Abstract
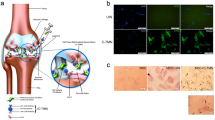
Despite the high prevalence and morbidity of osteoarthritis (OA), an effective treatment for this disease is currently lacking. Restoration of the diseased articular cartilage in patients with OA is, therefore, a challenge of considerable appeal to researchers and clinicians. Techniques that cause multipotent adult mesenchymal stem cells (MSCs) to differentiate into cells of the chondrogenic lineage have led to a variety of experimental strategies to investigate whether MSCs instead of chondrocytes can be used for the regeneration and maintenance of articular cartilage. MSC-based strategies should provide practical advantages for the patient with OA. These strategies include use of MSCs as progenitor cells to engineer cartilage implants that can be used to repair chondral and osteochondral lesions, or as trophic producers of bioactive factors to initiate endogenous regenerative activities in the OA joint. Targeted gene therapy might further enhance these activities of MSCs. Delivery of MSCs might be attained by direct intra-articular injection or by graft of engineered constructs derived from cell-seeded scaffolds; this latter approach could provide a three-dimensional construct with mechanical properties that are congruous with the weight-bearing function of the joint. Promising experimental and clinical data are beginning to emerge in support of the use of MSCs for regenerative applications.
Key Points
-
Osteoarthritis (OA), the most common joint disease, is characterized by degeneration of the articular cartilage that ultimately leads to joint destruction
-
Current treatment strategies for OA are inadequate
-
Delivery of an appropriate mesenchymal stem cell (MSC) population is currently being investigated in the search for new therapies for OA
-
MSCs could be used as trophic producers of bioactive factors to initiate endogenous regenerative activities in the OA joint; their activities might be further enhanced via targeted gene therapy
-
Delivery of MSCs might be achieved either by direct intra-articular injection or by implantation of engineered constructs derived from MSC-seeded scaffolds
-
In the long term, MSC-based technologies could permit the engineering and repair of cartilage as a treatment option for OA joints
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Elders MJ (2000) The increasing impact of arthritis on public health. J Rheumatol Suppl 60: 6–8
Brooks PM (2002) Impact of osteoarthritis on individuals and society: how much disability? Social consequences and health economic implications. Curr Opin Rheumatol 14: 573–577
Hochberg MC et al. (1995) Guidelines for the medical management of osteoarthritis. Part II. Osteoarthritis of the knee. American College of Rheumatology. Arthritis Rheum 38: 1541–1546
Gerwin N et al. (2006) Intraarticular drug delivery in osteoarthritis. Adv Drug Deliv Rev 58: 226–242
Gunther KP (2001) Surgical approaches for osteoarthritis. Best Pract Res Clin Rheumatol 15: 627–643
Moseley JB et al. (2002) A controlled trial of arthroscopic surgery for osteoarthritis of the knee. N Engl J Med 347: 81–88
Bartlett W et al. (2005) Autologous chondrocyte implantation versus matrix-induced autologous chondrocyte implantation for osteochondral defects of the knee: a prospective, randomised study. J Bone Joint Surg Br 87: 640–645
Bentley G et al. (2003) A prospective, randomised comparison of autologous chondrocyte implantation versus mosaicplasty for osteochondral defects in the knee. J Bone Joint Surg Br 85: 223–230
Hangody L and Fules P (2003) Autologous osteochondral mosaicplasty for the treatment of full-thickness defects of weight-bearing joints: ten years of experimental and clinical experience. J Bone Joint Surg Am 85A (Suppl 2): 25–32
Henderson I et al. (2005) Autologous chondrocyte implantation for treatment of focal chondral defects of the knee—a clinical, arthroscopic, MRI and histologic evaluation at 2 years. Knee 12: 209–216
Peterson L et al. (2003) Treatment of osteochondritis dissecans of the knee with autologous chondrocyte transplantation: results at two to ten years. J Bone Joint Surg Am 85A (Suppl 2): 17–24
Knutsen G et al. (2004) Autologous chondrocyte implantation compared with microfracture in the knee. A randomized trial. J Bone Joint Surg Am 86A: 455–464
Steinert AF et al. (2007) Major biological obstacles for persistent cell-based regeneration of articular cartilage. Arthritis Res Ther 9: 213
Bert JM and Gasser SI (2002) Approach to the osteoarthritic knee in the aging athlete: debridement to osteotomy. Arthroscopy 18: 107–110
Pittenger MF et al. (1999) Multilineage potential of adult human mesenchymal stem cells. Science 284: 143–147
Kolf CM et al. (2007) Mesenchymal stromal cells. Biology of adult mesenchymal stem cells: regulation of niche, self-renewal and differentiation. Arthritis Res Ther 9: 204
Chen FH et al. (2006) Technology insight: adult stem cells in cartilage regeneration and tissue engineering. Nat Clin Pract Rheumatol 2: 373–382
Aigner T et al. (2007) Mechanisms of disease: role of chondrocytes in the pathogenesis of osteoarthritis—structure, chaos and senescence. Nat Clin Pract Rheumatol 3: 391–399
Buckwalter JA et al. (2006) Perspectives on chondrocyte mechanobiology and osteoarthritis. Biorheology 43: 603–609
Martin JA et al. (2004) Chondrocyte senescence, joint loading and osteoarthritis. Clin Orthop Relat Res 427 (Suppl): S96–S103
Verbruggen G (2006) Chondroprotective drugs in degenerative joint diseases. Rheumatology (Oxford) 45: 129–138
Deschner J et al. (2003) Signal transduction by mechanical strain in chondrocytes. Curr Opin Clin Nutr. Metab Care 6: 289–293
Im GI et al. (2006) Chondrogenic differentiation of mesenchymal stem cells isolated from patients in late adulthood: the optimal conditions of growth factors. Tissue Eng 12: 527–536
Barry FP (2003) Biology and clinical applications of mesenchymal stem cells. Birth Defects Res C Embryo Today 69: 250–256
Murphy JM et al. (2002) Reduced chondrogenic and adipogenic activity of mesenchymal stem cells from patients with advanced osteoarthritis. Arthritis Rheum 46: 704–713
Muschler GF et al. (2001) Age- and gender-related changes in the cellularity of human bone marrow and the prevalence of osteoblastic progenitors. J Orthop Res 19: 117–125
Quarto R et al. (1995) Bone progenitor cell deficits and the age-associated decline in bone repair capacity. Calcif Tissue Int 56: 123–129
Leskela HV et al. (2003) Osteoblast recruitment from stem cells does not decrease by age at late adulthood. Biochem Biophys Res Commun 311: 1008–1013
De Bari C and Dell'Accio F (2007) Mesenchymal stem cells in rheumatology: a regenerative approach to joint repair. Clin Sci (Lond) 113: 339–348
Kafienah W et al. (2007) Three-dimensional cartilage tissue engineering using adult stem cells from osteoarthritis patients. Arthritis Rheum 56: 177–187
Scharstuhl A et al. (2007) Chondrogenic potential of human adult mesenchymal stem cells is independent of age or osteoarthritis etiology. Stem Cells 25: 3244–3251
Murphy JM et al. (2003) Stem cell therapy in a caprine model of osteoarthritis. Arthritis Rheum 48: 3464–3474
Lee KB et al. (2007) Injectable mesenchymal stem cell therapy for large cartilage defects––a porcine model. Stem Cells 25: 2964–2971
Chen X et al. (2006) Mesenchymal stem cells in immunoregulation. Immunol Cell Biol 84: 413–421
Uccelli A et al. (2007) Mesenchymal stem cells: a new strategy for immunosuppression. Trends Immunol 28: 219–226
Kan I et al. (2007) Autotransplantation of bone marrow-derived stem cells as a therapy for neurodegenerative diseases. Handb Exp Pharmacol 180: 219–242
Caplan AI and Dennis JE (2006) Mesenchymal stem cells as trophic mediators. J Cell Biochem 98: 1076–1084
Raghunath J et al. (2007) Biomaterials and scaffold design: key to tissue-engineering cartilage. Biotechnol Appl Biochem 46: 73–84
Li WJ et al. (2005) Application of nanofibrous scaffolds in skeletal tissue engineering. J Biomed Nanotechnol 1: 1–17
Mouw JK et al. (2005) Variations in matrix composition and GAG fine structure among scaffolds for cartilage tissue engineering. Osteoarthritis Cartilage 13: 828–836
Nöth U et al. (2002) In vitro engineered cartilage constructs produced by press-coating biodegradable polymer with human mesenchymal stem cells. Tissue Eng 8: 131–144
Li WJ et al. (2006) Chondrocyte phenotype in engineered fibrous matrix is regulated by fiber size. Tissue Eng 12: 1775–1785
Terada S et al. (2005) Hydrogel optimization for cultured elastic chondrocytes seeded onto a polyglycolic acid scaffold. J Biomed Mater Res A 75: 907–916
Kuo CK et al. (2006) Cartilage tissue engineering: its potential and uses. Curr Opin Rheumatol 18: 64–73
Nesic D et al. (2006) Cartilage tissue engineering for degenerative joint disease. Adv Drug Deliv Rev 58: 300–322
Nöth U et al. (2007) Chondrogenic differentiation of human mesenchymal stem cells in collagen type I hydrogels. J Biomed Mater Res A 83: 626–635
Nöth U et al. (2006). Matrix-based autologous chondrocyte transplantation for the treatment of large osteochondral defects. In. European Musculoskeletal Review 2006, 62–64. London: Touch Briefings
Wakitani S et al. (2004) Autologous bone marrow stromal cell transplantation for repair of full-thickness articular cartilage defects in human patellae: two case reports. Cell Transplant 13: 595–600
Kuroda R et al. (2007) Treatment of a full-thickness articular cartilage defect in the femoral condyle of an athlete with autologous bone-marrow stromal cells. Osteoarthritis Cartilage 15: 226–231
Evans CH et al. (2006) Will arthritis gene therapy become a clinical reality? Nat Clin Pract Rheumatol 2: 344–345
Evans CH et al. (2004) Osteoarthritis gene therapy. Gene Ther 11: 379–389
Robbins PD et al. (2003) Gene therapy for arthritis. Gene Ther 10: 902–911
Gouze E et al. (2007) Transgene persistence and cell turnover in the diarthrodial joint: implications for gene therapy of chronic joint diseases. Mol Ther 15: 1114–1120
Evans CH et al. (1996) Clinical trial to assess the safety, feasibility, and efficacy of transferring a potentially anti-arthritic cytokine gene to human joints with rheumatoid arthritis. Hum Gene Ther 7: 1261–1280
Evans CH et al. (2005) Gene transfer to human joints: progress toward a gene therapy of arthritis. Proc Natl Acad Sci USA 102: 8698–8703
Frisbie DD et al. (2002) Treatment of experimental equine osteoarthritis by in vivo delivery of the equine interleukin-1 receptor antagonist gene. Gene Ther 9: 12–20
Haupt JL et al. (2005) Dual transduction of insulin-like growth factor-I and interleukin-1 receptor antagonist protein controls cartilage degradation in an osteoarthritic culture model. J Orthop Res 23: 118–126
Nixon AJ et al. (2005) Gene-mediated restoration of cartilage matrix by combination insulin-like growth factor-I/interleukin-1 receptor antagonist therapy. Gene Ther 12: 177–186
Trippel SB et al. (2004) Gene-based approaches for the repair of articular cartilage. Gene Ther 11: 351–359
Mi Z et al. (2003) Adverse effects of adenovirus-mediated gene transfer of human transforming growth factor beta 1 into rabbit knees. Arthritis Res Ther 5: 132–139
Gelse K et al. (2003) Articular cartilage repair by gene therapy using growth factor-producing mesenchymal cells. Arthritis Rheum 48: 430–441
Tuli R et al. (2003) Current state of cartilage tissue engineering. Arthritis Res Ther 5: 235–238
Hollander AP et al. (2006) Maturation of tissue engineered cartilage implanted in injured and osteoarthritic human knees. Tissue Eng 12: 1787–1798
Acknowledgements
This work is supported by Deutsche Forschungsgemeinschaft (grant number DFG STE1051/2-1 to AF Steinert and U Nöth), Interdisziplinäres Zentrum für Klinische Forschung (grant number IZKF D-12/1 to U Nöth and D-23/1 to AF Steinert), and the Intramural Research Program of the National Institute of Arthritis, Musculoskeletal and Skin Diseases, National Institutes of Health (grant number Z01 AR 41131 to RS Tuan).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Nöth, U., Steinert, A. & Tuan, R. Technology Insight: adult mesenchymal stem cells for osteoarthritis therapy. Nat Rev Rheumatol 4, 371–380 (2008). https://doi.org/10.1038/ncprheum0816
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncprheum0816
This article is cited by
-
Self-organized insulin-producing β-cells differentiated from human omentum-derived stem cells and their in vivo therapeutic potential
Biomaterials Research (2023)
-
Mesenchymal stem cell implantation provides short‐term clinical improvement and satisfactory cartilage restoration in patients with knee osteoarthritis but the evidence is limited: a systematic review performed by the early‐osteoarthritis group of ESSKA‐European knee associates section
Knee Surgery, Sports Traumatology, Arthroscopy (2023)
-
Fibrin glue does not promote migration and proliferation of bone marrow derived mesenchymal stem cells in collagenic membranes: an in vitro study
Scientific Reports (2022)
-
Scintigraphic tracking of 99mTechnetium-labelled equine peripheral blood-derived mesenchymal stem cells after intravenous, intramuscular, and subcutaneous injection in healthy dogs
Stem Cell Research & Therapy (2021)
-
Bone marrow aspirate concentrate and scaffold for osteochondral lesions of the talus in ankle osteoarthritis: satisfactory clinical outcome at 10 years
Knee Surgery, Sports Traumatology, Arthroscopy (2021)