N Engl J Med 324: 93–103
Belchetz PE et al. (1978) Hypophysial responses to continuous and intermittent delivery of hypothalamic gonadotropin-releasing hormone. Science 202: 631–633
Nilsson B et al. (1986) Differences in the carbohydrate moieties of the common alpha-subunits of human chorionic gonadotropin, luteinizing hormone, follicle-stimulating hormone, and thyrotropin: preliminary structural inferences from direct methylation analysis. Endocrinology 119: 2737–2143
Spratt DI et al. (1988) Neuroendocrine–gonadal axis in men: frequent sampling of LH, FSH, and testosterone. Am J Physiol 254: E658–E666
Boyar R et al. (1972) Synchronization of augmented luteinizing hormone secretion with sleep during puberty. N Engl J Med 287: 582–586
Fehm HL et al. (1991) Sleep-associated augmentation and synchronization of luteinizing hormone pulses in adult men. Neuroendocrinology 54: 192–195
Veldhuis JD et al. (1987) Operating characteristics of the male hypothalamo–pituitary–gonadal axis: pulsatile release of testosterone and follicle-stimulating hormone and their temporal coupling with luteinizing hormone. J Clin Endocrinol Metab 65: 929–941
Winters SJ and Troen P (1986) Testosterone and estradiol are co-secreted episodically by the human testis. J Clin Invest 78: 870–873
Comhaire FH and Vermeulen A (1976) Testosterone concentration in the fluids of seminiferous tubules, the interstitium and the rete testis of the rat. J Endocrinol 70: 229–235
Chase DJ et al. (1992) Maintenance or stimulation of steroidogenic enzymes and testosterone production in rat Leydig cells by continuous and pulsatile infusions of luteinizing hormone during passive immunization against gonadotrophin-releasing hormone. J Reprod Fertil 95: 657–667
Gibson-Berry KL and Chase DJ (1990) Continuous and pulsatile infusions of luteinizing hormone have identical effects on steroidogenic capacity and sensitivity of Leydig cells in rats passively immunized against gonadotropin-releasing hormone. Endocrinology 126: 3107–3115
Bridges NA et al. (1993) The relationship between endogenous testosterone and gonadotrophin secretion. Clin Endocrinol (Oxf) 38: 373–378
Santen RJ (1975) Is aromatization of testosterone to estradiol required for inhibition of luteinizing hormone secretion in men? J Clin Invest 56: 1555–1563
Winters SJ and Troen P (1985) Evidence for a role of endogenous estrogen in the hypothalamic control of gonadotropin secretion in men. J Clin Endocrinol Metab 61: 842–845
Canovatchel WJ et al. (1994) Luteinizing hormone pulsatility in subjects with 5-alpha-reductase deficiency and decreased dihydrotestosterone production. J Clin Endocrinol Metab 78: 916–921
Veldhuis JD et al. (1989) Random and non-random coincidence between luteinizing hormone peaks and follicle-stimulating hormone, alpha subunit, prolactin, and gonadotropin-releasing hormone pulsations. J Neuroendocrinol 1: 185–194
Comhaire FH et al. (1995) Inhibin and steroid responses to testicular stimulation in normal men. Hum Reprod 10: 1740–1744
Anderson RA and Sharpe RM (2000) Regulation of inhibin production in the human male and its clinical applications. Int J Androl 23: 136–144
Ying SY (1988) Inhibins, activins, and follistatins: gonadal proteins modulating the secretion of follicle-stimulating hormone. Endocr Rev 9: 267–293
Hancock AD et al. (1992) Inhibin and inhibin alpha-chain precursors are produced by immature rat Sertoli cells in culture. Biol Reprod 46: 155–161
Tuohimaa P et al. (2000) Inhibin and activin subunits and spermatogenesis. Andrologia 32: 48–52
Bame JH et al. (1999) Effect of long-term immunization against inhibin on sperm output in bulls. Biol Reprod 60: 1360–1366
Hayes FJ et al. (2001) Importance of inhibin B in the regulation of FSH secretion in the human male. J Clin Endocrinol Metab 86: 5541–5546
Hayes FJ et al. (2001) Differential regulation of gonadotropin secretion by testosterone in the human male: absence of a negative feedback effect of testosterone on follicle-stimulating hormone secretion. J Clin Endocrinol Metab 86: 53–58
Bartke A (1977) Prolactin and the physiological regulation of the mammalian testis. In: The testis in normal and infertile men. New York: Raven Press
Winters SJ and Troen P (1984) Altered pulsatile secretion of luteinizing hormone in hypogonadal men with hyperprolactinaemia. Clin Endocrinol (Oxf) 21: 257–263
Magrini B et al. (1976) Study on the relationship between plasma prolactin levels and androgen metabolism in man. J Clin Endocrinol Metab 43: 944–947
Hair WM et al. (2002) Prolactin receptor expression in human testis and accessory tissues: localization and function. Mol Hum Reprod 8: 606–611
Luboshitzky R et al. (2000) Long-term melatonin administration does not alter pituitary–gonadal hormone secretion in normal men. Hum Reprod 15: 60–65
Deslypere JP and Vermeulen A (1984) Leydig cell function in normal men: effect of age, life-style, residence, diet, and activity. J Clin Endocrinol Metab 59: 955–962
Kaufman JM and Vermeulen A (2005) The decline of androgen levels in elderly men and its clinical and therapeutic implications. Endocr Rev 26: 833–876
Ferrini RL and Barrett-Connor E (1998) Sex hormones and age: a cross-sectional study of testosterone and estradiol and their bioavailable fractions in community-dwelling men. Am J Epidemiol 147: 750–754
Morley JE et al. (1997) Longitudinal changes in testosterone, luteinizing hormone, and follicle-stimulating hormone in healthy older men. Metabolism 46: 410–413
Mahmoud AM et al. (2003) Testicular volume in relation to hormonal indices of gonadal function in community-dwelling elderly men. J Clin Endocrinol Metab 88: 179–184
Vermeulen A (2005) Hormonal cut-offs of partial androgen deficiency: a survey of androgen assays. J Endocrinol Invest 28: 28–31
Snyder PJ et al. (1999) Effect of testosterone treatment on bone mineral density in men over 65 years of age. J Clin Endocrinol Metab 84: 1966–1972
O'Donnell AB et al. (2006) Testosterone, dehydroepiandrosterone, and physical performance in older men: results from the Massachusetts Male Aging Study. J Clin Endocrinol Metab 91: 425–431
Veldhuis JD et al. (2005) Mechanisms of ensemble failure of the male gonadal axis in aging. J Endocrinol Invest 28: 8–13
Neaves WB et al. (1984) Leydig cell numbers, daily sperm production, and serum gonadotropin levels in aging men. J Clin Endocrinol Metab 59: 756–763
Longcope C (1973) The effect of human chorionic gonadotropin on plasma steroid levels in young and old men. Steroids 21: 583–592
Mulligan T et al. (1999) Two-week pulsatile gonadotropin releasing hormone infusion unmasks dual (hypothalamic and Leydig cell) defects in the healthy aging male gonadotropic axis. Eur J Endocrinol 141: 257–266
Mulligan T et al. (2001) Pulsatile iv infusion of recombinant human LH in leuprolide-suppressed men unmasks impoverished Leydig-cell secretory responsiveness to midphysiological LH drive in the aging male. J Clin Endocrinol Metab 86: 5547–5553
Veldhuis JD et al. (2005) Age diminishes the testicular steroidogenic response to repeated intravenous pulses of recombinant human LH during acute GnRH-receptor blockade in healthy men. Am J Physiol Endocrinol Metab 288: E775–E781
Wielgos M et al. (1998) Doppler flowmetry measurements in testicular artery of aging men [Polish]. Ginekol Pol 69: 537–540
Regadera J et al. (1985) Testis, epididymis, and spermatic cord in elderly men. Correlation of angiographic and histologic studies with systemic arteriosclerosis. Arch Pathol Lab Med 109: 663–667
Svartberg J et al. (2006) Low testosterone levels are associated with carotid atherosclerosis in men. J Intern Med 259: 576–582
Kaufman JM and Vermeulen A (2005) The decline of androgen levels in elderly men and its clinical and therapeutic implications. Endocr Rev 26: 833–876
Comhaire F and Mahmoud A (2006) Male aging: wear and tear. In: Andrology for the Clinician, Berlin and Heidelberg: Springer-Verlag
Meuleman EJH and Comhaire F (2006) Male aging: organ failure and common diseases of the ageing male. In: Andrology for the Clinician, Berlin and Heidelberg: Springer-Verlag
Derby CA et al. (2006) Body mass index, waist circumference and waist to hip ratio and change in sex steroid hormones: the Massachusetts Male Aging Study. Clinical Endocrinology 65: 125–131
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Mahmoud, A., Comhaire, F. Mechanisms of Disease: late-onset hypogonadism. Nat Rev Urol 3, 430–438 (2006). https://doi.org/10.1038/ncpuro0560
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncpuro0560
This article is cited by
-
Plasma metabolome study reveals metabolic changes induced by pharmacological castration and testosterone supplementation in healthy young men
Scientific Reports (2022)
-
The association between 2,4-D and serum testosterone levels: NHANES 2013–2014
Journal of Endocrinological Investigation (2022)